Abstract
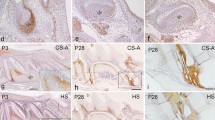
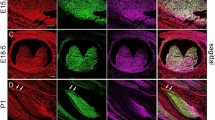
Recent studies have employed two markers, alpha-smooth muscle actin (α-SMA) and STRO-1, to detect cells with mesenchymal stem cell properties in dental pulp. The present study aimed to explore the expression profile of α-SMA and STRO-1 in intact dental pulp as well as during wound healing in adult dental pulp tissue. Healthy pulps were mechanically exposed and capped with the clinically used materials MTA (ProRoot White MTA) or Ca(OH)2 to induce a mineralized barrier at the exposed surface. After 7–42 days, the teeth were extracted and processed for immunohistochemical analysis using antibodies against α-SMA, STRO-1 and nestin (a neurogenic cytoskeletal protein expressed in odontoblasts). In normal pulp, α-SMA was detected in vascular smooth muscle cells and pericytes. Double immunofluorescent staining with STRO-1 and α-SMA showed that STRO-1 was localized in vascular smooth muscle cells, pericytes and endothelial cells, in addition to nerve fibers. During the process of dental pulp healing, numerous α-SMA-positive cells emerged at the wound margin at 14 days, and the initially formed mineralized barrier was lined with α-SMA-positive cells similar in appearance to reparative odontoblasts, some of which co-expressed nestin. STRO-1 was abundant in nerve fibers. In the advanced stage of mineralized barrier formation at 42 days, cells lining the barrier were stained with nestin, and no staining of α-SMA was detected in those cells. These observations indicate that α-SMA-positive cells temporarily appear along the wound margin during the earlier phase of mineralized barrier formation and STRO-1 is confined in vascular and neuronal elements.





Similar content being viewed by others
References
About I, Laurent-Maquin D, Lendahl U, Mitsiadis TA (2000) Nestin expression in embryonic and adult human teeth under normal and pathological conditions. Am J Pathol 157:287–295
Alliot-Licht B, Hurtrel D, Gregoire M (2001) Characterization of alpha-smooth muscle actin positive cells in mineralized human dental pulp cultures. Arch Oral Biol 46:221–228
Alongi DJ, Yamaza T, Song Y, Fouad AF, Romberg EE, Shi S, Tuan RS, Huang GT (2010) Stem/progenitor cells from inflamed human dental pulp retain tissue regeneration potential. Regen Med 5:617–631
Arora PD, Narani N, McCulloch CA (1999) The compliance of collagen gels regulates transforming growth factor-beta induction of alpha-smooth muscle actin in fibroblasts. Am J Pathol 154:871–882
Arthur A, Rychkov G, Shi S, Koblar SA, Gronthos S (2008) Adult human dental pulp stem cells differentiate toward functionally active neurons under appropriate environmental cues. Stem Cells 26:1787–1795
Brock DP, Marty-Roix R, Spector M (2002) Alpha-smooth-muscle actin in and contraction of porcine dental pulp cells. J Dent Res 81:203–208
Byers MR, Taylor PE (1993) Effect of sensory denervation on the response of rat molar pulp to exposure injury. J Dent Res 72:613–618
Chaponnier C, Gabbiani G (2004) Pathological situations characterized by altered actin isoform expression. J Pathol 204:386–395
Desmoulière A, Chaponnier C, Gabbiani G (2005) Tissue repair, contraction, and the myofibroblast. Wound Repair Regen 13:7–12
Feng J, Mantesso A, De Bari C, Nishiyama A, Sharpe PT (2011) Dual origin of mesenchymal stem cells contributing to organ growth and repair. Proc Natl Acad Sci USA 108:6503–6508
Ford TR, Torabinejad M, Abedi HR, Bakland LK, Kariyawasam SP (1996) Using mineral trioxide aggregate as a pulp-capping material. J Am Dent Assoc 127:1491–1494
Gronthos S, Zannettino AC, Graves SE, Ohta S, Hay SJ, Simmons PJ (1999) Differential cell surface expression of the STRO-1 and alkaline phosphatase antigens on discrete developmental stages in primary cultures of human bone cells. J Bone Miner Res 14:47–56
Hosoya A, Nakamura H, Ninomiya T, Yoshiba K, Yoshiba N, Nakaya H, Wakitani S, Yamada H, Kasahara E, Ozawa H (2006) Immunohistochemical localization of α-smooth muscle actin during rat molar tooth development. J Histochem Cytochem 54:1371–1378
Ishikawa Y, Ida-Yonemochi H, Nakakura-Ohshima K, Ohshima H (2012) The relationship between cell proliferation and differentiation and mapping of putative dental pulp stem/progenitor cells during mouse molar development by chasing BrdU-labeling. Cell Tissue Res 348:95–107
Kadar K, Kiraly M, Porcsalmy B, Molnar B, Racz GZ, Blazsek J, Kallo K, Szabo EL, Gera I, Gerber G, Varga G (2009) Differentiation potential of stem cells from human dental origin—promise for tissue engineering. J Physiol Pharmacol 7:167–175
Kaneko R, Akita H, Shimauchi H, Sasano Y (2009) Immunohistochemical localization of the STRO-1 antigen in developing rat teeth by light microscopy and electron microscopy. J Electron Microsc (Tokyo) 58:363–373
Kinner B, Zaleskas JM, Spector M (2002) Regulation of smooth muscle actin expression and contraction in adult human mesenchymal stem cells. Exp Cell Res 278:72–83
Király M, Porcsalmy B, Pataki A, Kádár K, Jelitai M, Molnár B, Hermann P, Gera I, Grimm WD, Ganss B, Zsembery A, Varga G (2009) Simultaneous PKC and cAMP activation induces differentiation of human dental pulp stem cells into functionally active neurons. Neurochem Int 55:323–332
Laino G, Graziano A, d’Aquino R, Pirozzi G, Lanza V, Valiante S, De Rosa A, Naro F, Vivarelli E, Papaccio G (2006) An approachable human adult stem cell source for hard-tissue engineering. J Cell Physiol 206:693–701
Martinez EF, Araújo VC, Sousa SO, Arana-Chavez VE (2007) TGF-beta1 enhances the expression of alpha-smooth muscle actin in cultured human pulpal fibroblasts: immunochemical and ultrastructural analyses. J Endod 33:1313–1318
McLachlan JL, Smith AJ, Sloan AJ, Cooper PR (2003) Gene expression analysis in cells of the dentine–pulp complex in healthy and carious teeth. Arch Oral Biol 48:273–283
Milara J, Serrano A, Peiró T, Gavaldà A, Miralpeix M, Morcillo EJ, Cortijo J (2011) Aclidinium inhibits human lung fibroblast to myofibroblast transition. Thorax. doi:10.1136/thoraxjnl-2011-200376
Miura M, Gronthos S, Zhao M, Lu B, Fisher LW, Robey PG, Shi S (2003) SHED: stem cells from human exfoliated deciduous teeth. Proc Natl Acad Sci USA 100:5807–5812
Morikawa S, Baluk P, Kaidoh T, Haskell A, Jain RK, McDonald DM (2002) Abnormalities in pericytes on blood vessels and endothelial sprouts in tumors. Am J Pathol 160:985–1000
Ning H, Lin G, Lue TF, Lin CS (2011) Mesenchymal stem cell marker Stro-1 is a 75 kd endothelial antigen. Biochem Biophys Res Commun 413:353–357
Powell DW, Mifflin RC, Valentich JD, Crowe SE, Saada JI, West AB (1999) Myofibroblasts. I. Paracrine cells important in health and disease. Am J Physiol 277:C1–C19
Racine-Samson L, Rockey DC, Bissell DM (1997) The role of alpha1beta1 integrin in wound contraction. A quantitative analysis of liver myofibroblasts in vivo and in primary culture. J Biol Chem 272:30911–30917
Ruch JV, Lesot H, Bègue-Kirn C (1995) Odontoblast differentiation. Int J Dev Biol 39:51–68
Schmidt M, Sun G, Stacey MA, Mori L, Mattoli S (2003) Identification of circulating fibrocytes as precursors of bronchial myofibroblasts in asthma. J Immunol 171:380–389
Schröder U (1985) Effects of calcium hydroxide-containing pulp-capping agents on pulp cell migration, proliferation, and differentiation. J Dent Res 64:541–548
Serini G, Bochaton-Piallat ML, Ropraz P, Geinoz A, Borsi L, Zardi L, Gabbiani G (1998) The fibronectin domain ED-A is crucial for myofibroblastic phenotype induction by transforming growth factor-beta1. J Cell Biol 142:873–881
Shi S, Gronthos S (2003) Perivascular niche of postnatal mesenchymal stem cells in human bone marrow and dental pulp. J Bone Miner Res 18:696–704
Singh SR, Hall IP (2008) Airway myofibroblasts and their relationship with airway myocytes and fibroblasts. Proc Am Thorac Soc 5:127–132
Tomasek JJ, Gabbiani G, Hinz B, Chaponnier C, Brown RA (2002) Myofibroblasts and mechano-regulation of connective tissue remodelling. Nat Rev Mol Cell Biol 3:349–363
Tomson PL, Grover LM, Lumley PJ, Sloan AJ, Smith AJ, Cooper PR (2007) Dissolution of bio-active dentine matrix components by mineral trioxide aggregate. J Dent 35:636–642
Yang X, Zhang W, van den Dolder J, Walboomers XF, Bian Z, Fan M, Jansen JA (2007) Multilineage potential of STRO-1 + rat dental pulp cells in vitro. J Tissue Eng Regen Med 2:128–135
Yoshiba N, Yoshiba K, Iwaku M, Nakamura H, Ozawa H (1994) A confocal laser scanning microscopic study of the immunofluorescent localization of fibronectin in the odontoblast layer of human teeth. Arch Oral Biol 39:395–400
Yoshiba K, Yoshiba N, Nakamura H, Iwaku M, Ozawa H (1996) Immunolocalization of fibronectin during reparative dentinogenesis in human teeth after pulp capping with calcium hydroxide. J Dent Res 75:1590–1597
Yoshiba N, Yoshiba K, Ohkura N, Hosoya A, Shigetani S, Yamanaka Y, Izumi N, Nakamura H, Okiji T (2012) Expressional alterations of fibrillin-1 during wound healing of human dental pulp. J Endod 38:177–184
Acknowledgments
This work was supported by a grant-in-aid for Scientific Research from the Ministry of Education, Science, Sports and Culture, Japan (no. 22592119).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yoshiba, N., Yoshiba, K., Ohkura, N. et al. Immunohistochemical analysis of two stem cell markers of α-smooth muscle actin and STRO-1 during wound healing of human dental pulp. Histochem Cell Biol 138, 583–592 (2012). https://doi.org/10.1007/s00418-012-0978-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-012-0978-4