Abstract
Objectives
This study was designed to assess survival and identify prognostic factors for liver metastases diagnosed by systematic screening in uveal melanoma patients.
Methods
Among 602 consecutive patients treated over 10 years for uveal melanoma and followed by systematic semi-annual hepatic screening (abdominal ultrasonography), 63 (10.5%) developed liver metastases; these patients form the basis of this study. Factors including patient demographics, characteristics of the uveal tumor, metastasis-free interval, severity of liver metastatic involvement, and treatments of metastases were studied retrospectively regarding their prognostic value, using univariate (Kaplan-Meier method) and multivariate (Cox model) analyses.
Results
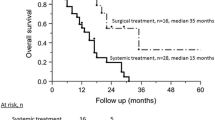
Thirty-five patients (55.6% of the metastatic population) received systemic chemotherapy or best supportive care only; 14 patients (22.2% of the metastatic population) diagnosed with diffuse liver involvement had cytoreductive surgery and intra-arterial chemotherapy; 14 (22.2% of the metastatic population) had complete surgical removal of liver metastases followed by postoperative intra-arterial chemotherapy. No significant surgical complications were experienced. The median overall survival after diagnosis of liver metastases was 15 months. It reached 25 months for selected patients with complete resection (P=0.0002). In this cohort of 63 patients, ten or fewer preoperatively diagnosed metastases and primary uveal melanoma not involving the ciliary body were independently associated with better prognosis.
Conclusions
This study suggests that selected patients with screened liver metastases from uveal melanoma may benefit from aggressive treatment, including surgery. The two independent favorable prognostic factors are fewer than ten metastases at screening and the absence of ciliary body involvement.



Similar content being viewed by others
References
Aoyama T, Mastrangelo MJ, Berd D, Nathan FE, Shields CL, Shields JA, Rosato EL, Rosato FE, Sato T (2000) Protracted survival after resection of metastatic uveal melanoma. Cancer 89:1561–1568
Bedikian AY, Kantarjian H, Young SE, Bodey GP (1981) Prognosis in metastatic choroidal melanoma. South Med J 74:574–577
Bedikian AY, Legha SS, Mavligit G, Carrasco CH, Khorana S, Plager C, Papadopoulos N, Benjamin RS (1995) Treatment of uveal melanoma metastatic to the liver: a review of the M. D. Anderson Cancer Center experience and prognostic factors. Cancer 76:1665–1670
Cox D (1972) Regression models and life-tables (with discussions). J R Statist Soc Series B 34:187–220
Eskelin S, Pyrhonen S, Hahka-Kemppinem M, Tuomaala S, Kivela T (2003) A prognostic model and staging for metastatic uveal melanoma. Cancer 97:465–475
Gragoudas ES, Egan KM, Seddon JM, Glynn RJ, Walsh SM, Finn SM, Munzenrider JE, Spar MD (1991) Survival of patients with metastases from uveal melanoma. Ophthalmology 98:383–389; discussion 90
Gragoudas ES, Seddon JM, Egan KM, Glynn RJ, Goitein M, Munzenrider J, Verhey L, Urie M, Koehler A (1988) Metastasis from uveal melanoma after proton beam irradiation. Ophthalmology 95:992–999
Gragoudas ES, Seddon JM, Egan KM, Polivogianis L, Hsieh CC, Goitein M, Verhey L, Munzenrider J, Austin-Seymour M, Urie M et al (1986) Prognostic factors for metastasis following proton beam irradiation of uveal melanomas. Ophthalmology 93:675–680
Kaplan E, Meier P (1958) Non-parametric estimation from incomplete observation. J Am Stat Assoc 53:457–481
Karakousis CP, Temple DF, Moore R, Ambrus JL (1983) Prognostic parameters in recurrent malignant melanoma. Cancer 52:575–579
Kath R, Hayungs J, Bornfeld N, Sauerwein W, Hoffken K, Seeber S (1993) Prognosis and treatment of disseminated uveal melanoma. Cancer 72:2219–2223
Kodjikian L, Roy P, Rouberol F, Chauvel P, Jean-Louis B, Garweg J, Little R, Sasco A, Grange J (2004) Survival after proton-beam irradiation of uveal melanomas. Am J Ophthalmol 137:1002–1010
Leyvraz S, Spataro V, Bauer J, Pampallona S, Salmon R, Dorval T, Meuli R, Gillet M, Lejeune F, Zografos L (1997) Treatment of ocular melanoma metastatic to the liver by hepatic arterial chemotherapy. J Clin Oncol 15:2589–2595
Li W, Gragoudas ES, Egan KM (2000) Metastatic melanoma death rates by anatomic site after proton beam irradiation for uveal melanoma. Arch Ophthalmol 118:1066–1070
McLean IW, Foster WD, Zimmerman LE (1982) Uveal melanoma: location, size, cell type, and enucleation as risk factors in metastasis. Hum Pathol 13:123–132
McLean IW, Foster WD, Zimmerman LE (1977) Prognostic factors in small malignant melanomas of choroid and ciliary body. Arch Ophtalmol 95:48–58
Oken M, Creech R, Tormey D (1982) Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 5:649–655
Patel JK, Didolkar MS, Pickren JW, Moore RH (1978) Metastatic pattern of malignant melanoma. A study of 216 autopsy cases. Am J Surg 135:807–810
Peto R, Peto J (1972) Asymptotically efficient rank invariant test procedures. J R Statist Soc Series A 135:185–206
Prescher G, Bornfeld N, Hirche H, Horsthemke B, Jockel KH, Becher R (1996) Prognostic implications of monosomy 3 in uveal melanoma. Lancet 347:1222–1225
Pyrhonen S (1998) The treatment of metastatic uveal melanoma. Eur J Cancer 34(Suppl 3):S27–S30
Raivio I (1977) Uveal melanoma in Finland. An epidemiological, clinical, histological and prognostic study. Acta Ophthalmol Suppl 133:1–64
Rajpal S, Moore R, Karakousis CP (1983) Survival in metastatic ocular melanoma. Cancer 52:334–336
Rouberol F, Roy P, Kodjikian L, Gérard J, Jean-Louis B, Grange J (2004) Survival, anatomic and functional long-term results in choroidal and ciliary body melanoma after ruthenium brachytherapy (a 15 years experience with betarays). Am J Ophthalmol 137:893–900
Salmon RJ, Levy C, Plancher C, Dorval T, Desjardins L, Leyvraz S, Pouillart P, Schlienger P, Servois V, Asselain B (1998) Treatment of liver metastases from uveal melanoma by combined surgery-chemotherapy. Eur J Surg Oncol 24:127–130
Seddon JM, Albert DM, Lavin PT, Robinson N (1983) A prognostic factor study of disease-free interval and survival following enucleation for uveal melanoma. Arch Ophthalmol 101:1894–1899
Seregard S (1996) Posterior uveal melanoma. The Swedish perspective. Acta Ophthalmol Scand 74:315–329
Shammas HF, Blodi FC (1977) Prognostic factors in choroidal and ciliary body melanomas. Arch Ophthalmol 95:63–69
Shields J, Shields C (1992) Introduction to melanocytic tumors of the uvea. In: Shields J, Shields C (eds) Intraocular tumors; a text and atlas. Williams & Wilkins, Philadelphia
Singh AD, Wang MX, Donoso LA, Shields CL, Potter PD, Shields JA, Elston RC, Fijal B (1996) Familial uveal melanoma, III. Is the occurrence of familial uveal melanoma coincidental? Arch Ophthalmol 114:1101–1104
Sobin L, Wittekind C (2002) TNM classification of malignant tumours. 6th ed. Wiley-Liss, Genève
The Collaborative Ocular Melanoma Study (2001) Assessment of metastatic disease status at death in 435 patients with large choroidal melanoma in the Collaborative Ocular Melanoma Study (COMS): COMS report no. 15. Arch Ophthalmol 119:670–676
The Collaborative Ocular Melanoma Study Group (1997) Factors predictive of growth and treatment of small choroidal melanoma: COMS report no. 5. Arch Ophthalmol 115:1537–1544
Vidal JL, Bacin F, Albuisson E, Rozan R, Desjardins L, D’Hermies F, Grange JD, Chauvel P, Caujolle JP, Sahel J et al (1995) “Melanoma 92”. Epidemiological study of uveal melanoma in France. J Fr Ophtalmol 18:520–528
Author information
Authors and Affiliations
Corresponding author
Additional information
Laurent Kodjikian had full access to all the data and takes responsibility for the integrity of the data in the study and the accuracy of the data analysis
Rights and permissions
About this article
Cite this article
Kodjikian, L., Grange, JD., Baldo, S. et al. Prognostic factors of liver metastases from uveal melanoma. Graefe's Arch Clin Exp Ophthalmo 243, 985–993 (2005). https://doi.org/10.1007/s00417-005-1188-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-005-1188-8