Abstract
Background
Enhanced S-cone syndrome is an orphan disease caused by mutations in the NR2E3 gene which result in an increased number of S-cones overpopulating the retina. Although the characteristic onset of enhanced S-cone syndrome can be well-documented by current ophthalmic imaging modalities, techniques such as spectral-domain optical coherence tomography (SD-OCT) and scanning laser ophthalmoscopy (SLO) fail to provide sufficient details regarding the microstructure of photoreceptors in retinal diseases. Adaptive optics (AO) provides a unique opportunity to analyze the effects of genetic mutations on photoreceptors by compensating aberrations of human eyes.
Methods
Three eyes of three young adults with enhanced S-cone syndrome were studied by clinical examination, genetic screening, fundus autofluorescence (FAF) imaging, SD-OCT, and electroretinography (ERG). Cone mosaic imaging was accomplished by an AO-SLO equipped with a dual crystal on silicon spatial light modulator. Qualitative image analyses and genetic findings were investigated in each patient.
Results
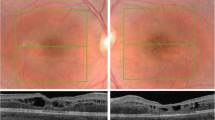
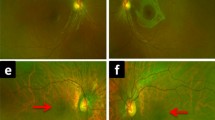
The diagnosis of patients was confirmed by ERG finding. Genetic screening confirmed the presence of two disease-causing mutations in the NR2E3 gene in each study patient, as well as identified a novel mutation (202 A > G, S68G). Fundus photograph, FAF, and SD-OCT found rosette-like lesion within the mid-periphery along the vascular arcades of the retina. In all AO-SLO images of patients, sparse distribution and asymmetric size of cone mosaic pattern were found within central retina. There were regions of dark space between groups of photoreceptors, distinguishable from shadowing and artifacts.
Conclusions
AO-SLO provided an in-depth window into the retina of live enhanced S-cone syndrome patients beyond the ability of other current imaging modalities. Dark lesions within the central retina in each patient contain structurally dysfunctional cones which account for retinal mosaic disorganization, and may predispose affected areas to other abnormalities such as rosette lesions. AO-SLO can be an efficient diagnostic tool in clinics for examining cellular-level pathologies in various retinal dystrophies.







Similar content being viewed by others
References
Haider NB, Jacobson SG, Cideciyan AV, Swiderski R, Streb LM, Searby C, Beck G, Hockey R, Hanna DB, Gorman S, Duhl D, Carmi R, Bennett J, Weleber RG, Fishman GA, Wright AF, Stone EM, Sheffield VC (2000) Mutation of a nuclear receptor gene, NR2E3, causes enhanced S-cone syndrome, a disorder of retinal cell fate. Nat Genet 24:127–131
Michaelides M, Holder G, Moore AT (2005) Inherited retinal dystrophies. In: Taylor D, Hoyt GS (eds) Pediatric ophthalmology and strabismus, 2nd edn. Elsevier Saunders, New York, pp 531–557
Fishman GA (2001) The enhanced S-cone syndrome. In: Fishman GA, Birch DG, Holder GE, Brigell MG (eds) Ophthalmology Monograph 2—Electrophysiologic Testing in Disorders of the Retina, Optic Nerve, and Visual Pathway, 2nd edition. American Academy of Ophthalmology, Singapore, p 120
Sharon D, Sandberg MA, Caruso RC, Berson EL, Dryja TP (2003) Shared mutations in NR2E3 in enhanced S-cone syndrome, Goldmann–Favre syndrome, and many cases of clumped pigmentary retinal degeneration. Arch Ophthalmol 121:1316–1323
Hayashi T, Gekka T, Goto-Omoto S, Takeuchi T, Kubo A, Kitahara K (2005) Novel NR2E3 mutations (R104Q, R334G) associated with a mild form of enhanced S-cone syndrome demonstrate compound heterozygosity. Ophthalmology 112:2115
Milam AH, Rose L, Cideciyan AV, Barakat MR, Tang WX, Gupta N, Aleman TS, Wright AF, Stone EM, Sheffield VC, Jacobson SG (2002) The nuclear receptor NR2E3 plays a role in human retinal photoreceptor differentiation and degeneration. Proc Natl Acad Sci USA 99:473–478
Fishman GA, Peachey NS (1989) Rod–cone dystrophy associated with a rod system electroretinogram obtained under photopic conditions. Ophthalmology 96:913–918
Kinori M, Pras E, Kolker A, Ferman-Attar G, Moroz I, Moisseiev J, Bandah-Rozenfeld D, Mizrahi-Meissonnier L, Sharon D, Rotenstreich Y (2011) Enhanced S-cone function with preserved rod function: a new clinical phenotype. Mol Vis 17:2241–2247
Yoon MK, Roorda A, Zhang Y, Nakanishi C, Wong LJ, Zhang Q, Gillum L, Green A, Duncan JL (2009) Adaptive optics scanning laser ophthalmoscopy images in a family with the mitochondrial DNA T8993C mutation. Invest Ophthalmol Vis Sci 50:1838–1847
Liang J, Williams DR, Miller DT (1997) Supernormal vision and high-resolution retinal imaging through adaptive optics. J Opt Soc Am A Opt Image Sci Vis 14:2884–2892
Roorda A, Romero-Borja F, Donnelly Iii W, Queener H, Hebert T, Campbell M (2002) Adaptive optics scanning laser ophthalmoscopy. Opt Express 10:405–412
Roorda A, Williams DR (1999) The arrangement of the three cone classes in the living human eye. Nature 397:520–522
Roorda A, Williams DR (2002) Optical fiber properties of individual human cones. J Vis 2:404–412
Pallikaris A, Williams DR, Hofer H (2003) The reflectance of single cones in the living human eye. Invest Ophthalmol Vis Sci 44:4580–4592
Zhang Y, Poonja S, Roorda A (2006) MEMS-based adaptive optics scanning laser ophthalmoscopy. Opt Lett 31:1268–1270
Zhang Y, Roorda A (2006) Evaluating the lateral resolution of the adaptive optics scanning laser ophthalmoscope. J Biomed Opt 11:014002
Merino D, Duncan JL, Tiruveedhula P, Roorda A (2011) Observation of cone and rod photoreceptors in normal subjects and patients using a new generation adaptive optics scanning laser ophthalmoscope. Biomed Opt Express 2:2189–2201
Duncan JL, Zhang Y, Gandhi J, Nakanishi C, Othman M, Branham KE, Swaroop A, Roorda A (2007) High-resolution imaging with adaptive optics in patients with inherited retinal degeneration. Invest Ophthalmol Vis Sci 48:3283–3291
Wolfing JI, Chung M, Carroll J, Roorda A, Williams DR (2006) High-resolution retinal imaging of cone-rod dystrophy. Ophthalmology 113:1014–1019
Choi SS, Doble N, Hardy JL, Jones SM, Keltner JL, Olivier SS, Werner JS (2006) In vivo imaging of the photoreceptor mosaic in retinal dystrophies and correlations with visual function. Invest Ophthalmol Vis Sci 47:2080–2092
Chen Y, Ratnam K, Sundquist SM, Lujan B, Ayyagari R, Gudiseva VH, Roorda A, Duncan JL (2011) Cone photoreceptor abnormalities correlate with vision loss in patients with stargardt disease. Invest Ophthalmol Vis Sci 52:3281–3292
Futoshi H, Koji N, Ken-ichi S, Yasuyuki N (2011) A compact adaptive optics scanning laser ophthalmoscope with high-efficiency wavefront correction using dual liquid crystal on silicon–spatial light modulator. Proc SPIE 7885:788515
Marmor MF, Fulton AB, Holder GE, Miyake Y, Brigell M, Bach M (2009) International society for clinical electrophysiology of vision. ISCEV standard for full-field clinical electroretinography–2008 update. Doc Ophthalmol 1:69–77
Audo I, Michaelides M, Robson AG, Hawlina M, Vaclavik V, Sandbach JM, Neveu MM, Hogg CR, Hunt DM, Moore AT, Bird AC, Webster AR, Holder GE (2008) Phenotypic variation in enhanced S-cone syndrome. Invest Ophthalmol Vis Sci 49:2082–2093
Escher P, Gouras P, Roduit R, Tiab L, Bolay S, Delarive T, Chen S, Tsai CC, Hayashi M, Zernant J, Merriam JE, Mermod N, Allikmets R, Munier FL, Schorderet DF (2009) Mutations in NR2E3 can cause dominant or recessive retinal degenerations in the same family. Hum Mutat 30:342–351
Wang NK, Fine HF, Chang S, Chou CL, Cella W, Tosi J, Lin CS, Nagasaki T, Tsang SH (2009) Cellular origin of fundus autofluorescence in patients and mice with a defective NR2E3 gene. Br J Ophthalmol 93:1234–1240
Marmor MF, Jacobson SG, Foerster MH, Kellner U, Weleber RG (1990) Diagnostic clinical findings of a new syndrome with night blindness, maculopathy, and enhanced S-cone sensitivity. Am J Ophthalmol 110:124–134
Pachydaki SI, Klaver CC, Barbazetto IA, Roy MS, Gouras P, Allikmets R, Yannuzzi LA (2009) Phenotypic features of patients with NR2E3 mutations. Arch Ophthalmol 127:71–75
Haider NB, Naggert JK, Nishina PM (2001) Excess cone cell proliferation due to lack of a functional NR2E3 causes retinal dysplasia and degeneration in rd7/rd7 mice. Hum Mol Genet 10:1619–1626
Ooto S, Hangai M, Takayama K, Sakamoto A, Tsujikawa A, Oshima S, Inoue T, Yoshimura N (2011) High-resolution imaging of the photoreceptor layer in epiretinal membrane using adaptive optics scanning laser ophthalmoscopy. Ophthalmology 118:873–881
Bessho K, Fujikado T, Mihashi T, Yamaguchi T, Nakazawa N, Tano Y (2008) Photoreceptor images of normal eyes and of eyes with macular dystrophy obtained in vivo with an adaptive optics fundus camera. Jpn J Ophthalmol 52:380–385
Mustafi D, Kevany BM, Genoud C, Okano K, Cideciyan AV, Sumaroka A, Roman AJ, Jacobson SG, Engel A, Adams MD, Palczewski K (2011) Defective photoreceptor phagocytosis in a mouse model of enhanced S-cone syndrome causes progressive retinal degeneration. FASEB J 25:3157–3176
Baraas RC, Carroll J, Gunther KL, Chung M, Williams DR, Foster DH, Neitz M (2007) Adaptive optics retinal imaging reveals S-cone dystrophy in tritan color-vision deficiency. J Opt Soc Am A Opt Image Sci Vis 24:1438–1447
Marmor MF, Tan F, Sutter EE, Bearse MA Jr (1999) Topography of cone electrophysiology in the enhanced S-cone syndrome. Invest Ophthalmol Vis Sci 40:1866–1873
Acknowledgement
Publication of this article was supported by multiple grants. These included the following: a Core Support for Vision Research grant (P30EY019007: SHT, RA) and National Eye Institute of The National Institutes of Health (Bethesda, MD, USA) grants (EY018213:SHT, EY019861: RA, SHT). Funds were also received from the Foundation Fighting Blindness (Owings Mills, MD, USA), as were unrestricted funds from Research to Prevent Blindness Inc, (New York, NY, USA). Dr. Tsang is a fellow of the Burroughs–Wellcome Program in Biomedical Sciences, and has been supported by the following grants: the Bernard Becker Association of University Professors in Ophthalmology Research to Prevent Blindness Award, the Foundation Fighting Blindness, Dennis W. Jahnigen Award of the American Geriatrics Society, Joel Hoffman Scholarship, and the Crowley Family Fund and Barbara & Donald Jonas Family Funds. The authors have no financial support or conflicts of interest from the materials or methods used in this investigation. Individual author contributions: Design and conduct of study (SPP, IHH, SHT); management, analysis, and interpretation of data (SPP, IHH, WL, SY, SHT); and preparation, review and/or approval of manuscript (SPP, WL, SC, RA, JH, SHT). The authors thank Canon Inc. (Tokyo, Japan) for their technical support with the AO-SLO system, and Elena N. Bukanova for technical support and contributions with genetic analyses. Further gratitude is extended to Drs. Lawrence Yannuzzi and Irene Barbazetto for patient referral.
Financial disclosure
The authors indicated no financial support or conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, S.P., Hong, I.H., Tsang, S.H. et al. Disruption of the human cone photoreceptor mosaic from a defect in NR2E3 transcription factor function in young adults. Graefes Arch Clin Exp Ophthalmol 251, 2299–2309 (2013). https://doi.org/10.1007/s00417-013-2296-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-013-2296-5