Abstract
Purpose
To report the clinical and electrophysiological features of cone dystrophy with supernormal rod response (CDSRR).
Methods
Retrospective cohort study of 15 unrelated patients (nine males and six females, median age 16, range 5–47 years) diagnosed with CDSRR by clinical examination, full-field electroretinography (ERG) and genetic testing.
Observations
History, ophthalmic examination including near vision, color vision and contrast sensitivity assessment, multimodal retinal imaging and ERG. Genetic testing was done for all patients using next-generation sequencing.
Results
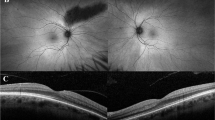
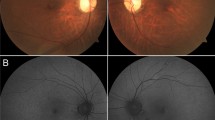
The rate of consanguinity was 86.7%. Color vision was defective in 56.3%. Near vision was defective in all patients (mean 20/160). Contrast sensitivity was affected in all patients at low contrast of 2.5%. A parafoveal ring of increased autofluorescence imaging was seen in most patients (75%). Supernormal mixed maximal response b-wave was seen bilaterally in 63% of patients (and high normal in 37%). Rod dysfunction with prolonged rod b-wave latency was detected in all. The 30-Hz flicker response was more reduced and delayed compared to the single-flash cone response. A novel homozygous missense variant c.530G>C (p.Cys177Ser) in KCNV2 was detected in one patient, the nonsense homozygous mutation c.427G>T (p.Glu143*) was found in 13 patients, and the nonsense c.159C>G (p.Tyr53*) was found in one patient.
Conclusion
This is the largest cohort of CDSRR from a single ethnic background. Rod dysfunction and reduced 30-Hz flicker response were demonstrated in all patients. In contrast to previous descriptions in the literature, a supernormal combined dark-adapted rod-cone ERG was present in the majority of the patients at standard stimulus intensity. Considering the consistent genotype and the demonstration of likely pathogenic genetic variants in all the patients, we argue that the combination of delayed rod b-wave and subnormal flicker response strongly suggests the diagnosis of CDSRR.





Similar content being viewed by others
References
Gouras P, Eggers HM, MacKay CJ (1983) Cone dystrophy, nyctalopia, and supernormal rod responses. A new retinal degeneration. Arch Ophthalmol 101(5):718–724
Michaelides M, Holder GE, Webster AR, Hunt DM, Bird AC, Fitzke FW et al (2005) A detailed phenotypic study of “cone dystrophy with supernormal rod ERG”. Br J Ophthalmol 89(3):332–339
Wu H, Cowing JA, Michaelides M, Wilkie SE, Jeffery G, Jenkins SA et al (2006) Mutations in the gene KCNV2 encoding a voltage-gated potassium channel subunit cause “cone dystrophy with supernormal rod electroretinogram” in humans. Am J Hum Genet 79(3):574–579
Wissinger B, Dangel S, Jagle H, Hansen L, Baumann B, Rudolph G et al (2008) Cone dystrophy with supernormal rod response is strictly associated with mutations in KCNV2. Invest Ophthalmol Vis Sci 49(2):751–757
Alexander KR, Fishman GA (1984) Supernormal scotopic ERG in cone dystrophy. Br J Ophthalmol 68(2):69–78
Stockman A, Henning GB, Michaelides M, Moore AT, Webster AR, Cammack J et al (2014) Cone dystrophy with “supernormal” rod ERG: psychophysical testing shows comparable rod and cone temporal sensitivity losses with no gain in rod function. Investig Ophthalmol Vis Sci 55(2):832–840
Vincent A, Robson AG, Holder GE (2013) Pathognomonic (diagnostic) ERGs. A review and update. Retina 33(1):5–12
Khan AO, Alrashed M, Alkuraya FS (2012) ‘Cone dystrophy with supranormal rod response’ in children. Br J Ophthalmol 96(3):422–426
McCulloch DL, Marmor MF, Brigell MG, Hamilton R, Holder GE, Tzekov R et al (2015) ISCEV Standard for full-field clinical electroretinography (2015 update). Doc Ophthalmol 131(1):81–83
Fujinami K, Tsunoda K, Nakamura N, Kato Y, Noda T, Shinoda K et al (2013) Molecular characteristics of four Japanese cases with KCNV2 retinopathy: report of novel disease-causing variants. Mol Vis 19:1580–1590
Robson AG, Webster AR, Michaelides M, Downes SM, Cowing JA, Hunt DM et al (2010) Cone dystrophy with supernormal rod electroretinogram: a comprehensive genotype/phenotype study including fundus autofluorescence and extensive electrophysiology. Retina 30(1):51–62
Funding
No funding was received for this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Statement of human rights
All procedures performed in studies involving human participants were in accordance with the ethical standards of the KKESH IRB and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Statement on the welfare of animals
No animals were involved in this research.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Abdelkader, E., Yasir, Z.H., Khan, A.M. et al. Analysis of retinal structure and function in cone dystrophy with supernormal rod response. Doc Ophthalmol 141, 23–32 (2020). https://doi.org/10.1007/s10633-020-09748-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10633-020-09748-1