Abstract
Background
Cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) is the most common monogenic form of stroke and is characterised by early onset stroke and dementia. Most strokes are lacunar ischaemic strokes, but intracerebral haemorrhage (ICH) has also been reported, although there are limited published data on its frequency and characteristics.
Methods
A retrospective review of a prospectively recruited CADASIL register from the British National Referral clinic was performed to identify acute ICH cases and their characteristics. In addition, a systematic review of ICH in CADASIL was performed. MEDLINE (Pubmed), Embase, and Web of Science were searched for articles published from inception until 31/05/2023.
Results
Ten cases of ICH were identified from the National clinic register of 516 symptomatic patients, giving an estimated point prevalence of 1.9%. An additional 119 cases were identified from the systematic review, comprising 129 cases and 142 ICH events in total. Including all identified cases, the mean age at onset of ICH was 56.6 ± 15.7 (SD) years, and 74 (57.4%) were male. ICH was the first manifestation of the disease in 32 patients (38.1%), and ICH recurrence occurred in 16 (12.4%). Most ICHs were subcortical, with the thalamus, 58 (40.8%), and basal ganglia, 34 (23.9%), being the commonest sites. Anticoagulation, but not antiplatelet agents, was associated with an increased risk of ICH (20.0% vs. 1.9%, p = 0.006).
Conclusions
ICH is a relatively rare manifestation of CADASIL, occurring in about 2% of symptomatic cases. Most of the haemorrhages occurred in the subcortical regions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) is the most common monogenic form of stroke, characterised by the early onset of ischaemic stroke and dementia. It is caused by a mutation in the NOTCH3 gene, which usually changes the number of cysteine residues within the extracellular epidermal growth factor-like repeats (EGFr) in the NOTCH3 receptor [1]. Common manifestations of CADASIL include stroke, usually an ischaemic stroke of lacunar subtype, migraine with aura, psychiatric syndromes, vascular cognitive impairment, and dementia.
Until recently, intracerebral haemorrhage had been little described in CADASIL. Several ICH cases in CADASIL have been reported, especially in Asian populations [2]. However, the published data on the prevalence and characteristics of ICH in CADASIL are still limited. Therefore, to better understand ICH in CADASIL, we identified cases of ICH in a prospective CADASIL register to determine its prevalence and characteristics. We also performed a systematic review to identify previously published cases and analysed clinical features, risk factors, and outcomes in our case series combined with the previously published cases.
Methods
Case series
Data acquisition
We performed a retrospective review of a prospectively collected CADASIL register from the CADASIL National Referral clinic in Cambridge (from 2001 to 2023) and from the UK Familial Small Vessel Disease Study (from 2016 to 2023), which has been recruiting patients with monogenic small vessel disease (SVD) from 6 stroke centres in the UK. Patients’ data were collected at the first study encounter and follow-up visits using standardized forms. All CADASIL patients were diagnosed with a typical cysteine-changing NOTCH3 mutation, and only symptomatic patients were included in the analysis. ICH was defined as an acute neurological syndrome with a confirmed presence of intraparenchymal blood by either computed tomography (CT) or magnetic resonance imaging (MRI).
All available imaging studies were reviewed to identify evidence of ICH lesions, complications, and radiological changes, including white matter lesions, lacunes, and cerebral microbleeds (CMBs). As per STRIVE criteria [3], lacune was defined as a subcortical infarct between 3 and 15 mm in diameter. CMB was defined as a small area, not larger than 10 mm in diameter, of signal void with associated blooming seen on T2*-weighted or susceptibility-weighted MRI sequences. Scans at the time of ICH were available in 7 patients and were graded individually. ICH details of the remaining 3 were extracted from the original radiology reports and, in addition, in a post-acute gradient echo sequence in one (A6).
Statistical analysis
Categorical variables were presented in the number of cases and percentage (%), and continuous variables were displayed by mean and standard deviation (SD). Baseline characteristics between CADASIL patients with and without ICH were compared using Student’s t test and Pearson’s chi-squared test as appropriate. A p value of less than 0.05 was considered significant. An estimated point prevalence of ICH in CADASIL patients was calculated in percentage (%) within the prospective cohort. All analyses were carried out using the R software version 4.3.1.
Systematic review
Search strategy
The study protocol was pre-registered on PROSPERO (CRD42023425877), provided by the University of York, United Kingdom. We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Statement guidelines, 2020 [4]. MEDLINE (Pubmed), Embase, and Web of Science were searched to identify studies published from inception until 31/05/2023. Search terms included "cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy" OR "CADASIL", AND, "hemorrhage" OR "hemorrhages" OR "haemorrhage" OR "haemorrhages" OR "bleed", and were refined according to each database. In addition, reference lists of the relevant articles were also searched.
Study selection and eligibility criteria
Studies were deemed eligible if they met the following criteria: (1) CADASIL diagnosed by one of the following criteria: confirmed NOTCH3 mutation, the presence of granular osmiophilic material (GOM) from skin biopsy, or known family mutation with clinical features; (2) original human data; and (3) availability of acute ICH details from patients’ history or scans. Asymptomatic haemorrhagic lesions were not included. Quality was assessed using the Joanna Briggs Institute (JBI) critical appraisal checklists [5, 6]. Regardless of language, all studies that fulfilled the criteria were included. In case there was a potential of data overlapping between studies, we contacted the corresponding authors to get the original patient data.
Data extraction and analysis
Demographic data extracted included age, sex, mutation detail, history of any CADASIL features, radiographic changes due to CADASIL, vascular risk factors, and antithrombotic medication used. Regarding the ICH detail, clinical features, radiological characteristics, and location of ICH were extracted. The cases, including demographics, risk factor details, and clinical and radiographic features identified from the systemic review, were pooled with the UK case series for the combined analysis.
Results
Case series
A total of 544 CADASIL patients were identified from the register. Of these, 516 were symptomatic and had typical heterozygous cysteine-changing mutations and were included in the analysis. We excluded 26 who were asymptomatic and diagnosed on predictive testing, one with a compound heterozygous mutation, and one with a homozygous mutation. The data from 516 symptomatic CADASIL patients were available, of whom ten patients had suffered from ICH, giving an ICH point prevalence of 1.9%. One patient also suffered one ICH recurrence. The mean age at ICH was 53.3 ± 7.6 (SD) years. In two cases, ICH was the presenting feature of CADASIL. There was no difference in the proportion of those with a mutation in EGFr 1–6, which was previously associated with increased ischaemic stroke risk [7], between those with and without ICH (50.0% vs. 79.4%, p value = 0.112). In those cases with ICH, there was a nonsignificant trend to a higher history of hypertension (50.0% vs. 23.9%, p value = 0.126), and a significant increase of anticoagulant use (20.0% vs. 1.9%, p value = 0.006), but not of antiplatelet agents. Details of individual patients with ICH are shown in Table 1. Patients’ mutation details and differences in demographic and clinical features of those with and without ICH are available in the online resources (Table S1, S2).
Six patients recovered to a modified Rankin score (mRS) of less than 2, one had a mRS of 4, while two died from the ICH. Outcome details were not available in one patient (A10). One patient (A7) had a recurrent ICH at the left medial temporal cortex, occurred 14 days after he had a brain biopsy at that region due to the suspicion of a primary brain tumour, which showed gliotic changes.
On brain imaging, the location of the ICH was thalamic in 3 patients, caudate in 1, basal ganglia in 1, medial temporal cortex in 3, subcortical white matter in 1, cerebellum in 1, and brainstem in 1. Intraventricular extension was observed in 5 (45.5%), most of which were deep ICHs. Figure 1 shows examples of imaging characteristics of ICH in two patients. Out of the 8 patients with original gradient echo or susceptibility scans available for review, subcortical and/or infratentorial CMBs were present in 4 patients.
A–C Brain imaging findings of patient A1. A non-contrast CT scan A, B revealed a left caudate haemorrhage with intraventricular extension. T2-weighted MRI sequences C demonstrated diffuse WMH with bilateral temporal pole involvement. D–F Brain imaging findings of patient A4. T2-weighted MRI D demonstrated diffuse WMH involving bilateral external capsules. Susceptibility-weighted E and T1-weighted F MRI sequences revealed ICH at the subcortical white matter of right superior parietal lobule
Systematic review
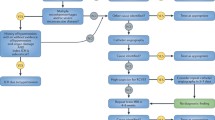
Through the search, a total of 756 articles were identified. Fifty-five articles were eligible for full-text assessment and 25 were excluded for four reasons, summarised in Fig. 2. We contacted the corresponding authors regarding possible data overlapping issues in 8 articles [8,9,10,11,12,13,14,15]. Following this, we included 4 of them, two which had been confirmed to have no overlapping data [11, 15] and for 2 where there was overlap but we were provided with the individual patient data [13, 14]. We received no reply for one potential overlap [8,9,10,11] so we omitted smaller studies from this group [8,9,10] and only included the larger study [11].
Thirty studies were eligible, including 21 case reports [16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36], 7 case series [13, 14, 37,38,39,40,41], and 2 observational studies [11, 15], comprising 119 CADASIL patients with history of ICH. Half of the studies were published in Asia, which consisted of 98 cases (76.0%). The details of each study are summarised in Table 2. The details of the quality assessment and mutation and diagnostic means in each study are available in the online resources (Table S3–S6).
Combined analysis of case series and systematic review of cases
Combining the result with our case series, 129 CADASIL patients with 142 ICH events were identified. Data on genetic testing were available in 124 (96.1%). CADASIL was diagnosed on the basis of a NOTCH3 mutation in 123 (95.3%), a known family mutation with clinical features in the absence of genetic confirmation in 2 (1.6%) [13, 40], GOM in the absence of genetic testing in 3 (2.3%) [16, 17, 30], and GOM in 1 patient with negative testing in exon 2–24 [19], Of those with a diagnosed NOTCH3 mutation, 119 (96.7%) were typical cysteine altering, 3 (2.4%) were cysteine sparing, and 1 with genetically confirmed CADASIL but NOTCH3 mutation detail wasn’t mentioned [33].
A summary of the characteristics of all patients is shown in Table 3. The mean age at the time of ICH was 56.6 ± 15.7 (SD), and 74 patients (57.4%) were male. Asian CADASIL patients had ICH at an older mean age compared to other regions (58.1 ± 15.4 and 51.9 ± 10.6 years, respectively) (online resources, Table S7). Of 84 patients with available information, 32 (38.1%) had ICH as the first manifestation of CADASIL.
Hypertension was the most common vascular risk factor present in 91 patients (71.1%) and was more commonly seen in Asian patients (78.6% vs. 46.7%). Six patients (7.8%) were on anticoagulants (warfarin in 3 patients, low molecular weight heparin (LMWH) in 1, and two, the unspecified type).
A total of 142 ICH lesions were identified from brain imaging or autopsy studies. Most lesions were subcortical haemorrhages (130, 91.5%). The sites of haemorrhages were thalamus (58, 40.8%), basal ganglia (34, 23.9%), subcortical white matter (18, 12.7%), cerebellum (12, 8.5%), brainstem (8, 5.6%), and cortical (10, 7.0%). One ICH was in the parietal lobe but was not specified if it was cortical or subcortical (1, 0.7%), and the remaining case was reported as having multiple lesions (1, 0.7%). This trend was similarly seen across different ethnicities. All patients with brain MRI (128, 99.2%) had widespread white matter hyperintensities. CMBs were present in 99 of 105 patients (89.3%) of whom information was reported, and lacunes 86 of 93 patients (92.5%).
Intraventricular extension was observed in 25 patients (21.6%), with most being basal ganglia or thalamic haemorrhages. Out of 129 cases, 11 patients (8.5%) died acutely from the ICH; of these, three patients had large basal ganglia or thalamic haemorrhages with intraventricular extension, two had brainstem haemorrhages, one had an extensive left temporal lobe haemorrhage with severe mass effect, one with right frontal subcortical haemorrhage developed aspiration pneumonia, and in the remaining four details were not specified.
Discussion
In this study, we describe the point prevalence of ICH in a large prospectively recruited CADASIL cohort at about 2%. In a systematic review of published literature, we further identified 119 cases, giving a total of 129 patients with 142 ICHs when combined with our case series. This allowed us to characterise the characteristics of ICH in CADASIL. About 92% were subcortical, with the commonest locations being the thalamus (40.8%) and basal ganglia (23.9%), while 14.1% were infratentorial. Intraventricular extension occurred in 21.6%, and the ICH was fatal in 8.5%.
We observed a similar point prevalence of ICH to that previously reported from European CADASIL cohorts, which ranged from 0.5% to 2.7% [42,43,44]; however, ICH was more commonly observed in the Asian cohorts. A recent meta-analysis reported the pooled prevalence of ICH in CADASIL of 10.1%, with 17.7% and 2.0% within the Asian and European subgroups, respectively [45,46,47]. The mechanism leading to this difference is unknown, although the Asian population has been reported to have a higher prevalence of hypertension in CADASIL patients [48].
In our cohort, there was no difference in the proportion of patients with a mutation in the EGFr 1–6, which has been associated with an increased risk of ischaemic stroke [7], between those with and without ICH. Whether the mutation site is related to the ICH phenotype remains to be further explored. Interestingly, the mutation profiles in CADASIL patients are slightly different as most of the mutations in Far East Asia occur in exon 11 [45, 47], while in our cohort, mutations mostly occur in exon 4 (63.6% in the whole cohort and 40.0% in ICH patients) (online resource Table S2).
We also describe the risk factor profile in patients with ICH, although a comparison group without ICH was only available in our case series. Hypertension was the most common vascular risk factor present in 71.1%. The prevalence was higher than those observed in large CADASIL cohorts (23.0%–39.4%) [44, 45, 49, 50]. Hypertension has been associated with an increased risk of ischaemic stroke in CADASIL [12] and has been suggested as a risk factor for ICH in CADASIL in previously published studies [15, 51]. Our results suggest that CADASIL is associated with ICH rather than occurring independently in patients with risk factors such as hypertension. In support of this, our observed ICH point prevalence at 2% is considerably higher than the ICH prevalence rate one would expect in the UK population (about 185.1 cases per 100,000 or 0.2%) [52]. Furthermore, about half of the ICH patients in our cohort did not have hypertension at the time of ICH. About half (48.1%) were prescribed antiplatelet during the time of ICH, and 7.8% were prescribed anticoagulants. In our case series, there was a significant increase in ICH in patients taking anticoagulants but not antiplatelets. Concern over this risk of ICH has led European Guidelines to suggest anticoagulants should be avoided in CADASIL, unless there is an alternative reason why they must be given [53].
CMBs were also common in CADASIL cases with ICH, occurring in more than 90% of those with appropriate MRI sequences performed. CMBs are frequently present in CADASIL, being reported in 31–66% [51, 54], but the figure we found seems higher.
Our study has a number of strengths. The case series was identified from large, prospectively recruited CADASIL patients, making it possible to estimate point prevalence. Furthermore, we identified all other published cases from the literature. However, it also has limitations. The CADASIL cohort only included patients presenting to the UK National CADASIL clinic and other clinical stroke services, so there may be a referral bias. Owing to the rarity of ICH in CADASIL, most included studies were case reports, and there was no control group for the systematic review cases. Therefore, it wasn’t possible to reliably determine the association between the potential risk factors and ICH. In this review, three patients from Asian studies had a cysteine-sparing NOTCH3 mutation. It is still controversial about the pathogenicity of these mutations in CADASIL, which might be different from typical CADASIL.
In conclusion, ICH is rare in CADASIL but does occur in about 2% of individuals. It is usually subcortical, appears more common in Far East Asia, and may be associated with hypertension and anticoagulants.
Data availability
The anonymised data are available from the corresponding author upon reasonable request.
References
Chabriat H, Joutel A, Dichgans M, Tournier-Lasserve E, Bousser MG (2009) Cadasil. Lancet Neurol 8:643–653
Kim Y, Bae JS, Lee JY, Song HK, Lee JH, Lee M, Kim C, Lee SH (2022) Genotype and phenotype differences in CADASIL from an Asian perspective. Int J Mol Sci 23:11506
Wardlaw JM, Smith EE, Biessels GJ, Cordonnier C, Fazekas F, Frayne R, Lindley RI, O’Brien JT, Barkhof F, Benavente OR, Black SE, Brayne C, Breteler M, Chabriat H, Decarli C, de Leeuw FE, Doubal F, Duering M, Fox NC, Greenberg S, Hachinski V, Kilimann I, Mok V, Oostenbrugge R, Pantoni L, Speck O, Stephan BC, Teipel S, Viswanathan A, Werring D, Chen C, Smith C, van Buchem M, Norrving B, Gorelick PB, Dichgans M (2013) Neuroimaging standards for research into small vessel disease and its contribution to ageing and neurodegeneration. Lancet Neurol 12:822–838
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. J Clin Epidemiol 134:178–189
Moola S, Munn Z, Tufanaru C, Aromataris E, Sears K, Sfetcu R, Currie M, Lisy K, Qureshi R, Mattis P, Mu P (2020) Chapter 7: Systematic reviews of etiology and risk. In: Aromataris E, Munn Z (eds) JBI Manual for Evidence Synthesis. JBI, Available from: https://synthesismanual.jbi.global
Munn Z, Barker TH, Moola S, Tufanaru C, Stern C, McArthur A, Stephenson M, Aromataris E (2020) Methodological quality of case series studies: an introduction to the JBI critical appraisal tool. JBI Evid Synth 18:2127–2133
Rutten JW, Van Eijsden BJ, Duering M, Jouvent E, Opherk C, Pantoni L, Federico A, Dichgans M, Markus HS, Chabriat H, Lesnik Oberstein SAJ (2019) The effect of NOTCH3 pathogenic variant position on CADASIL disease severity: NOTCH3 EGFr 1–6 pathogenic variant are associated with a more severe phenotype and lower survival compared with EGFr 7–34 pathogenic variant. Genet Med 21:676–682
Tang SC, Chen YR, Chi NF, Chen CH, Cheng YW, Hsieh FI, Hsieh YC, Yeh HL, Sung PS, Hu CJ, Chern CM, Lin HJ, Lien LM, Peng GS, Chiou HY, Jeng JS (2019) Prevalence and clinical characteristics of stroke patients with p. R544C NOTCH3 mutation in Taiwan. Ann Clin Transl Neurol 6:121–128
Chen CH, Tang SC, Cheng YW, Tsai HH, Chi NF, Sung PS, Yeh HL, Lien LM, Lin HJ, Lee MJ, Hu CJ, Chiou HY, Jeng JS (2019) Detrimental effects of intracerebral haemorrhage on patients with CADASIL harbouring NOTCH3 R544C mutation. J Neurol Neurosurg Psychiatry 90:841–843
Chen CH, Hsu HC, Cheng YW, Chen YF, Tang SC, Jeng JS (2022) Prominent juxtacortical white matter lesion hallmarks NOTCH3-related intracerebral haemorrhage. Stroke Vasc Neurol 7:38–46
Chen CH, Chu YT, Chen YF, Ko TY, Cheng YW, Lee MJ, Chen PL, Tang SC, Jeng JS (2022) Comparison of clinical and neuroimaging features between NOTCH3 mutations and nongenetic spontaneous intracerebral haemorrhage. Eur J Neurol 29:3243–3254
Adib-Samii P, Brice G, Martin RJ, Markus HS (2010) Clinical spectrum of CADASIL and the effect of cardiovascular risk factors on phenotype: study in 200 consecutively recruited individuals. Stroke 41:630–634
Choi JC, Kang SY, Kang JH, Park JK (2006) Intracerebral hemorrhages in CADASIL. Neurology 67:2042–2044
Choi JC, Song SK, Lee JS, Kang SY, Kang JH (2013) Diversity of stroke presentation in CADASIL: study from patients harboring the predominant NOTCH3 mutation R544C. J Stroke Cerebrovasc Dis 22:126–131
Liao YC, Hu YC, Chung CP, Wang YF, Guo YC, Tsai YS, Lee YC (2021) Intracerebral hemorrhage in cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy: prevalence, clinical and neuroimaging features and risk factors. Stroke 52:985–993
Sourander P, Wälinder J (1977) Hereditary multi-infarct dementia. Lancet 1:1015
Baudrimont M, Dubas F, Joutel A, Tournier-Lasserve E, Bousser MG (1993) Autosomal dominant leukoencephalopathy and subcortical ischemic stroke. A Clinicopathol Study Stroke 24:122–125
Maclean AV, Woods R, Alderson LM, Salloway SP, Correia S, Cortez S, Stopa EG (2005) Spontaneous lobar haemorrhage in CADASIL. J Neurol Neurosurg Psychiatry 76:456–457
Ragoschke-Schumm A, Axer H, Fitzek C, Dichgans M, Peters N, Mueller-Hoecker J, Witte OW, Isenmann S (2005) Intracerebral haemorrhage in CADASIL. J Neurol Neurosurg Psychiatry 76:1606–1607
Werbrouck BF, De Bleecker JL (2006) Intracerebral haemorrhage in CADASIL A case report. Acta Neurol Belg 106:219–221
Kotorii S, Goto H, Kondo T, Matsuo H, Takahashi K, Shibuya N (2006) Case of CADASIL showing spontaneous subcortical hemorrhage with a novel mutation of Notch3 gene. Rinsho Shinkeigaku 46:644–648
Oh JH, Lee JS, Kang SY, Kang JH, Choi JC (2008) Aspirin-associated intracerebral hemorrhage in a patient with CADASIL. Clin Neurol Neurosurg 110:384–386
Mizuno T, Muranishi M, Torugun T, Tango H, Nagakane Y, Kudeken T, Kawase Y, Kawabe K, Oshima F, Yaoi T, Itoh K, Fushiki S, Nakagawa M (2008) Two Japanese CADASIL families exhibiting Notch3 mutation R75P not involving cysteine residue. Intern Med 47:2067–2072
Delgado MG, Coto E, Tuñon A, Sáiz A (2011) CADASIL: how to avoid the unavoidable? BMJ Case Rep. https://doi.org/10.1136/bcr.08.2011.4727
Sano Y, Shimizu F, Kawai M, Omoto M, Negoro K, Kurokawa T, Fujisawa H, Suzuki M, Okayama N, Suehiro Y, Hinoda Y, Kanda T (2011) p.Arg332Cys mutation of NOTCH3 gene in two unrelated Japanese families with CADASIL. Intern Med 50:2833–2838
Pradotto L, Orsi L, Daniele D, Caroppo P, Lauro D, Milesi A, Sellitti L, Mauro A (2012) A new NOTCH3 mutation presenting as primary intracerebral haemorrhage. J Neurol Sci 315:143–145
Lian L, Li D, Xue Z, Liang Q, Xu F, Kang H, Liu X, Zhu S (2013) Spontaneous intracerebral hemorrhage in CADASIL. J Headache Pain 14:98
Mehta S, Mehndiratta P, Sila CA (2013) Spontaneous cerebellar hemorrhage associated with a novel Notch3 mutation. J Clin Neurosci 20:1034–1036
Rinnoci V, Nannucci S, Valenti R, Donnini I, Bianchi S, Pescini F, Dotti MT, Federico A, Inzitari D, Pantoni L (2013) Cerebral hemorrhages in CADASIL: report of four cases and a brief review. J Neurol Sci 330:45–51
Marlen G, Andrea S (2016) Cerebral Hemorrhage in CADASIL: first report in entre Ríos. J Alzheimer’s Disease Parkinsonism. https://doi.org/10.4172/2161-0460.1000292
Koutroulou I, Karapanayiotides T, Grigoriadis N, Karacostas D (2016) CADASIL presenting with spontaneous intracerebral hemorrhage: report of a case and description of the first family in Northern Greece. Hippokratia 20:76–79
Zhang C, Li W, Li S, Niu S, Wang X, Tang H, Yu X, Chen B, Shi Y, Chen Q, Guo L, Pan Y, Wang Y, Zhang Z (2017) CADASIL: two new cases with intracerebral hemorrhage. Ann Clin Transl Neurol 4:266–271
Chiang CC, Christiansen ME, O’Carroll CB (2019) Fatal intracerebral hemorrhage in cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL): a case report. Neurologist 24:136–138
Wang W, Ren Z, Shi Y, Zhang J (2020) A novel mutation outside of the EGFr encoding exons of NOTCH3 gene in a Chinese with CADASIL. J Stroke Cerebrovasc Dis 29:105410
Hu L, Liu G, Fan Y (2022) R558C NOTCH3 mutation in a CADASIL patient with intracerebral hemorrhage: a case report with literature review. J Stroke Cerebrovasc Dis 31:106541
Chu Y, Wang Q, Ma Y, Xu L, Ren K, Liu J, Tao D, Cao H, Ji X (2023) A case of recurrent intracranial hemorrhage in CADASIL caused by NOTCH3 c.1759C > T heterozygous mutation. J Clin Lab Anal 37:e24840
Lee YC, Liu CS, Chang MH, Lin KP, Fuh JL, Lu YC, Liu YF, Soong BW (2009) Population-specific spectrum of NOTCH3 mutations, MRI features and founder effect of CADASIL in Chinese. J Neurol 256:249–255
Bersano A, Bedini G, Markus HS, Vitali P, Colli-Tibaldi E, Taroni F, Gellera C, Baratta S, Mosca L, Carrera P, Ferrari M, Cereda C, Grieco G, Lanfranconi S, Mazucchelli F, Zarcone D, De Lodovici ML, Bono G, Boncoraglio GB, Parati EA, Calloni MV, Perrone P, Bordo BM, Motto C, Agostoni E, Pezzini A, Padovani A, Micieli G, Cavallini A, Molini G, Sasanelli F, Sessa M, Comi G, Checcarelli N, Carmerlingo M, Corato M, Marcheselli S, Fusi L, Grampa G, Uccellini D, Beretta S, Ferrarese C, Incorvaia B, Tadeo CS, Adobbati L, Silani V, Faragò G, Trobia N, Grond-Ginsbach C, Candelise L (2018) The role of clinical and neuroimaging features in the diagnosis of CADASIL. J Neurol 265:2934–2943
Kim Y, Lee SH (2019) Novel characteristics of race-specific genetic functions in Korean CADASIL. Medicina (Kaunas) 55:521
Palazzo P, Le Guyader G, Neau JP (2021) Intracerebral hemorrhage in CADASIL. Rev Neurol (Paris) 177:422–430
Nogueira R, Couto CM, Oliveira P, Martins B, Montanaro VVA (2023) Clinical and epidemiological profiles from a case series of 26 Brazilian CADASIL patients. Arq Neuropsiquiatr 81:417–425
Nannucci S, Rinnoci V, Pracucci G, MacKinnon AD, Pescini F, Adib-Samii P, Bianchi S, Dotti MT, Federico A, Inzitari D, Markus HS, Pantoni L (2018) Location, number and factors associated with cerebral microbleeds in an Italian-British cohort of CADASIL patients. PLoS ONE 13:e0190878
Mönkäre S, Kuuluvainen L, Schleutker J, Myllykangas L, Pöyhönen M (2022) Clinical features and spectrum of NOTCH3 variants in Finnish patients with cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL). Acta Neurol Scand 146:643–651
Dupé C, Guey S, Biard L, Dieng S, Lebenberg J, Grosset L, Alili N, Hervé D, Tournier-Lasserve E, Jouvent E, Chevret S, Chabriat H (2023) Phenotypic variability in 446 CADASIL patients: Impact of NOTCH3 gene mutation location in addition to the effects of age, sex and vascular risk factors. J Cereb Blood Flow Metab 43:153–166
Liao YC, Hsiao CT, Fuh JL, Chern CM, Lee WJ, Guo YC, Wang SJ, Lee IH, Liu YT, Wang YF, Chang FC, Chang MH, Soong BW, Lee YC (2015) Characterization of CADASIL among the Han Chinese in Taiwan: distinct genotypic and phenotypic profiles. PLoS ONE 10:e0136501
Chen S, Ni W, Yin XZ, Liu HQ, Lu C, Zheng QJ, Zhao GX, Xu YF, Wu L, Zhang L, Wang N, Li HF, Wu ZY (2017) Clinical features and mutation spectrum in Chinese patients with CADASIL: a multicenter retrospective study. CNS Neurosci Ther 23:707–716
Min JY, Park SJ, Kang EJ, Hwang SY, Han SH (2022) Mutation spectrum and genotype-phenotype correlations in 157 Korean CADASIL patients: a multicenter study. Neurogenetics 23:45–58
Lai QL, Zhang YX, Wang JJ, Mo YJ, Zhuang LY, Cheng L, Weng ST, Qiao S, Liu L (2022) Occurrence of intracranial hemorrhage and associated risk factors in cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy: a systematic review and meta-analysis. J Clin Neurol 18:499–506
Bianchi S, Zicari E, Carluccio A, Di Donato I, Pescini F, Nannucci S, Valenti R, Ragno M, Inzitari D, Pantoni L, Federico A, Dotti MT (2015) CADASIL in central Italy: a retrospective clinical and genetic study in 229 patients. J Neurol 262:134–141
Mukai M, Mizuta I, Watanabe-Hosomi A, Koizumi T, Matsuura J, Hamano A, Tomimoto H, Mizuno T (2020) Genotype-phenotype correlations and effect of mutation location in Japanese CADASIL patients. J Hum Genet 65:637–646
Lee JS, Ko K, Oh JH, Park JH, Lee HK, Floriolli D, Paganini-Hill A, Fisher M (2017) Cerebral microbleeds, hypertension, and intracerebral hemorrhage in cerebral autosomal-dominant arteriopathy with subcortical infarcts and leukoencephalopathy. Front Neurol 8:203
Krishnamurthi RV, Ikeda T, Feigin VL (2020) Global, regional and country-specific burden of ischaemic stroke, intracerebral haemorrhage and subarachnoid haemorrhage: a systematic analysis of the global Burden of Disease Study 2017. Neuroepidemiology 54:171–179
Mancuso M, Arnold M, Bersano A, Burlina A, Chabriat H, Debette S, Enzinger C, Federico A, Filla A, Finsterer J, Hunt D, Lesnik Oberstein S, Tournier-Lasserve E, Markus HS (2020) Monogenic cerebral small-vessel diseases: diagnosis and therapy. Consensus recommendations of the European Academy of Neurology. Eur J Neurol 27:909–927
Lesnik Oberstein SA, van den Boom R, van Buchem MA, van Houwelingen HC, Bakker E, Vollebregt E, Ferrari MD, Breuning MH, Haan J (2001) Cerebral microbleeds in CADASIL. Neurology 57:1066–1070
Acknowledgements
The authors would like to thank all CADASIL patients who participated in the UK Familial SVD study and the recruiting centres: Cambridge University Hospital NHS Foundation Trust: Markus H. S., Edwards H., Jolly A., and Henthorn M.; Leeds Teaching Hospital NHS Trust: Hassan A. and Waugh D.; Royal Hallamshire Hospital, Sheffield: Harkness K., Howe J., Edwards M., and Richard E.; St. George’s Healthcare NHS Trust, London: Khan U., Ghalata R., Stratton S., and Williams R.; University College London Hospitals NHS Foundation Trust: Werring D., Banara A., and Scheherazade F. The authors would like to thank Professor Yi-Chu Liao, Taiwan Veterans General Hospital, Taiwan, and Professor Jay Chol Choi, Jeju National University, South Korea, for providing additional information on their literature to be included in the systematic review.
Funding
N. Sukhonpanich is supported by the Prince Mahidol Award Youth Programme Scholarship, Prince Mahidol Foundation, Thailand. The UK Familial Cerebral Small Vessel Disease study is funded by the British Heart Foundation programme grant (RG/4/32218). Recruitment was supported by the NIHR Clinical Research Network. This research was supported by the NIHR Cambridge Biomedical Research Centre (BRC-1215–20014) and the Cambridge BHF Centre of Research Excellence (RE/18/1/34212). The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care.
Author information
Authors and Affiliations
Contributions
NS involved in the study conceptualisation, search strategy design, literature search, data extraction, analysis and manuscript drafting and revision. HSM is the consultant neurologist of the National CADASIL referral clinic. HSM involved in the study conceptualisation, search strategy design, data analysis, supervision, and manuscript revision.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that there is no conflict of interest.
Ethical approval
All patients gave written informed consent for their data to be used for research. Ethical approval of the UK Familial Small Vessel Disease study was obtained from the East of England Cambridge Central Research Ethics Committee (16/EE/0118).
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sukhonpanich, N., Markus, H.S. Prevalence, clinical characteristics, and risk factors of intracerebral haemorrhage in CADASIL: a case series and systematic review. J Neurol 271, 2423–2433 (2024). https://doi.org/10.1007/s00415-023-12177-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-023-12177-0