Abstract
Purpose
The retinal relaxing factor (RRF) is a continuously released factor from the retina that causes vasorelaxation, the identity and potential role in physiology of which remain largely unknown. Experiments were performed to find out whether the RRF-induced relaxation is influenced by serotonin, glutamate, L-cysteine, the cytochrome P450 pathway, the cyclooxygenase pathway, or oxidative stress. In addition, the sensitivity of retinal and non-retinal arteries towards the RRF was compared.
Methods
In vitro tension measurements were performed on isolated mouse femoral or bovine retinal arteries to study the vasorelaxing effect of the RRF, induced by mouse or bovine retinas.
Results
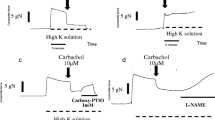
The presence of serotonin, glutamate, or L-cysteine did not alter the RRF-induced relaxation. Increasing oxidative stress by hydroquinone and diethyldithiocarbamic acid sodium salt enhanced the RRF response. Inhibition of the cytochrome P450 or the cyclooxygenase pathway did not cause any alteration. Surprisingly, the RRF-induced relaxation was enhanced by the presence of flufenamic acid or carbenoxolone. Furthermore, bringing retinal tissue in close contact with retinal or non-retinal arteries induced comparable relaxations.
Conclusions
Serotonin, glutamate, L-cysteine, the cytochrome P450, and the cyclooxygenase pathway do not influence the RRF-induced relaxation and the RRF-induced relaxation seems to be resistant to oxidative stress. The mechanism responsible for the enhanced RRF-induced relaxation in the presence of flufenamic acid or carbenoxolone remains elusive and the RRF does not show more effectivity on retinal arteries.






Similar content being viewed by others
References
Delaey C, Van de Voorde J (1998) Retinal arterial tone is controlled by a retinal-derived relaxing factor. Circ Res 83(7):714–720
Vanden Daele L, Boydens C, Pauwels B, Van de Voorde J (2017) The retinal relaxing factor: update on an enigmatic regulator of the retinal circulation. Invest Ophthalmol Vis Sci 58(3):1702–1708. https://doi.org/10.1167/iovs.16-20904
Brandstatter JH, Koulen P, Wassle H (1998) Diversity of glutamate receptors in the mammalian retina. Vis Res 38(10):1385–1397
Cornide-Petronio ME, Anadon R, Barreiro-Iglesias A, Rodicio MC (2015) Tryptophan hydroxylase and serotonin receptor 1A expression in the retina of the sea lamprey. Exp Eye Res 135:81–87. https://doi.org/10.1016/j.exer.2015.04.017
Ghai K, Zelinka C, Fischer AJ (2009) Serotonin released from amacrine neurons is scavenged and degraded in bipolar neurons in the retina. J Neurochem 111(1):1–14. https://doi.org/10.1111/j.1471-4159.2009.06270.x
Takir S, Ortakoylu GZ, Toprak A, Uydes-Dogan BS (2015) NaHS induces relaxation response in prostaglandin F(2alpha) precontracted bovine retinal arteries partially via K(v) and K(ir) channels. Exp Eye Res 132:190–197. https://doi.org/10.1016/j.exer.2015.02.002
Winther AK, Dalsgaard T, Hedegaard ER, Simonsen U (2015) Involvement of hydrogen sulfide in perivascular and hypoxia-induced inhibition of endothelin contraction in porcine retinal arterioles. Nitric Oxide 50:1–9. https://doi.org/10.1016/j.niox.2015.07.002
Newman EA (2015) Glial cell regulation of neuronal activity and blood flow in the retina by release of gliotransmitters. Philos Trans R Soc Lond Ser B Biol Sci 370(1672). https://doi.org/10.1098/rstb.2014.0195
Holmgaard K, Aalkjaer C, Lambert JD, Hessellund A, Bek T (2008) The relaxing effect of perivascular tissue on porcine retinal arterioles in vitro is mimicked by N-methyl-D-aspartate and is blocked by prostaglandin synthesis inhibition. Acta Ophthalmol 86(1):26–33. https://doi.org/10.1111/j.1600-0420.2007.01010.x
Boussery K, Delaey C, Van de Voorde J (2002) Rat retinal tissue releases a vasorelaxing factor. Invest Ophthalmol Vis Sci 43(10):3279–3286
Boussery K, Franki AS, Delaey C, Van de Voorde J (2002) A vasorelaxing factor is released from mouse retinal tissue. Ophthalmic Res 34(3):172–177. https://doi.org/10.1159/000063662
Lee YC, Chang HH, Liu CH, Chen MF, Chen PY, Kuo JS, Lee TJ (2010) Methyl palmitate: a potent vasodilator released in the retina. Invest Ophthalmol Vis Sci 51(9):4746–4753. https://doi.org/10.1167/iovs.09-5132
Jarrett SG, Boulton ME (2012) Consequences of oxidative stress in age-related macular degeneration. Mol Asp Med 33(4):399–417. https://doi.org/10.1016/j.mam.2012.03.009
Kowluru RA, Chan PS (2007) Oxidative stress and diabetic retinopathy. Exp Diabetes Res. https://doi.org/10.1155/2007/43603
Takir S, Uydes-Dogan BS, Ozdemir O (2011) Retina evokes biphasic relaxations in retinal artery unrelated to endothelium, K(V), K(ATP), K(Ca) channels and methyl palmitate. Microvasc Res 81(3):295–302. https://doi.org/10.1016/j.mvr.2011.02.007
Caputo A, Caci E, Ferrera L, Pedemonte N, Barsanti C, Sondo E, Pfeffer U, Ravazzolo R, Zegarra-Moran O, Galietta LJ (2008) TMEM16A, a membrane protein associated with calcium-dependent chloride channel activity. Science 322(5901):590–594. https://doi.org/10.1126/science.1163518
LeMaistre JL, Sanders SA, Stobart MJ, Lu L, Knox JD, Anderson HD, Anderson CM (2012) Coactivation of NMDA receptors by glutamate and D-serine induces dilation of isolated middle cerebral arteries. J Cereb Blood Flow Metab 32(3):537–547. https://doi.org/10.1038/jcbfm.2011.161
Kulkarni M, Njie-Mbye YF, Okpobiri I, Zhao M, Opere CA, Ohia SE (2011) Endogenous production of hydrogen sulfide in isolated bovine eye. Neurochem Res 36(8):1540–1545. https://doi.org/10.1007/s11064-011-0482-6
Hobbs AJ, Tucker JF, Gibson A (1991) Differentiation by hydroquinone of relaxations induced by exogenous and endogenous nitrates in non-vascular smooth muscle: role of superoxide anions. Br J Pharmacol 104(3):645–650
Kelner MJ, Bagnell R, Hale B, Alexander NM (1989) Inactivation of intracellular copper-zinc superoxide dismutase by copper chelating agents without glutathione depletion and methemoglobin formation. Free Radic Biol Med 6(4):355–360
Vanden Daele L, Boydens C, Pauwels B, Van de Voorde J (2016) Vasorelaxing effect of resveratrol on bovine retinal arteries. Invest Ophthalmol Vis Sci 57(4):1655–1661. https://doi.org/10.1167/iovs.15-18369
Metea MR, Newman EA (2006) Glial cells dilate and constrict blood vessels: a mechanism of neurovascular coupling. J Neurosci 26(11):2862–2870. https://doi.org/10.1523/JNEUROSCI.4048-05.2006
Wang MH, Brand-Schieber E, Zand BA, Nguyen X, Falck JR, Balu N, Schwartzman ML (1998) Cytochrome P450-derived arachidonic acid metabolism in the rat kidney: characterization of selective inhibitors. J Pharmacol Exp Ther 284(3):966–973
Guinamard R, Simard C, Del Negro C (2013) Flufenamic acid as an ion channel modulator. Pharmacol Ther 138(2):272–284. https://doi.org/10.1016/j.pharmthera.2013.01.012
Bencze M, Behuliak M, Vavrinova A, Zicha J (2015) Broad-range TRP channel inhibitors (2-APB, flufenamic acid, SKF-96365) affect differently contraction of resistance and conduit femoral arteries of rat. Eur J Pharmacol 765:533–540. https://doi.org/10.1016/j.ejphar.2015.09.014
Gilliam JC, Wensel TG (2011) TRP channel gene expression in the mouse retina. Vis Res 51(23–24):2440–2452. https://doi.org/10.1016/j.visres.2011.10.009
Chi Y, Li K, Yan QJ, Koizumi S, Shi LY, Takahashi S, Zhu Y, Matsue H, Takeda M, Kitamura M, Yao J (2011) Nonsteroidal anti-inflammatory drug flufenamic acid is a potent activator of AMP-activated protein kinase. J Pharmacol Exp Ther 339(1):257–266. https://doi.org/10.1124/jpet.111.183020
Ge RL, Hu L, Tai YL, Xue F, Yuan L, Wei GT, Wang Y (2013) Flufenamic acid promotes angiogenesis through AMPK activation. Int J Oncol 42(6):1945–1950. https://doi.org/10.3892/ijo.2013.1891
Rae MG, Hilton J, Sharkey J (2012) Putative TRP channel antagonists, SKF 96365, flufenamic acid and 2-APB, are non-competitive antagonists at recombinant human alpha 1 beta 2 gamma 2 GABA(A) receptors. Neurochem Int 60(6):543–554. https://doi.org/10.1016/j.neuint.2012.02.014
Hinds K, Monaghan KP, Frolund B, McGeown JG, Curtis TM (2013) GABAergic control of arteriolar diameter in the rat retina. Invest Ophthalmol Vis Sci 54(10):6798–6805. https://doi.org/10.1167/iovs.13-12362
Liu YN, Zhang HR, Huang DY, Qi JL, Xu JX, Gao HX, Du XN, Gamper N, Zhang HL (2015) Characterization of the effects of Cl- channel modulators on TMEM16A and bestrophin-1 Ca2+ activated Cl- channels. Pflugers Arch Eur J Physiol 467(7):1417–1430. https://doi.org/10.1007/s00424-014-1572-5
Qi JL, Wang Y, Liu YN, Zhang F, Guan BC, Zhang HL (2014) Development and validation of HTS assay for screening the calcium-activated chloride channel modulators in TMEM16A stably expressed CHO cells. Anal Bioanal Chem 406(6):1713–1721. https://doi.org/10.1007/s00216-013-7550-5
Yang YD, Cho HW, Koo JY, Tak MH, Cho YY, Shim WS, Park SP, Lee J, Lee B, Kim BM, Raouf R, Shin YK, Oh U (2008) TMEM16A confers receptor-activated calcium-dependent chloride conductance. Nature 455(7217):1210–U1236. https://doi.org/10.1038/nature07313
Heinze C, Seniuk A, Sokolov MV, Huebner AK, Klementowicz AE, Szijarto IA, Schleifenbaum J, Vitzthum H, Gollasch M, Ehmke H, Schroeder BC, Hubner CA (2014) Disruption of vascular Ca2+-activated chloride currents lowers blood pressure. J Clin Invest 124(2):675–686. https://doi.org/10.1172/Jci70025
Pan F, Mills SL, Massey SC (2007) Screening of gap junction antagonists on dye coupling in the rabbit retina. Vis Neurosci 24(4):609–618. https://doi.org/10.1017/S0952523807070472
Srinivas M, Spray DC (2003) Closure of gap junction channels by arylaminobenzoates. Mol Pharmacol 63(6):1389–1397. https://doi.org/10.1124/mol.63.6.1389
Tao L, Harris AL (2007) 2-aminoethoxydiphenyl borate directly inhibits channels composed of connexin26 and/or connexin32. Mol Pharmacol 71(2):570–579. https://doi.org/10.1124/mol.106.027508
Yang Y, Cao MH, Wang Q, Yuan DD, Li L, Tao L (2011) The effects of 2-aminoethoxydiphenyl borate and diphenylboronic anhydride on gap junctions composed of connexin43 in TM4 Sertoli cells. Biol Pharm Bull 34(9):1390–1397
Verselis VK, Srinivas M (2013) Connexin channel modulators and their mechanisms of action. Neuropharmacology 75:517–524. https://doi.org/10.1016/j.neuropharm.2013.03.020
Mese G, Richard G, White TW (2007) Gap junctions: basic structure and function. J Invest Dermatol 127(11):2516–2524. https://doi.org/10.1038/sj.jid.5700770
Dahl G, Qiu F, Wang JJ (2013) The bizarre pharmacology of the ATP release channel pannexin1. Neuropharmacology 75:583–593. https://doi.org/10.1016/j.neuropharm.2013.02.019
Ma WH, Hui H, Pelegrin P, Surprenant A (2009) Pharmacological characterization of pannexin-1 currents expressed in mammalian cells. J Pharmacol Exp Ther 328(2):409–418. https://doi.org/10.1124/jpet.108.146365
Sosinsky GE, Boassa D, Dermietzel R, Duffy HS, Laird DW, MacVicar B, Naus CC, Penuela S, Scemes E, Spray DC, Thompson RJ, Zhao HB, Dahl G (2011) Pannexin channels are not gap junction hemichannels. Channels (Austin) 5(3):193–197
Meens MJ, Kwak BR, Duffy HS (2015) Role of connexins and pannexins in cardiovascular physiology. Cell Mol Life Sci 72(15):2779–2792. https://doi.org/10.1007/s00018-015-1959-2
Pelegrin P, Surprenant A (2006) Pannexin-1 mediates large pore formation and interleukin-1beta release by the ATP-gated P2X7 receptor. EMBO J 25(21):5071–5082. https://doi.org/10.1038/sj.emboj.7601378
Wang J, Ma M, Locovei S, Keane RW, Dahl G (2007) Modulation of membrane channel currents by gap junction protein mimetic peptides: size matters. Am J Physiol Cell Physiol 293(3):C1112–C1119. https://doi.org/10.1152/ajpcell.00097.2007
Acknowledgements
The authors would like to thank Lies Vancraeynest and Tom Vanthuyne for the excellent technical assistance. We also thank the slaughterhouse Flanders Meat group in Zele to provide us bovine eyes.
Funding
This work is supported by the Fund of Research in Ophthalmology (FRO).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 56 kb)
Rights and permissions
About this article
Cite this article
Vanden Daele, L., Boydens, C. & Van de Voorde, J. Characterization of the retina-induced relaxation in mice. Graefes Arch Clin Exp Ophthalmol 256, 1905–1912 (2018). https://doi.org/10.1007/s00417-018-4096-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-018-4096-4