Abstract
Purpose
To investigate the prevalence and progression of vitreo-macular interface disorders (VMID) phenotypes and their natural history in retinitis pigmentosa (RP).
Methods
A total of 257 eyes of 145 RP patients with VMID were retrospectively evaluated. Patients were divided according to the VMID subtypes into epiretinal membranes (ERMs), vitreo-macular traction (VMT) group, and macular hole (MH). Serial eye-tracked spectral-domain optical coherence tomography (SD-OCT) and best-corrected visual acuity (BCVA) changes were analyzed for a mean follow-up of 36.95 months. The status of posterior vitreous cortex was also considered. A control group of 65 eyes belonging to 65 RP patients with no macular changes was also recruited.
Results
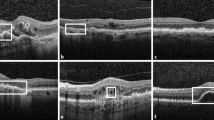
VMID and control groups had the same baseline BCVA (0.50 vs 0.44 LogMAR) and did not differ in terms of phakic status. Different VMID groups had similar BCVA at baseline (p = 0.98). ERM represented the most prevalent disorder (207/257 eyes, 80.5%), followed by 35/257 (13.6%) VMT, and 15/257 Lamellar MH (LMH) eyes (5.8%). There were no cases of full thickness MH. Throughout the 36.9 months of follow-up, BCVA decreased an average 0.09 LogMAR from 0.31 to 0.4 in VMID patients and 0.01 in controls. VMID subgroup analysis showed a significant BCVA decrease in ERM patients (− 20.29%, p < 0.001), while VMT and LMH did not change significantly. Foveal thickness also remained stable over time. Complete PVD was present in 11 eyes in ERM, VMT, and LMH.
Conclusions
Our study confirms the high prevalence of VMID in RP patients; however, only ERMs determined a significant loss of vision over 24 months. The high prevalence of VMID in RP patients suggests that macular alteration other than edema represents part of disease spectrum.






Similar content being viewed by others
References
Hartong DT, Berson EL, Dryja TP (2006) Retinitis pigmentosa. Lancet 368:1795–1809. https://doi.org/10.1016/S0140-6736(06)69740-7
Testa F, Rossi S, Colucci R, Gallo B, Di Iorio V, della Corte M, Azzolini C, Melillo P, Simonelli F (2014) Macular abnormalities in Italian patients with retinitis pigmentosa. Br J Ophthalmol 98:946–950. https://doi.org/10.1136/bjophthalmol-2013-304082
Lupo S, Grenga PL, Vingolo EM (2011) Fourier-domain optical coherence tomography and microperimetry findings in retinitis pigmentosa. Am J Ophthalmol 151:106–111. https://doi.org/10.1016/j.ajo.2010.07.026
Giusti C, Forte R, Vingolo EM (2002) Clinical pathogenesis of macular holes in patients affected by retinitis pigmentosa. Eur Rev Med Pharmacol Sci 6:45–48
Hirakawa H, Iijima H, Gohdo T, Tsukahara S (1999) Optical coherence tomography of cystoid macular edema associated with retinitis pigmentosa. Am J Ophthalmol 128:185–191
Hajali M, Fishman GA (2009) The prevalence of cystoid macular oedema on optical coherence tomography in retinitis pigmentosa patients without cystic changes on fundus examination. Eye (Lond) 23:915–919. https://doi.org/10.1038/eye.2008.110
Hajali M, Fishman GA, Anderson RJ (2008) The prevalence of cystoid macular oedema in retinitis pigmentosa patients determined by optical coherence tomography. Br J Ophthalmol 92:1065–1068. https://doi.org/10.1136/bjo.2008.138560
Jin ZB, Gan DK, Xu GZ, Nao IN (2008) Macular hole formation in patients with retinitis pigmentosa and prognosis of pars plana vitrectomy. Retina 28:610–614. https://doi.org/10.1097/IAE.0b013e31815ec341
Hagiwara A, Yamamoto S, Ogata K, Sugawara T, Hiramatsu A, Shibata M, Mitamura Y (2011) Macular abnormalities in patients with retinitis pigmentosa: prevalence on OCT examination and outcomes of vitreoretinal surgery. Acta Ophthalmol 89:e122–e125. https://doi.org/10.1111/j.1755-3768.2010.01866.x
Gandorfer A, Haritoglou C, Kampik A (2008) Toxicity of indocyanine green in vitreoretinal surgery. Dev Ophthalmol 42:69–81. https://doi.org/10.1159/000138974
Park SH, Kim YJ, Lee SJ (2016) Incidence of and risk factors for dissociated optic nerve fiber layer after epiretinal membrane surgery. Retina 36:1469–1473. https://doi.org/10.1097/IAE.0000000000000949
Kusuhara S, Matsumiya W, Imai H, Honda S, Tsukahara Y, Negi A (2014) Evaluating dissociated optic nerve fiber layer appearance using en face layer imaging produced by optical coherence tomography. Ophthalmologica 232:170–178. https://doi.org/10.1159/000364811
Chader GJ, Weiland J, Humayun MS (2009) Artificial vision: needs, functioning, and testing of a retinal electronic prosthesis. Prog Brain Res 175:317–332. https://doi.org/10.1016/S0079-6123(09)17522-2
Chang LK, Fine HF, Spaide RF, Koizumi H, Grossniklaus HE (2008) Ultrastructural correlation of spectral-domain optical coherence tomographic findings in vitreomacular traction syndrome. Am J Ophthalmol 146:121–127. https://doi.org/10.1016/j.ajo.2008.03.001
Koizumi H, Spaide RF, Fisher YL, Freund KB, Klancnik JM Jr, Yannuzzi LA (2008) Three-dimensional evaluation of vitreomacular traction and epiretinal membrane using spectral-domain optical coherence tomography. Am J Ophthalmol 145:509–517. https://doi.org/10.1016/j.ajo.2007.10.014
dell’ Omo R, Virgili G, Rizzo S, De Turris S, Coclite G, Giorgio D, dell’ Omo E, Costagliola C (2016) Role of lamellar hole-associated epiretinal proliferation in lamellar macular holes. Am J Ophthalmol. https://doi.org/10.1016/j.ajo.2016.11.007
Duker JS, Kaiser PK, Binder S, de Smet MD, Gaudric A, Reichel E, Sadda SR, Sebag J, Spaide RF, Stalmans P (2013) The International Vitreomacular Traction Study Group classification of vitreomacular adhesion, traction, and macular hole. Ophthalmology 120:2611–2619. https://doi.org/10.1016/j.ophtha.2013.07.042
Govetto A, Dacquay Y, Farajzadeh M, Platner E, Hirabayashi K, Hosseini H, Schwartz SD, Hubschman JP (2016) Lamellar macular hole: two distinct clinical entities? Am J Ophthalmol 164:99–109. https://doi.org/10.1016/j.ajo.2016.02.008
Pang CE, Spaide RF, Freund KB (2014) Epiretinal proliferation seen in association with lamellar macular holes: a distinct clinical entity. Retina 34:1513–1523. https://doi.org/10.1097/IAE.0000000000000163
Pang CE, Spaide RF, Freund KB (2015) Comparing functional and morphologic characteristics of lamellar macular holes with and without lamellar hole-associated epiretinal proliferation. Retina 35:720–726. https://doi.org/10.1097/IAE.0000000000000390
Pang CE, Maberley DA, Freund KB, White VA, Rasmussen S, To E, Matsubara JA (2016) Lamellar hole-associated epiretinal proliferation: a clinicopathologic correlation. Retina 36:1408–1412. https://doi.org/10.1097/IAE.0000000000001069
Uchino E, Uemura A, Ohba N (2001) Initial stages of posterior vitreous detachment in healthy eyes of older persons evaluated by optical coherence tomography. Arch Ophthalmol 119:1475–1479
Holladay JT (2004) Visual acuity measurements. J Cataract Refract Surg 30:287–290. https://doi.org/10.1016/j.jcrs.2004.01.014
Fusi-Rubiano W, Awad M, Manjunath R, Elaraoud I, Narendran N, Yang Y (2015) Spectrum of morphological and visual changes due to vitreomacular interface disorders encountered in a large consecutive cohort of patients. Eye (Lond) 29:397–402. https://doi.org/10.1038/eye.2014.313
Zur D, Iglicki M, Feldinger L, Schwartz S, Goldstein M, Loewenstein A, Barak A (2018) Disorganization of retinal inner layers as a biomarker for idiopathic epiretinal membrane after macular surgery-the DREAM study. Am J Ophthalmol 196:129–135. https://doi.org/10.1016/j.ajo.2018.08.037
Fishman GA, Fishman M, Maggiano J (1977) Macular lesions associated with retinitis pigmentosa. Arch Ophthalmol 95:798–803
Johnson MW (2010) Posterior vitreous detachment: evolution and complications of its early stages. Am J Ophthalmol 149:371–382 e371. https://doi.org/10.1016/j.ajo.2009.11.022
Romano MR, Comune C, Ferrara M, Cennamo G, De Cilla S, Toto L, Cennamo G (2015) Retinal changes induced by epiretinal tangential forces. J Ophthalmol 2015:372564. https://doi.org/10.1155/2015/372564
Takahashi H, Nomura Y, Tan X, Fujino Y, Kawashima H, Yanagi Y (2015) Effects of posterior vitreous detachment on aqueous humour levels of VEGF and inflammatory cytokines. Br J Ophthalmol 99:1065–1069. https://doi.org/10.1136/bjophthalmol-2014-306051
Rahi AH, Addison DJ (1983) Autoimmunity and the outer retina. Trans Ophthalmol Soc U K 103(Pt 4):428–437
Stunkel M, Bhattarai S, Kemerley A, Stone EM, Wang K, Mullins RF, Drack AV (2015) Vitritis in pediatric genetic retinal disorders. Ophthalmology 122:192–199. https://doi.org/10.1016/j.ophtha.2014.07.037
Chester GH, Blach RK, Cleary PE (1976) Inflammation in the region of the vitreous base. Pars planitis. Trans Ophthalmol Soc U K 96:151–157
Hikichi T, Akiba J, Trempe CL (1995) Prevalence of posterior vitreous detachment in retinitis pigmentosa. Ophthalmic Surg 26:34–38
Vingolo EM, Valente S, Gerace E, Spadea L, Nebbioso M (2015) Macular hole in retinitis pigmentosa patients: microincision vitrectomy with polydimethylsiloxane as possible treatment. Eye (Lond) 29:699–702. https://doi.org/10.1038/eye.2014.344
Ikeda Y, Yoshida N, Murakami Y, Nakatake S, Notomi S, Hisatomi T, Enaida H, Ishibashi T (2015) Long-term surgical outcomes of epiretinal membrane in patients with retinitis pigmentosa. Sci Rep 5:13078. https://doi.org/10.1038/srep13078
Battaglia Parodi M, La Spina C, Triolo G, Riccieri F, Pierro L, Gagliardi M, Bandello F (2016) Correlation of SD-OCT findings and visual function in patients with retinitis pigmentosa. Graefes Arch Clin Exp Ophthalmol 254:1275–1279. https://doi.org/10.1007/s00417-015-3185-x
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fragiotta, S., Rossi, T., Carnevale, C. et al. Vitreo-macular interface disorders in retinitis pigmentosa. Graefes Arch Clin Exp Ophthalmol 257, 2137–2146 (2019). https://doi.org/10.1007/s00417-019-04418-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-019-04418-8



