Abstract
Purpose
To evaluate whether anti-drug antibodies (ADAs) are present in the ocular fluid of patients with ranibizumab-recalcitrant neovascular age-related macular degeneration (nAMD).
Methods
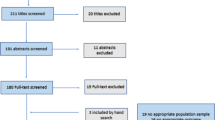
Two serum ADA-positive ranibizumab-recalcitrant patients and two serum ADA-negative controls were recruited from patients with nAMD treated with ranibizumab monotherapy. Recalcitrance was defined as persistent fluid after ≥6 monthly ranibizumab injections. Serum and aqueous humor ADAs were detected by enzyme-linked immunosorbent assay and immunoprecipitation, respectively.
Results
Two of 156 ranibizumab-treated patients were ADA-positive. The patients received six and 14 ranibizumab injections, respectively, up to 4 weeks prior to blood collection. The serum ADA concentration was estimated to be approximately 50,000 ng/mL. Neutralizing ADAs were confirmed in both samples. A specific band was detected by immunoprecipitation only in ADA-positive samples, consistent with the results of enzyme-linked immunosorbent assay. Based on an assessment of the degree of sensitivity of commercially available anti-ranibizumab antibodies, it was estimated that the immunoprecipitation method could detect ADA levels >30 ng. Nevertheless, ADAs were not detected in the aqueous humor of either the experimental or control group.
Conclusion
In the aqueous humor, ADAs are either not present or are present at a lower concentration than that which can be detected by immunoprecipitation. This presumably reflects the fact that blood ADA is the product of systemic circulation clearance through anterior elimination of intravitreal ranibizumab. Based on our results, ADAs do not return to the eye in sufficient quantities to interfere with the action of ranibizumab in the vitreous cavity.



Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Rosenfeld PJ, Brown DM, Heier JS et al (2006) Ranibizumab for neovascular age-related macular degeneration. N Engl J Med 355:1419–1431. https://doi.org/10.1056/NEJMoa054481
Brown DM, Kaiser PK, Michels M et al (2006) Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N Engl J Med 355:1432–1444. https://doi.org/10.1056/NEJMoa062655
Gupta OP, Shienbaum G, Patel AH, Fecarotta C, Kaiser RS, Regillo CD (2010) A treat and extend regimen using ranibizumab for neovascular age-related macular degeneration clinical and economic impact. Ophthalmology 117:2134–2140. https://doi.org/10.1016/j.ophtha.2010.02.032
Otsuji T, Nagai Y, Sho K et al (2013) Initial non-responders to ranibizumab in the treatment of age-related macular degeneration (AMD). Clin Ophthalmol 7:1487–1490. https://doi.org/10.2147/OPTH.S46317
Eghøj MS, Sørensen TL (2012) Tachyphylaxis during treatment of exudative age-related macular degeneration with ranibizumab. Br J Ophthalmol 96:21–23. https://doi.org/10.1136/bjo.2011.203893
Forooghian F, Cukras C, Meyerle CB, Chew EY, Wong WT (2009) Tachyphylaxis after intravitreal bevacizumab for exudative age-related macular degeneration. Retina 29:723–731. https://doi.org/10.1097/IAE.0b013e3181a2c1c3
Fassnacht-Riederle H, Becker M, Graf N, Michels S (2014) Effect of aflibercept in insufficient responders to prior anti-VEGF therapy in neovascular AMD. Graefes Arch Clin Exp Ophthalmol 252:1705–1709. https://doi.org/10.1007/s00417-014-2589-3
Heussen FM, Shao Q, Ouyang Y, Joussen AM, Müller B (2014) Clinical outcomes after switching treatment from intravitreal ranibizumab to aflibercept in neovascular age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol 252:909–915. https://doi.org/10.1007/s00417-013-2553-7
Amoaku WM, Chakravarthy U, Gale R et al (2015) Defining response to anti-VEGF therapies in neovascular AMD. Eye (Lond) 29:1397–1398. https://doi.org/10.1038/eye.2015.48
Sarao V, Parravano M, Veritti D, Arias L, Varano M, Lanzetta P (2016) Intravitreal aflibercept for choroidal neovascularization due to age-related macular degeneration unresponsive to ranibizumab therapy. Retina 36:770–777. https://doi.org/10.1097/IAE.0000000000000751
Atzeni F, Talotta R, Salaffi F et al (2013) Immunogenicity and autoimmunity during anti-TNF therapy. Autoimmun Rev 12:703–708. https://doi.org/10.1016/j.autrev.2012.10.021
De Groot AS, Scott DW (2007) Immunogenicity of protein therapeutics. Trends Immunol 28:482–490. https://doi.org/10.1016/j.it.2007.07.011
Fox EJ, Vartanian TK, Zamvil SS (2007) The immunogenicity of disease-modifying therapies for multiple sclerosis: clinical implications for neurologists. Neurologist 13:355–362. https://doi.org/10.1097/NRL.0b013e318148c08e
van Schouwenburg PA, Rispens T, Wolbink GJ (2013) Immunogenicity of anti-TNF biologic therapies for rheumatoid arthritis. Nat Rev Rheumatol 9:164–172. https://doi.org/10.1038/nrrheum.2013.4
Atiqi S, Hooijberg F, Loeff FC, Rispens T, Wolbink GJ (2020) Immunogenicity of TNF-inhibitors. Front Immunol 11:312. https://doi.org/10.3389/fimmu.2020.00312
Heier JS, Boyer DS, Ciulla TA et al (2006) Ranibizumab combined with verteporfin photodynamic therapy in neovascular age-related macular degeneration: year 1 results of the FOCUS Study. Arch Ophthalmol 124:1532–1542. https://doi.org/10.1001/archopht.124.11.1532
Bressler NM, Kim T, Oh I, Russo P, Kim MY, Woo SJ (2023) Immunogenicity with ranibizumab biosimilar SB11 (Byooviz) and reference product Lucentis and association with efficacy, safety, and pharmacokinetics: a post hoc analysis of a phase 3 randomized clinical trial. JAMA Ophthalmol 141:117–127. https://doi.org/10.1001/jamaophthalmol.2022.5403
Gaudreault J, Fei D, Rusit J, Suboc P, Shiu V (2005) Preclinical pharmacokinetics of ranibizumab (rhuFabV2) after a single intravitreal administration. Invest Ophthalmol Vis Sci 46:726–733. https://doi.org/10.1167/iovs.04-0601
Yoon CK, Oh J, Bae K, Park UC, Yu KS, Yu HG (2022) Efficacy and safety of a new ranibizumab biosimilar CKD-701 using a pro re nata treatment regimen in neovascular age-related macular degeneration: a phase 3 randomized clinical trial. PLoS One 17:e0275611. https://doi.org/10.1371/journal.pone.0275611
Shen WY, Yu MJ, Barry CJ, Constable IJ, Rakoczy PE (1998) Expression of cell adhesion molecules and vascular endothelial growth factor in experimental choroidal neovascularisation in the rat. Br J Ophthalmol 82:1063–1071. https://doi.org/10.1136/bjo.82.9.1063
Wu E, Palmer N, Tian Z et al (2008) Comprehensive dissection of PDGF-PDGFR signaling pathways in PDGFR genetically defined cells. PLoS One 3:e3794. https://doi.org/10.1371/journal.pone.0003794
Rusnati M, Presta M (2007) Fibroblast growth factors/fibroblast growth factor receptors as targets for the development of anti-angiogenesis strategies. Curr Pharm Des 13:2025–2044. https://doi.org/10.2174/138161207781039689
Stein-Streilein J (2008) Immune regulation and the eye. Trends Immunol 29:548–554. https://doi.org/10.1016/j.it.2008.08.002
Niederkorn JY (2007) The induction of anterior chamber-associated immune deviation. Chem Immunol Allergy 92:27–35. https://doi.org/10.1159/000099251
Blana SA, Musshoff F, Hoeller T, Fimmers R, Madea B (2011) Variations in vitreous humor chemical values as a result of pre-analytical treatment. Forensic Sci Int 210:263–270. https://doi.org/10.1016/j.forsciint.2011.03.023
Laude A, Cackett PD, Vithana EN et al (2010) Polypoidal choroidal vasculopathy and neovascular age-related macular degeneration: same or different disease? Prog Retin Eye Res 29:19–29. https://doi.org/10.1016/j.preteyeres.2009.10.001
Cheung GCM, Lai TYY, Gomi F, Ruamviboonsuk P, Koh A, Lee WK (2017) Anti-VEGF therapy for neovascular AMD and polypoidal choroidal vasculopathy. Asia Pac J Ophthalmol (Phila) 6:527–534. https://doi.org/10.22608/APO.2017260
Abdullah SE, Perez-Soler R (2012) Mechanisms of resistance to vascular endothelial growth factor blockade. Cancer 118:3455–3467. https://doi.org/10.1002/cncr.26540
Inai T, Mancuso M, Hashizume H et al (2004) Inhibition of vascular endothelial growth factor (VEGF) signaling in cancer causes loss of endothelial fenestrations, regression of tumor vessels, and appearance of basement membrane ghosts. Am J Pathol 165:35–52. https://doi.org/10.1016/S0002-9440(10)63273-7
Campa C, Costagliola C, Incorvaia C et al (2010) Inflammatory mediators and angiogenic factors in choroidal neovascularization: pathogenetic interactions and therapeutic implications. Mediators Inflamm 2010:546826. https://doi.org/10.1155/2010/546826
Lorés-Motta L, Riaz M, Grunin M et al (2018) Association of genetic variants with response to anti- vascular endothelial growth factor therapy in age-related macular degeneration. JAMA Ophthalmol 136:875–884. https://doi.org/10.1001/jamaophthalmol.2018.2019
Acknowledgements
The authors thank Youngjae Lee of the Korea Advanced Institute of Science and Technology for his technical advice and support.
Funding
This work was supported by Inha University Hospital Research Grant.
Author information
Authors and Affiliations
Contributions
Conceptualization: Kanghoon Lee, Hee Seung Chin; methodology: Kanghoon Lee, Soyoung Lee, Hee Seung Chin; formal analysis and investigation: Kanghoon Lee, Soyoung Lee, Soyeon Jung, Hee Seung Chin; writing—original draft preparation: Kanghoon Lee, Soyoung Lee, Soyeon Jung, Hee Seung Chin; Resources: Soyeon Jung, Hee Seung Chin. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was approved by the Ethics Committee of Inha University Hospital (No. 202106031).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent to publish
Additional informed consent was obtained from all individual participants for whom identifying information is included in this article.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 699 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lee, K., Lee, S., Jung, S. et al. Analysis of ocular fluid in patients with ranibizumab-recalcitrant neovascular age-related macular degeneration who have serum anti-ranibizumab antibodies. Graefes Arch Clin Exp Ophthalmol 261, 3581–3587 (2023). https://doi.org/10.1007/s00417-023-06146-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-023-06146-6