Abstract
Purpose
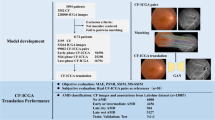
A low quality of fundus photograph with artifacts may lead to false diagnosis. Recently, a cycle-consistent generative adversarial network (CycleGAN) has been introduced as a tool to generate images without matching paired images. Therefore, herein, we present a deep learning technique that removes the artifacts automatically in a fundus photograph using a CycleGAN model.
Methods
This study included a total of 2206 anonymized retinal images including 1146 with artifacts and 1060 without artifacts. In this experiment, we applied the CycleGAN model to color fundus photographs with a pixel resolution of 256 × 256 × 3. To apply the CycleGAN to an independent dataset, we randomly divided the data into training (90%) and test (10%) datasets. Additionally, we adopted the automated quality evaluation (AQE) to assess the retinal image quality.
Results
From the results, we observed that the artifacts such as overall haze, edge haze, lashes, arcs, and uneven illumination were successfully reduced by the CycleGAN in the generated images, and the main information of the retina was essentially retained. Further, we observed that most of the generated images exhibited improved AQE grade values when compared with the original images with artifacts.
Conclusion
Thus, we could conclude that the CycleGAN technique can effectively reduce the artifacts and improve the quality of fundus photographs, and it may be beneficial for clinicians in analyzing the low-quality fundus photographs. Future studies should improve the quality and resolution of the generated image to provide a more detailed fundus photography.





Similar content being viewed by others
Data availability
All the datasets are available at Mendeley Data repositories (https://data.mendeley.com/datasets/dh2x8v6nf8).
References
Fleming AD, Philip S, Goatman KA et al (2006) Automated assessment of diabetic retinal image quality based on clarity and field definition. Invest Ophthalmol Vis Sci 47:1120–1125. https://doi.org/10.1167/iovs.05-1155
Bartling H, Wanger P, Martin L (2009) Automated quality evaluation of digital fundus photographs. Acta Ophthalmol 87:643–647. https://doi.org/10.1111/j.1755-3768.2008.01321.x
Marrugo AG, Sorel M, Sroubek F, Millán MS (2011) Retinal image restoration by means of blind deconvolution. J Biomed Opt 16:116016. https://doi.org/10.1117/1.3652709
Mora AD, Soares J, Fonseca JM (2013) A template matching technique for artifacts detection in retinal images. In: 2013 8th international symposium on image and signal processing and analysis (ISPA). pp 717–722
Gondara L (2016) Medical image denoising using convolutional denoising autoencoders. In: 2016 IEEE 16th international conference on data mining workshops (ICDMW). pp 241–246
Yoo TK, Choi JY, Seo JG et al (2019) The possibility of the combination of OCT and fundus images for improving the diagnostic accuracy of deep learning for age-related macular degeneration: a preliminary experiment. Med Biol Eng Comput 57:677–687. https://doi.org/10.1007/s11517-018-1915-z
Yoo TK, Ryu IH, Lee G et al (2019) Adopting machine learning to automatically identify candidate patients for corneal refractive surgery. Npj Digit Med 2:59. https://doi.org/10.1038/s41746-019-0135-8
Goodfellow I, Pouget-Abadie J, Mirza M, et al (2014) Generative adversarial nets. In: Advances in neural information processing systems. pp 2672–2680
Isola P, Zhu J-Y, Zhou T, Efros AA (2017) Image-to-image translation with conditional adversarial networks. In: Proceedings of the IEEE conference on computer vision and pattern recognition. pp 1125–1134
Liu Y, Khosravan N, Liu Y et al (2019) Cross-modality knowledge transfer for prostate segmentation from CT scans. In: Wang Q, Milletari F, Nguyen HV et al (eds) Domain adaptation and representation transfer and medical image learning with less labels and imperfect data. Springer International Publishing, Cham, pp 63–71
Liu Y, Guo Y, Chen W, Lew MS (2018) An extensive study of cycle-consistent generative networks for image-to-image translation. In: 2018 24th international conference on pattern recognition (ICPR). pp 219–224
Yoo TK, Choi JY, Kim HK (2020) A generative adversarial network approach to predicting postoperative appearance after orbital decompression surgery for thyroid eye disease. Comput Biol Med 103628. https://doi.org/10.1016/j.compbiomed.2020.103628
Tang C, Li J, Wang L, et al (2019) Unpaired low-dose CT denoising network based on cycle-consistent generative adversarial network with prior image information. In: Comput. Math. Methods Med. https://www.hindawi.com/journals/cmmm/2019/8639825/. Accessed 16 Jan 2020
Zhu J-Y, Park T, Isola P, Efros AA (2017) Unpaired image-to-image translation using cycle-consistent adversarial networks. In: Proceedings of the IEEE international conference on computer vision. pp 2223–2232
Carneiro T, Medeiros Da NóBrega RV, Nepomuceno T et al (2018) Performance analysis of Google CoLaboratory as a tool for accelerating deep learning applications. IEEE Access 6:61677–61685. https://doi.org/10.1109/ACCESS.2018.2874767
Suresh K (2011) An overview of randomization techniques: an unbiased assessment of outcome in clinical research. J Hum Reprod Sci 4:8–11. https://doi.org/10.4103/0974-1208.82352
Sang J, Lei Z, Li SZ (2009) Face image quality evaluation for ISO/IEC standards 19794-5 and 29794-5. In: Tistarelli M, Nixon MS (eds) Advances in biometrics. Springer, Berlin, pp 229–238
You Q, Wan C, Sun J, et al (2019) Fundus image enhancement method based on CycleGAN. In: 2019 41st annual international conference of the IEEE engineering in medicine and biology society (EMBC). pp 4500–4503
Wang S, Jin K, Lu H et al (2016) Human visual system-based fundus image quality assessment of portable fundus camera photographs. IEEE Trans Med Imaging 35:1046–1055. https://doi.org/10.1109/TMI.2015.2506902
Suzuki N, Yamane K (2012) Determination of the optimal colour space for distinguishing small retinal haemorrhages from dust artefacts. Acta Ophthalmol 90:1–2. https://doi.org/10.1111/j.1755-3768.2012.4721.x
Köhler T, Hornegger J, Mayer M, Michelson G (2012) Quality-guided denoising for low-cost fundus imaging. In: Tolxdorff T, Deserno TM, Handels H, Meinzer H-P (eds) Bildverarbeitung für die Medizin 2012: Algorithmen - Systeme - Anwendungen. Proceedings des workshops vom 18. bis 20. März 2012 in Berlin. Springer, Berlin, Heidelberg, pp 292–297
Gulshan V, Peng L, Coram M et al (2016) Development and validation of a deep learning algorithm for detection of diabetic retinopathy in retinal fundus photographs. JAMA 316:2402–2410. https://doi.org/10.1001/jama.2016.17216
Ihle SJ, Reichmuth AM, Girardin S et al (2019) Unsupervised data to content transformation with histogram-matching cycle-consistent generative adversarial networks. Nat Mach Intell 1:461–470. https://doi.org/10.1038/s42256-019-0096-2
Wolterink JM, Dinkla AM, Savenije MHF et al (2017) Deep MR to CT synthesis using unpaired data. In: Tsaftaris SA, Gooya A, Frangi AF, Prince JL (eds) Simulation and synthesis in medical imaging. Springer International Publishing, Cham, pp 14–23
Wang L, Xu X, Yu Y et al (2019) SAR-to-optical image translation using supervised cycle-consistent adversarial networks. IEEE Access 7:129136–129149
Burlina PM, Joshi N, Pacheco KD et al (2019) Assessment of deep generative models for high-resolution synthetic retinal image generation of age-related macular degeneration. JAMA Ophthalmol 137:258–264. https://doi.org/10.1001/jamaophthalmol.2018.6156
Son J, Park SJ, Jung K-H (2018) Towards accurate segmentation of retinal vessels and the optic disc in fundoscopic images with generative adversarial networks. J Digit Imaging. https://doi.org/10.1007/s10278-018-0126-3
Becker AS, Jendele L, Skopek O et al (2019) Injecting and removing suspicious features in breast imaging with CycleGAN: a pilot study of automated adversarial attacks using neural networks on small images. Eur J Radiol 120:108649. https://doi.org/10.1016/j.ejrad.2019.108649
Zhang Z, Yang L, Zheng Y (2018) Translating and segmenting multimodal medical volumes with cycle- and shape-consistency generative adversarial network. In: 2018 IEEE/CVF conference on computer vision and pattern recognition. pp 9242–9251
Author information
Authors and Affiliations
Contributions
Tae Keun Yoo and Joon Yul Choi conceived and designed this study. Tae Keun Yoo and Joon Yul Choi analyzed and described the data. Joon Yul Choi and Hong Kyu Kim collected the data. All authors contributed to the writing and approval of the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with human participants performed by any of the authors.
Code availability
We used the CoLaboratory’s CycleGAN tutorial page to develop and validate the CycleGAN model, and all these codes are available in the webpage (https://www.tensorflow.org/tutorials/generative/cyclegan). We only modified the input pipeline to import our dataset, and this code is presented in the Supplementary Material.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 310 kb).
Rights and permissions
About this article
Cite this article
Yoo, T.K., Choi, J.Y. & Kim, H.K. CycleGAN-based deep learning technique for artifact reduction in fundus photography. Graefes Arch Clin Exp Ophthalmol 258, 1631–1637 (2020). https://doi.org/10.1007/s00417-020-04709-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-020-04709-5