Abstract
Background
The semantic fluency test is one of the most widely used neuropsychological tests in dementia diagnosis. Research utilizing the qualitative, psycholinguistic information embedded in its output is currently underexplored in presymptomatic and prodromal genetic FTD.
Methods
Presymptomatic MAPT (n = 20) and GRN (n = 43) mutation carriers, and controls (n = 55) underwent up to 6 years of neuropsychological assessment, including the semantic fluency test. Ten mutation carriers became symptomatic (phenoconverters). Total score and five qualitative fluency measures (lexical frequency, age of acquisition, number of clusters, cluster size, number of switches) were calculated. We used multilevel linear regression modeling to investigate longitudinal decline. We assessed the co-correlation of the qualitative measures at each time point with principal component analysis. We explored associations with cognitive decline and grey matter atrophy using partial correlations, and investigated classification abilities using binary logistic regression.
Results
The interrater reliability of the qualitative measures was good (ICC = 0.75–0.90). There was strong co-correlation between lexical frequency and age of acquisition, and between clustering and switching. At least 4 years pre-phenoconversion, GRN phenoconverters had fewer but larger clusters (p < 0.001), and fewer switches (p = 0.004), correlating with lower executive function (r = 0.87–0.98). Fewer switches was predictive of phenoconversion, correctly classifying 90.3%. Starting at least 4 years pre-phenoconversion, MAPT phenoconverters demonstrated an increase in lexical frequency (p = 0.009) and a decline in age of acquisition (p = 0.034), correlating with lower semantic processing (r = 0.90). Smaller cluster size was predictive of phenoconversion, correctly classifying 89.3%. Increase in lexical frequency and decline in age of acquisition were associated with grey matter volume loss of predominantly temporal areas, while decline in the number of clusters, cluster size, and switches correlated with grey matter volume loss of predominantly frontal areas.
Conclusions
Qualitative aspects of semantic fluency could give insight into the underlying mechanisms as to why the “traditional” total score declines in the different FTD mutations. However, the qualitative measures currently demonstrate more fluctuation than the total score, the measure that seems to most reliably deteriorate with time. Replication in a larger sample of FTD phenoconverters is warranted to identify if qualitative measures could be sensitive cognitive biomarkers to identify and track mutation carriers converting to the symptomatic stage of FTD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Frontotemporal dementia (FTD) is a clinically and pathologically heterogeneous type of early-onset dementia, typically characterized by atrophy of the frontal and/or temporal lobes [1]. The clinical profile of FTD shows behavioural and language disturbances, with cognitive deficits in executive function and relative sparing of memory and visuospatial abilities [2]. Up to 40% of FTD cases have an autosomal dominant pattern of inheritance. Mutations in the progranulin (GRN), microtubule-associated protein tau (MAPT) and chromosome 9 open reading frame 72 (C9orf72) genes are the most common causes [3]. Early diagnosis—albeit difficult due to the heterogeneous symptoms and overlap with other forms of dementia and psychiatric disorders—is essential for proper patient management and planning, non-pharmacological treatment, and patient stratification in upcoming disease-modifying clinical trials [4].
Research in the genetic FTD field has been increasingly moving towards the presymptomatic and early prodromal stages, as the critical time-window for treatment most likely lies prior to overt symptom onset. With promising avenues opening for clinical trials, identifying robust biomarkers is of utmost importance [5]. Previous neuropsychological studies showed that subtle cognitive deficits and decline are present in the presymptomatic stage, and gene-specific cognitive profiles can be detected [6,7,8]. These findings suggest that neuropsychological assessment in the presymptomatic and early prodromal stages can provide sensitive cognitive markers for FTD.
The semantic fluency test is one of the most widely used tests in neuropsychological assessments. In this brief, easy-to-apply test, people have to generate items from a particular semantic category (e.g., animals, foods) in 1 min [9]. The semantic fluency test presents high sensitivity and specificity for dementia diagnosis [9], with impaired performance found in both symptomatic [10] and presymptomatic FTD [7, 8]. Although the total number of items generated is commonly used to quantify test performance, qualitative, psycholinguistic information embedded in the output can also be investigated, including clusters (number of multiword stringsFootnote 1), switches (number of transitions between clusters), age of acquisition (AoA; the age at which a word is learned), and lexical frequency (LF; how often a word occurs in daily language) [12, 13]. Previous research demonstrated the prognostic value of qualitative fluency measures in cognitively healthy subjects at-risk for and in conversion from prodromal to overt Alzheimer’s Dementia (AD) [14, 15]. This approach has been underexplored in presymptomatic and/or prodromal genetic FTD, while this psycholinguistic information may be able to detect the subtle development of FTD’s characteristic language symptoms at an early stage.
The aim of this study was therefore to investigate longitudinal changes in five qualitative aspects of semantic fluency (i.e., number of clusters and switches, cluster size, AoA, and LF) in mutation carriers that developed FTD (phenoconverters), presymptomatic mutation carriers, and non-carriers from autosomal dominant GRN- and MAPT-FTD families. We were specifically interested in the inflection point (i.e., when in the disease trajectory) at which the qualitative measures start to deviate from normal. Additionally, we explored the co-correlation between the qualitative measures, and their associations with cognitive decline and grey matter (GM) volume loss, and the prognostic value of decline in qualitative measures in predicting symptomatic onset.
Methods
Participants
We included longitudinal data of 118 participants from the FTD Risk Cohort of the Erasmus MC University Medical Center (Rotterdam, the Netherlands). This is an ongoing study in which first-degree family members of FTD patients due to a pathogenic mutation are followed on a 1- or 2-year basis [16]. Participants were recruited between February 2010 and October 2019. DNA genotyping at study entry assigned participants to the mutation carrier (n = 63; MAPT n = 20, GRN n = 43) or non-carrier group (controls; n = 55). Upon study entry, all mutation carriers were presymptomatic according to clinical diagnostic criteria for FTD [2, 17, 18], and had global CDR®-plus-NACC-FTLD [19] scores of 0. Ten mutation carriers (MAPT n = 6, GRN n = 4) developed symptoms during follow-up (phenoconverters). Diagnoses were made in multidisciplinary consensus meetings, using information from the standardized clinical assessment (see below). Phenoconverters met the following criteria: [i] progressive deterioration of behaviour, language and/or motor functioning; [ii] functional decline, evidenced by multiple study visits with global CDR®-plus-NACC-FTLD [19] ≥ 0.5 without reversing back to 0; and [iii] cognitive decline, evidenced by ≥ 1.5 SD below age-, sex- and education-specific means in ≥ 1 domain on neuropsychological assessment. Eight phenoconverters had clinical features of bvFTD (MAPT n = 6, GRN n = 2), and two had non-fluent variant PPA (GRN n = 2). The presymptomatic mutation carriers that did not develop FTD symptoms are referred to as non-converters (n = 53).
Clinical assessment
Every 1–2 years, all participants underwent a standardized clinical assessment, consisting of a structured interview with the participant and a knowledgeable informant (incorporating the CDR®-plus-NACC-FTLD [19]), medical history taking, neurological examination, neuropsychological assessment, and brain MRI. The neuropsychological assessment consisted of cognitive screening tests (Mini-Mental State Examination, MMSE, and Frontal Assessment Battery, FAB), and tests within the major cognitive domains. Neuropsychiatric symptoms were assessed with the Beck’s Depression Inventory (BDI) and the brief questionnaire form of the Neuropsychiatric Inventory (NPI-Q) (see [7, 8] for full test battery).
Qualitative fluency measures
The semantic fluency task was part of the neuropsychological assessment at each study visit. In this task, participants were asked to verbally generate as many different animals as possible in 60 s [20]. The total score is the number of animals produced minus errors. Additionally, we calculated five qualitative fluency measures from the output: LF, AoA, number of clusters, cluster size and number of switches (Box 1, Appendix 1 for scoring guidelines).
Study design
Phenoconverters, non-converters and controls were compared at five time points: baseline, and follow-up after 2, 4, 5 and 6 years, for the purpose of this study restructured as (Fig. 1):
-
1.
Four years before phenoconversion—data were available for six phenoconverters. The other four developed symptoms between baseline and the first follow-up visit, therefore no data 4 years prior to phenoconversion were available. The data were compared to the baseline data of non-converters and controls.
-
2.
Two years before phenoconversion—data were available for all ten phenoconverters. The data were compared to the 2-year follow-up data of non-converters and controls.
-
3.
Phenoconversion—data were available for all ten phenoconverters. The data were compared to the 4-year follow-up data of non-converters and controls.
-
4.
One year after phenoconversion—data were available for four phenoconverters. The other six were clinically too impaired to undergo neuropsychological testing or passed away (n = 4), or converted recently, therefore no data 1 year after phenoconversion were available yet (n = 2). The data were compared to the 5-year follow-up data of non-converters and controls.
-
5.
Two years after phenoconversion—data were available for four phenoconverters. The other six were clinically too impaired to undergo neuropsychological testing or passed away (n = 5), or converted recently therefore no data 1 year after phenoconversion were available yet (n = 1). The data were compared to the 6-year follow-up data of non-converters and controls.
Subject sample and study design. The total sample (n = 118) was divided into mutation carriers (n = 63) and non-carriers (healthy controls; n = 55), the mutation carrier group was split into phenoconverters (n = 10) and non-converters (n = 53). The original data was restructured for phenoconverters, so that there were five time points: 4 years before phenoconversion, 2 years before phenoconversion, phenoconversion, 1 year post-phenoconversion and 2 years post-phenoconversion. Four years before phenoconversion data was available for only 6 phenoconverters, as the other four phenoconverters developed symptoms between baseline and the first follow-up visit, and therefore no data 4 years prior to phenoconversion were available. One and 2 years after phenoconversion data was available for only 4 phenoconverters; the other 6 were either lost to follow-up, as they were clinically too impaired to undergo neuropsychological testing or passed away (n = 4), or converted recently so that follow-up data post-phenoconversion was not available at this time point (n = 2). The data of phenoconverters was compared to respectively baseline, and follow-up after 2, 4, 5 and 6 years in non-converters and healthy controls. The remaining six converters were either lost to follow-up, as they were clinically too impaired to undergo neuropsychological testing or passed away (n = 4), or converted recently so that follow-up data post-phenoconversion was not available at this time point (n = 2). The data were compared to the 5-year follow-up data of non-converters and controls
MRI acquisition and (pre)processing
On each study visit, we performed volumetric T1-weighted MRI scanning on a Philips 3 T Achieva MRI scanner (Philips Medical Systems, Best, the Netherlands) using either an 8- or 32-channel SENSE head coil and the following scan parameters: inversion/repetition time = 933/2200 ms, flip angle = 8°, voxel size = 1.1 × 1.1 × 1.1 mm, matrix size = 256 × 256 × 208, total scan time = 4.43 min. All scans underwent visual quality control. The DICOM images were subsequently corrected for gradient nonlinearity distortions and converted to NifTI format. They were then pre-processed in the Voxel-Based Morphometry (VBM) pipeline in Statistical Parametric Mapping 12 (SPM12; Functional Imaging Laboratory, University College London, London, UK; www.fil.ion.ucl.ac.uk/spm) implemented in Matlab R2018a (Mathworks, USA). First, the T1-weighted images were normalized to a template space and segmented into GM, white matter (WM) and cerebrospinal fluid (CSF), after which they were rigidly aligned. We calculated total intracranial volume (TIV) by adding GM, WM and CSF. Second, the segmentations were spatially normalized to a DARTEL template by applying the flow fields of all the individual scans. Images were smoothed using a 6 mm full width at half maximum (FWHM) isotropic Gaussian kernel. After every preprocessing step, images were visually inspected.
Statistical analysis
We performed statistical analyses using SPSS Statistics 25.0 (IBM Corp., Armonk, NY). Alpha was set at 0.05 across all comparisons (two-tailed). We compared continuous demographic data between groups using one-way ANOVAs for normally distributed data (with Bonferroni post-hoc tests), or Kruskal–Wallis tests for non-normally distributed data (with Mann–Whitney U post-hoc tests). We analysed between-group differences in sex and gene with Pearson Χ2 tests. Interrater reliability was explored using intraclass correlation analysis. We assessed the co-correlation of the five qualitative measures at each time point by means of principal component analysis using Varimax rotation. Only factors accounting for 3% or more of variance and Eigenvalues > 1 were retained. Factor loadings were only considered meaningful when r > 0.450, and any item that did not load sufficiently onto a factor was removed [23]. For ease of interpretation, we first converted raw fluency and relevant neuropsychological measures (Boston Naming Test (BNT), Semantic Association Test (SAT), Trailmaking test B (TMT-B), Stroop colour-word test card III) into z-scores by subtracting the mean of controls from each individual’s raw score at that time point, divided by the SDs of controls at that time point. We then used multilevel linear regression modeling to investigate longitudinal decline in total and qualitative fluency measures. We performed two separate analyses to assess longitudinal change in qualitative fluency measures per [i] clinical status (phenoconverters, non-converters, controls) and [ii] gene (MAPT, GRN). We entered [i] or [ii], time, and first-order interactions, with age, sex, education and total number of words generated as covariates. Assumptions were checked (non-linearity, dependence of errors, outliers, heteroscedasticity). Significant interactions between covariates and dependent variables were included in the model. We based the covariance structure (Toeplitz Heterogeneous) on the lowest Akaike Information Criterion (AIC), as a lower AIC indicates a better model fit. We included the random intercept as this model presented with a lower AIC. For converters, we calculated deltas of the standardized values for the [i] qualitative fluency measures and [ii] relevant neuropsychological tests between restructured time-point 1 in the six phenoconverters that had data 4 years before phenoconversion, or time-point 2 in the four phenoconverters that only had data 2 years before phenoconversion, and time-point 3 (phenoconversion). We then explored the association between the delta fluency measures and delta neuropsychological tests (corrected for age, sex and education) using partial correlations. Change over time maps were generated by subtracting the GM maps calculated at time-point 3 (phenoconversion) from the maps calculated at time-point 1 or 2 in SPM12. We then explored the relationship between the delta qualitative fluency measures and delta GM maps by means of multiple regression models. Age, sex, TIV, and head coil were entered as covariates. We set the statistical threshold at p < 0.05, adjusted for multiple comparisons with familywise error (FWE) correction. Lastly, to investigate classification abilities of these delta z-scores, we performed binary logistic regression analyses. Assumptions were checked (non-linearity, dependence of errors, outliers, multicollinearity). The models were selected with a forward stepwise method according to the likelihood ratio test and applying the standard p-values for variable inclusion (0.05) and exclusion (0.10). Goodness of fit was evaluated with the HL Χ2 test, with Nagelkerke R2 as measure of effect size. The analyses were adjusted for age, sex, education, and total number of words generated. All models were corrected for multiple comparisons (Bonferroni).
Results
Demographics and clinical data
Demographic and clinical data are shown in Table 1. There were no differences between phenoconverters, non-converters, and controls in age [F(2,117) = 0.212, p = 0.809], sex [X(2) = 1.568, p = 0.457], gene [X(2) = 4.830, p = 0.089] or education level [F(2,117) = 1.290, p = 0.279]. There were no differences between MAPT and GRN phenoconverters in age [F(2,9) = 2.966, p = 0.123], sex [X(2) = 0.524, p = 0.262] or education level [F(2,9) = 0.022, p = 0.945]. We found no differences between phenoconverters, non-converters, and controls regarding baseline MMSE [F(2,117) = 0.229, p = 0.796], FAB [F(2,40) = 2.504, p = 0.095], BDI [F(2,116) = 0.607, p = 0.547] or NPI-Q [F(2,73) = 0.031, p = 0.969]. There were no differences between MAPT and GRN phenoconverters in these measures (all p > 0.05). There were no differences in neuropsychological test scores between group at study entry (all p > 0.05).
Interrater reliability
Two independent raters (LCJ, SAAML), blinded to participant’s clinical and genetic status, scored the number of clusters, cluster sizes, and number of switches (see Methods—Qualitative fluency measures, and Appendix 1). The interrater reliability of all three measures was considered ‘good’ (i.e., intraclass correlation coefficients 0.75–0.90) [25]: number of clusters 0.81 [95% CI 0.76–0.85], cluster sizes 0.85 [95% CI 0.81–0.87], and number of switches 0.85 [95% CI 0.81–0.88].
Co-correlation between the qualitative measures
Appendix 2 shows the results of the principal component analysis on the five qualitative measures per time-point. Based on our criteria and visual inspection of the scree plot, we could extract two components. The first component explained between 39.5 and 49.7% of the total variance, and both LF and AoA had high loadings (r > 0.900). The second factor explained between 29.0 and 43.3% of the total variance, and both the number of clusters and the number of switches, and at some time-points also the cluster size, had high loadings (r > 0.59).
Longitudinal qualitative fluency trajectories
The inflection points and longitudinal trajectories of the fluency measures are shown in Table 2. The individual trajectories in phenoconverters are displayed in Fig. 2. Phenoconverters had declining total scores from at least 4 years pre-phenoconversion (p < 0.001), while there was no decline in non-converters or controls (p > 0.05). As can be noted from Fig. 2, the qualitative measures demonstrated more noise in their fluctuation than the total score. Phenoconverters had higher LF from 4 years pre-phenoconversion in comparison to controls (p = 0.016). Also the number of clusters (p < 0.001) and switches (p = 0.004) started to decline at this point. AoA declined from phenoconversion onwards (p = 0.050). No longitudinal change was found for cluster size (p > 0.05). There was no change in qualitative fluency measures in non-converters or controls (p > 0.05). Different inflection points and longitudinal trajectories were found for GRN and MAPT phenoconverters. At least 4 years pre-phenoconversion, GRN phenoconverters started producing fewer words in comparison to controls (p = 0.005). Moreover, they started producing fewer but larger clusters (both p < 0.001), and used fewer switches (p = 0.004). GRN phenoconverters had larger cluster sizes than MAPT phenoconverters (p < 0.001). LF and AoA did not change in GRN phenoconverters compared to controls (p > 0.05). Starting at least 4 years pre-phenoconversion, MAPT phenoconverters produced fewer words than controls (p < 0.001). Moreover, LF started to increase (p = 0.007), while AoA (p = 0.034), the number of clusters (p = 0.009), and cluster size (p = 0.010) declined. The number of switches did not change (p > 0.05).
Individual verbal fluency trajectories in the ten FTD phenoconverters. Lines represent the individual longitudinal changes (years to phenoconversion, X-axis) in semantic fluency total scores, lexical, age of acquisition, cluster count and size, and switches (z-scores, Y-axis) in the 10 phenoconverters. The 6 MAPT phenoconverters are displayed in blue, the 3 GRN phenoconverters in green. Only the total score was available for phenoconverter 10 (GRN 4). Abbreviations: MAPT microtubule-associated protein tau, GRN progranulin
Associations with cognitive decline
Partial correlation coefficients between the fluency measures and relevant neuropsychological tests are shown in Table 3. In phenoconverters, decline on the SAT verbal correlated with an increase in LF (p = 0.031) and a decline in AoA (p = 0.037). Worse performance on TMT-B (p = 0.031) and Stroop-III (p = 0.026) correlated with an increase in cluster size, while worse performance on Stroop-III correlated with a decline in the number of switches (p = 0.031). In GRN phenoconverters, decline on the SAT verbal correlated with a decline in AoA (p = 0.020). Worse performance on Stroop-III correlated with a decline in the number of clusters (p = 0.022), and an increase in cluster size (p = 0.014), while decline on TMT-B correlated with a decline in the number of switches (p = 0.024). In MAPT phenoconverters, decline on the SAT verbal correlated with an increase in LF (p = 0.015) and a decline in AoA (p = 0.014).
Associations with GM volume loss
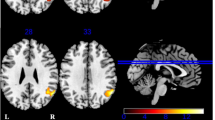
The relationships between the qualitative fluency measures and GM volume loss are displayed in Fig. 3 and Appendix 3. In the total group of phenoconverters, worse performance on all five qualitative fluency measures was associated with GM volume loss in large overlapping areas spanning the frontal (e.g., middle frontal gyrus) and temporal lobes (e.g., middle and superior temporal gyrus), and the anterior insular and cingulate cortices (pFWE-corrected < 0.05). In GRN phenoconverters, an increase in LF and a decline in AoA were associated with GM volume loss of the cerebellum and predominantly temporal areas (e.g., medial temporal lobe, inferior temporal gyrus). A decline in the number of clusters, cluster size, and the number of switches correlated with GM volume loss of the cerebellum, insular cortex, and putamen (pFWE-corrected < 0.05). In MAPT phenoconverters, an increase in LF and a decline in AoA were associated with GM volume loss of the cerebellum and predominantly temporal areas (e.g., anterior temporal pole, inferior temporal gyrus). A decline in the number of clusters, cluster size, and the number of switches correlated with GM volume loss of the cerebellum and predominantly frontal areas (e.g., middle and superior frontal gyrus, frontal pole) (pFWE-corrected < 0.05).
Grey matter atrophy patterns associated with lower qualitative fluency performance. VBM analyses demonstrated grey matter volume loss to be associated with lower performance in lexical frequency (red), age of acquisition (green), clusters (blue), cluster size (yellow), and switches (copper) in the total group of phenoconverters (top), GRN phenoconverters (middle), and MAPT phenoconverters (bottom). We set the statistical threshold at p < 0.05 (FWE-corrected). Abbreviations: L left, GRN progranulin, MAPT microtubule-associated protein tau
Classification abilities of qualitative fluency measures
Decline in the total score differentiated between phenoconverters and non-converters [X2(1) = 7.669, p = 0.006] and controls [X2(1) = 7.643, p = 0.006]. This was primarily driven by MAPT phenoconverters, as it differentiated well between this group and non-converters [X2(1) = 5.919, p = 0.015] and controls [X2(1) = 5.902, p = 0.015], but not between GRN phenoconverters, non-converters, and controls (p > 0.05). A decline in switches was predictive of phenoconversion in GRN [X2(1) = 5.069, p = 0.024], correctly classifying 90.3% of cases; a decline in cluster size was predictive of phenoconversion in MAPT [X2(1) = 3.894, p = 0.048], correctly classifying 89.3% of cases.
Discussion
This study examined longitudinal changes in qualitative aspects of the semantic fluency task in a large cohort of FTD phenoconverters, presymptomatic mutation carriers, and non-carriers from GRN- and MAPT-FTD families. Phenoconverters showed a decline in the total score from at least 4 years pre-phenoconversion, with individually-varying inflection points and longitudinal trajectories in qualitative fluency measures in GRN and MAPT. At least 4 years pre-phenoconversion, GRN phenoconverters started producing fewer but larger clusters, and switched less between clusters, which was correlated with executive dysfunction. A decline in switching was predictive of phenoconversion. At least 4 years pre-phenoconversion, MAPT phenoconverters demonstrated an increase in LF and a decline in AoA, which was correlated with semantic deficits. A decline in cluster size was predictive of phenoconversion. Increase in LF and decline in AoA were associated with GM volume loss of predominantly temporal areas, while decline in the number of clusters, cluster size, and switches correlated with GM volume loss of predominantly frontal areas.
The semantic fluency total score is strongly intertwined with the qualitative aspects of the task. For accurate and timely word retrieval, both AoA and LF [26], and clustering and switching components [27] are required. Coinciding with the results of our principal component analysis, in which we found a AoA-LF component and a clusters-switches component, previous studies found strong correlations between AoA and LF, and between the number of clusters and the number of switches. With respect to the relation between AoA and LF, correlations have found to be high in natural languages, as early-acquired words tend to occur more frequently than late-required words [28]. The number of switches and the number of clusters are correlated, as by identifying the number of clusters, one can generate the number of switches (corresponds to the number of clusters minus one) [27].
Irrespective of the underlying FTD mutation, we showed a decline in the total score in mutation carriers from at least 4 years prior to phenoconversion. Decline in semantic fluency was found to be an early cognitive marker in other neurodegenerative diseases. For instance, in preclinical AD and Huntington’s disease, as early as 12 years before the onset of dementia, decline in a measure of semantic memory was found [29, 30]. In another study, it was amongst the most statistically sensitive cognitive measures of symptomatic conversion [31]. Studies into semantic fluency decline in presymptomatic FTD have shown somewhat contrasting results. One study demonstrated decline in semantic fluency in MAPT mutation carriers from 6 years before estimated symptom onset [7], whereas another only found decline at estimated symptom onset [6]. It should be noted that both studies used estimated years to symptom onset as a proxy for actual onset, which can be less reliable in familial FTD [6]. Utilizing a similar research design as our current study, decline of semantic fluency from 4 years before symptom onset in MAPT converters, and decline of semantic fluency was found to be the best predictor for having an MAPT mutation [8]. Although semantic fluency did not decline in GRN converters, decline on phonemic fluency was found to be predictive of an GRN mutation, confirming the value of fluency tasks in presymptomatic FTD as they can distinguish the underlying genotype [8].
Starting at least 4 years pre-phenoconversion, GRN mutation carriers produced fewer but larger clusters, and had fewer switches, than MAPT phenoconverters and controls. A likely explanation for the decline in their total score is that GRN phenoconverters deteriorate in cognitive flexibility [32], and thereby lose the ability to switch between semantic clusters in order to generate more words. Indeed, in our study the decline in the number of clusters and switches, and the increase in cluster size, correlated with decline in executive function. Executive dysfunction is known to be a distinctive cognitive feature in GRN mutations, demonstrating deficits in symptomatic mutation carriers [33], extending to the presymptomatic stage [7, 8]. When converting to the symptomatic stage, GRN mutation carriers also show the most decline in executive function [34].
Starting at least 4 years pre-phenoconversion, MAPT mutation carriers produced words with a higher LF and a lower AoA, and had fewer clusters and smaller clusters than GRN phenoconverters and controls. These findings point towards deterioration of the semantic system as an explanation as to why qualitative fluency measures change in MAPT. First, semantic decline is most likely to affect words with a lower LF and a higher AoA first, as the categorical organization of the system retrieves the ‘typical’ exemplars faster and more accurately, and they are better represented and more interconnected to other concepts than those that enter the semantic system later in life [35]. The reliance of both processes on semantic processing is further supported by their correlation with the verbal SAT which assesses verbal semantic deficits [36]. Clustering relies on lexical retrieval, vocabulary size and lexical access, and thus is mainly supported by the integrity of the semantic system [13].
We demonstrated that—irrespective of the underlying mutation—decline in the number of clusters, cluster size, and switches correlated with GM volume loss of predominantly frontal areas, while worse LF and AoA performance was associated with GM volume loss of predominantly temporal areas. These neuroanatomical correlates are in line with the predominant frontal involvement in GRN mutation carriers [6, 34], and link the degradation of the fronto-insula network to less cognitive flexibility—and as a consequence early clustering-switching impairment—as the most likely underpinning of declining fluency performance in conversion to GRN-associated FTD. The finding that LF and AoA rely on temporal lobe functioning could explain why these qualitative features are changing early in MAPT mutation carriers, as temporal volume loss is considered the neuroimaging hallmark of MAPT [37, 38], being present up to several decades before symptom onset [39]. Although PPA is not a frequent clinical phenotype, semantic impairments are well-described in MAPT-related FTD [40]. Our cohort includes three P301L and three G272V phenoconverters, which is too small to investigate differences between the two tau mutations. Nevertheless, with larger sample sizes it would be interesting to explore if there is clinical heterogeneity across the MAPT mutations [41], as the P301L mutation often presents with a language phenotype with semantic deficits [42]. Impairments in semantic fluency are found to be common as the result of cerebellar pathology, as this subcortical region plays a crucial role in motor performance and executive processes necessary for organizing and monitoring word output [43].
The key strength of our study is our longitudinal design, spanning up to 6 years of follow-up in a large single-centre sample of participants from GRN and MAPT FTD-families. This design allowed the investigation of mutation carriers as they were converting to the symptomatic stage, which provides us more accurate information about the underlying disease process than previous studies that used estimated years to onset as a proxy [40]. We chose multilevel linear modelling to handle potential missing data and unbalanced time-points that were the result of our ongoing prospective study. The small sample of phenoconverters, in combination with the large fluctuations in the data of the qualitative measures, are the largest drawback of the study, which has hampered our statistical power and interpretation of results. As the multilevel model assumes a linear relationship between genetic status and fluency performance it is possible that we have missed non-linear effects. Ideally, the semantic fluency test should not have been used in determining phenoconversion, however in our multidisciplinary approach we have used all available clinical information—e.g., MR brain imaging, anamnestic and heteroanamnestic information, questionnaires—so that symptom onset did not solely depend on the neuropsychological assessment. Although theoretically a cluster can consist of a single word, one cannot measure interword intervals for fewer than two words, so that a single-word “cluster” cannot be corroborated by the measurement of interword intervals. Following Ledoux et al. [11], we therefore defined clusters not as single words, but only as multiword strings (i.e. two or more consecutive words) whose relationship is defined by one of the scoring rules, but realize this could have penalized patients with a low total output. Lastly, the analyses on the presymptomatic mutation carriers were performed using the original baseline and follow-up visits, regardless of years from potential phenoconversion, therefore they might have lost some sensitivity to detect decline. Future directions include replication of our findings in larger multicentre cohorts, including C9orf72 mutation carriers. Moreover, using qualitative measures in discriminative event-based models could help us understand the dynamics of disease progression and how other biomarkers (e.g., NfL) fit into this [44]. Lastly, future studies could look into the effect of using time-bins next to the usual 60-s output, as most people start with readily available animals and produce less familiar exemplars as the task develops (which affects the qualitative measures), and was found to be particularly sensitive to mutation status in presymptomatic APOE-ε4 carriers at-risk of developing AD [14].
Conclusion
Our pilot study shows that qualitative aspects of semantic fluency change in presymptomatic FTD, and shows different profiles and inflection points depending on the mutation involved. This could provide important insight into the mechanisms as to why the “traditional” total score is declining. Its brief and easy-to-apply nature makes the total score of the semantic fluency test a likely candidate cognitive biomarker for upcoming clinical trials for FTD, but more research with a larger sample of phenoconverters is needed to replicate our findings, and to explore the additional value of qualitative measures in identifying and tracking mutation carriers as they convert to the symptomatic stage.
Data availability
Anonymized data not published within this article will be made available upon reasonable request from any qualified investigator.
Notes
We used the scoring rules of Ledoux et al. [11] here, in which the scoring of clusters is restricted to strings of two or more successive words. Although theoretically a cluster can consist of only a single word, one cannot measure interword intervals for fewer than two words, thus a single-word cluster cannot be corroborated by the measurement of interword intervals. Reference: Ledoux et al. [11].
References
Olney NT, Ong E, Goh SYM et al (2020) Clinical and volumetric changes with increasing functional impairment in familial frontotemporal lobar degeneration. Alzheimer’s Dementia 16(1):49–59. https://doi.org/10.1016/j.jalz.2019.08.196
Rascovsky K, Hodges JR, Knopman D et al (2011) Sensitivity of revised diagnostic criteria for the behavioural variant of frontotemporal dementia. Brain 134(Pt 9):2456–2477. https://doi.org/10.1093/brain/awr179
Rohrer JD, Warren JD (2011) Phenotypic signatures of genetic frontotemporal dementia. Curr Opin Neurol 24(6):542–549. https://doi.org/10.1097/WCO.0b013e32834cd442
Musa G, Slachevsky A, Munoz-Neira C et al (2020) Alzheimer’s disease or behavioral variant frontotemporal dementia? Review of key points toward an accurate clinical and neuropsychological diagnosis. J Alzheimers Dis 73(3):833–848. https://doi.org/10.3233/JAD-190924
Rohrer JD, Warren JD, Fox NC, Rossor MN (2013) Presymptomatic studies in genetic frontotemporal dementia. Revue Neurologique 169(10):820–824. https://doi.org/10.1016/j.neurol.2013.07.010
Rohrer JD, Nicholas JM, Cash DM et al (2015) Presymptomatic cognitive and neuroanatomical changes in genetic frontotemporal dementia in the Genetic Frontotemporal dementia Initiative (GENFI) study: a cross-sectional analysis. Lancet Neurol 14(3):253–262. https://doi.org/10.1016/s1474-4422(14)70324-2
Jiskoot LC, Dopper EGP, Heijer TD et al (2016) Presymptomatic cognitive decline in familial frontotemporal dementia: a longitudinal study. Neurology 87(4):384–391. https://doi.org/10.1212/wnl.0000000000002895
Jiskoot LC, Panman JL, van Asseldonk L et al (2018) Longitudinal cognitive biomarkers predicting symptom onset in presymptomatic frontotemporal dementia. J Neurol 265(6):1381–1392. https://doi.org/10.1007/s00415-018-8850-7
Lopes M, Brucki SMD, Giampaoli V, Mansur LL (2009) Semantic verbal fluency test in dementia: preliminary retrospective analysis. Dement Neuropsychol 3(4):315–320. https://doi.org/10.1590/S1980-57642009DN30400009
Libon DJ, McMillan C, Gunawardena D, Powers C, Massimo L, Khan A et al (2009) Neurocognitive contributions to verbal fluency deficits in frontotemporal lobar degeneration. Neurology 73:535–542. https://doi.org/10.1212/WNL.0b013e3181b2a4f5
Ledoux K, Vannorsdall TD, Pickett EJ, Bosley LV, Gordon B, Schretlen DJ (2014) Capturing additional information about the organization of entries in the lexicon from verbal fluency productions. J Clin Exp Neuropsychol 36(2):205–220. https://doi.org/10.1080/13803395.2013.878689
Vonk JMJ, Jonkers R, Hubbard HI, Gorno-Tempini ML, Brickman AM, Obler LK (2019) Semantic and lexical features of words dissimilarly affected by non-fluent, logopenic, and semantic primary progressive aphasia. J Int Neuropsychol Soc 25(10):1011–1022
van den Berg E, Jiskoot LC, Grosveld MJH, van Swieten JC, Papma JM (2017) Qualitative assessment of verbal fluency performance in frontotemporal dementia. Dement Geriatr Cogn Disord 44(1–2):35–44. https://doi.org/10.1159/000477538
Vonk JMJ, Flores RJ, Rosado D et al (2019) Semantic network function captured by word frequency in nondemented APOE epsilon4 carriers. Neuropsychology 33(2):256–262. https://doi.org/10.1037/neu0000508
Pakhomov SV, Hemmy LS (2014) A computational linguistic measure of clustering behavior on semantic verbal fluency task predicts risk of future dementia in the nun study. Cortex 55:97–106. https://doi.org/10.1016/j.cortex.2013.05.009
Dopper EGP, Rombouts SARB, Jiskoot LC et al (2014) Structural and functional brain connectivity in presymptomatic familial frontotemporal dementia. Neurology 83(2):e19–e26. https://doi.org/10.1212/wnl.0000000000000583
Gorno-Tempini ML, Hillis AE, Weintraub S et al (2011) Classification of primary progressive aphasia and its variants. Neurology 76(11):1006–1014. https://doi.org/10.1212/WNL.0b013e31821103e6
Brooks BR, Miller RG, Swash M, Munsat TL, World Federation of Neurology Research Group on Motor Neuron D (2000) El Escorial revisited: revised criteria for the diagnosis of amyotrophic lateral sclerosis. Amyotroph Lateral Scler Other Motor Neuron Disord 1(5):293–299. https://doi.org/10.1080/146608200300079536
Miyagawa T, Brushaber D, Syrjanen J et al (2020) Use of the CDR® plus NACC FTLD in mild FTLD: Data from the ARTFL/LEFFTDS consortium. Conference Paper. Alzheimer’s Dementia 16(1):79–90. https://doi.org/10.1016/j.jalz.2019.05.013
Thurstone LL (1938) Primary mental abilities. University of Chicago Press, Chicago
Keuleers E, Brysbaert M, New B (2010) SUBTLEX-NL: a new measure for Dutch word frequency based on film subtitles. Behav Res Methods 42(3):643–650. https://doi.org/10.3758/BRM.42.3.643
Brysbaert M, Stevens M, De Deyne S, Voorspoels W, Storms G (2014) Norms of age of acquisition and concreteness for 30,000 Dutch words. Acta Psychol (Amst) 150:80–84. https://doi.org/10.1016/j.actpsy.2014.04.010
DeVellis RF (2003) Scale development: theory and applications, vol 26, 2nd edn. Sage Publications, Thousand Oaks
Hendriks MKR, Gorissen M, Schmand BDA (eds) (2014) Neuropsychologische diagnostiek. Boom, pp 176–178
Koo TK, Li MY (2016) A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J Chiropr Med 15(2):155–163. https://doi.org/10.1016/j.jcm.2016.02.012
Meschyan G, Hernandez A (2002) Age of acquisition and word frequency: determinants of object-naming speed and accuracy. Mem Cognit 30:262–269
Carlesso K, Eduara P, Fernandes G, Diehl M, Rodrigues C, Rochele P, Fonseca P, Fernanda Altmann R (2022) Clustering and switching in verbal fluency: a comparison between control and individuals with brain damage. CoDAS 34(2):e20200365
Yum YN, Law S-P (2019) Interactions of age of acquisition and lexical frequency effects with phonological regularity: an ERP study. Psychophysiology 56:e13433
Amieva H, le Goff M, Millet X et al (2008) Prodromal Alzheimer’s disease: successive emergence of the clinical symptoms. Ann Neurol 64(5):492–498. https://doi.org/10.1002/ana.21509
Larsson MU, Almkvist O, Luszcz MA, Robins Wahlin TB (2008) Phonemic fluency deficits in asymptomatic gene carriers for Huntington’s disease. Neuropsychology 22(5):596–605
Langbaum JB, Hendrix SB, Ayutyanont N et al (2014) An empirically derived composite cognitive test score with improved power to track and evaluate treatments for preclinical Alzheimer’s disease. Alzheimers Dement 10(6):666–674. https://doi.org/10.1016/j.jalz.2014.02.002
Ho AK, Sahakian BJ, Robbins TW, Barker RA, Rosser AE, Hodges JR (2002) Verbal fluency in Huntington’s disease: a longitudinal analysis of phonemic and semantic clustering and switching. Neuropsychologia 40(8):1277–1284. https://doi.org/10.1016/s0028-3932(01)00217-2
Poos JM, Jiskoot LC, Leijdesdorff SMJ et al (2020) Cognitive profiles discriminate between genetic variants of behavioral frontotemporal dementia. J Neurol. https://doi.org/10.1007/s00415-020-09738-y
Jiskoot LC, Panman JL, Meeter LH et al (2019) Longitudinal multimodal MRI as prognostic and diagnostic biomarker in presymptomatic familial frontotemporal dementia. Brain 142(1):193–208
Bird H, Lambon Ralph MA, Patterson K, Hodges JR (2000) The rise and fall of frequency and imageability: noun and verb production in semantic dementia. Brain Lang 73(1):17–49. https://doi.org/10.1006/brln.2000.2293
Visch-Brink ESD, Denes G (2005) Semantische associatie test. Swets & Zeitlinger
Rohrer JD (2012) Structural brain imaging in frontotemporal dementia. Review. Biochimica Et Biophysica Acta-Mol Basis Dis 1822(3):325–332
Young AL, Marinescu RV, Oxtoby NP et al (2018) Uncovering the heterogeneity and temporal complexity of neurodegenerative diseases with Subtype and Stage Inference. Nat Commun 4273:1–16
Chu SA, Flagan TM, Staffaroni AM et al (2021) Brain volumetric deficits in MAPT mutation carriers: a multisite study. Ann Clin Transl Neurol 8(1):95–110. https://doi.org/10.1002/acn3.51249
Moore KM, Nicholas JM, Grossman M et al (2020) Age at symptom onset and death and disease duration in genetic frontotemporal dementia: an international retrospective cohort study. Lancet Neurol 19(2):145–156. https://doi.org/10.1016/S1474-4422(19)30394-1
Whitwell JL, Jack CR, Boeve BF et al (2009) Atrophy patterns in IVS10+16, IVS10+3, N279K, S305N, P301L, and V337M MAPT mutations. Neurology 73(13):1058–1065. https://doi.org/10.1212/WNL.0b013e3181b9c8b9
Borrego-Écija S, Morgado J, Palencia-Madrid L et al (2017) Frontotemporal dementia caused by the P301L mutation in the MAPT gene: clinicopathological features of 13 cases from the same geographical origin in Barcelona, Spain. Dementia Geriatr Cogn Disord 44(3–4):213–221. https://doi.org/10.1159/000480077
Arasanz CP, Staines WR, Roy EA, Schweizer TA (2012) The cerebellum and its role in word generation: a cTBS study. Cortex 48:718–724
van der Ende EL, Bron EE, Poos JM et al (2021) A data-driven disease progression model of fluid biomarkers in genetic frontotemporal dementia. Brain. https://doi.org/10.1093/brain/awab382
Cardoso MJ, Modat M, Wolz R et al (2015) Geodesic information flows: spatially-variant graphs and their application to segmentation and fusion. IEEE Trans Med Imaging 34(9):1976–1988. https://doi.org/10.1109/TMI.2015.2418298
Troyer AK, Moscovitch M, Winicur G (1997) Clustering and switching as two components of verbal fluency: evidence from younger and older adults. Neuropsychology 11(1):138–146. https://doi.org/10.1037/0894-4105.11.1.138
Acknowledgements
The authors thank all the participants and their families for taking part in the FTD-RisC study.
Funding
The FTD-RisC study is supported by Dioraphte Foundation Grant 09-02-00, the Association for frontotemporal Dementias Research Grant 2009, The Netherlands organization for Scientific Research (NWO) grant HCMI 056-13-018, ZonMw Memorabel project numbers 733050103 & 733050813, the Bluefield project, JPND PreFrontAls consortium project number 733051042, and ZonMw Onderzoeksprogramma Dementie (project number 10510032120002).
Author information
Authors and Affiliations
Contributions
LCJ contributed to the conception and design of the study, acquired and analysed data, and drafted the manuscript, figures and tables. JMP, SAAML, LAAG and JvH contributed to data collection. EvdB, DDS and JMJV contributed to the design and/or data interpretation of the study. HS contributed to the conception and design of the manuscript, and is PI of the FTD-RisC study. All authors were involved in editing and approval of the final draft of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Ethical approval
The investigators and participants were blinded for the genetic status of the at-risk participants, except for those that underwent predictive testing (27.8%). All participants provided written informed consent. The Erasmus MC ethics committee gave approval for the study (MEC 2009-409). This study was conducted in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Appendices
Appendix 1: Scoring guidelines for qualitative aspects of semantic fluency
General scoring rules
When more than three words were missing from the fluency output (e.g., word was illegible or there was a tick instead of a word written), only the total number of words generated was used for further analyses (i.e., no qualitative aspects were calculated). For the total number of words generated, only correct words were used, thus no perseverative, pseudo or rule-break errors were included. Clusters were allowed to (partially) overlap. ‘Embedded’ clusters (i.e., a cluster within a cluster) were not taken into account. In determining the total cluster size, words that belonged to more than one cluster were counted twice (e.g., pig belongs to the cluster “farm animals” as well as the cluster “porcine”). In determining the number of switches, overlapping clusters were also counted as a switch. The beginning and end of the output were not taken into account as a switch.
Ad hoc scoring rules for LF and AoA
For LF and AoA the original fluency output was followed if possible. If a word was not found in the databases for either LF [25] or AoA [26] the following ad hoc scoring rules were followed: 1. finding the singular of this word in the database (e.g., pig instead of pigs); 2. finding the word in the database with the adjective removed (e.g., bear instead of polar bear). When the word was still not available, a different value was used: for LF it was assumed that the missing word occurred only once in the language corpus. With this assumed frequency a Zipf-value of ‘’zero frequency’’ was calculated. For AoA, an imputed value was used when the word was not available in the database. The imputed value was based on the highest age of acquisition score (15.1) within this study.
Cluster scoring rules*
Words that belong to the same subcategory | E.g., pets; dog, cat, rabbit |
Words that have a strong association in the real world | E.g., cat, mouse |
Words that share the same first letter | E.g., fish, ferret |
Words that share the same first sound | E.g., cat, kangaroo |
Words that share the same word | E.g., horse, seahorse |
Words that rhyme | E.g. cat, rat |
Appendix 2: Principal component analysis on the five qualitative fluency measures
Items | Component coefficients | Communalities | |
|---|---|---|---|
1 | 2 | ||
4 years before phenoconversion | |||
LF | − 0.958 | 0.025 | 0.915 |
AoA | 0.950 | 0.033 | 0.904 |
Clusters | 0.061 | 0.888 | 0.792 |
Cluster size | 0.535 | − 0.593 | 0.637 |
Switching | − 0.014 | 0.907 | 0.824 |
Eigenvalues | 2.314 | 1.758 | |
Variance, % | 46.280 | 35.167 | |
2 years before phenoconversion | |||
LF | 0.975 | 0.025 | 0.951 |
AoA | 0.970 | − 0.024 | 0.941 |
Clusters | 0.172 | 0.837 | 0.731 |
Cluster size | 0.235 | − 0.807 | 0.706 |
Switching | 0.039 | 0.902 | 0.815 |
Eigenvalues | 1.977 | 2.167 | |
Variance, % | 39.542 | 43.332 | |
Phenoconversion | |||
LF | − 0.942 | 0.034 | 0.888 |
AoA | 0.948 | − 0.137 | 0.918 |
Clusters | 0.424 | 0.093 | 0.188 |
Cluster size | 0.228 | − 0.839 | 0.756 |
Switching | 0.249 | 0.855 | 0.793 |
Eigenvalues | 2.096 | 1.448 | |
Variance, % | 41.922 | 28.965 | |
1 year after phenoconversion | |||
LF | − 0.959 | − 0.015 | 0.920 |
AoA | 0.950 | 0.103 | 0.913 |
Clusters | 0.047 | − 0.350 | 0.629 |
Cluster size | 0.711 | 0.919 | 0.847 |
Switching | − 0.124 | 0.924 | 0.869 |
Eigenvalues | 2.420 | 1.758 | |
Variance, % | 48.396 | 35.156 | |
2 years after phenoconversion | |||
LF | − 0.929 | − 0.220 | 0.912 |
AoA | 0.938 | 0.168 | 0.907 |
Clusters | 0.197 | 0.903 | 0.853 |
Cluster size | 0.606 | − 0.624 | 0.756 |
Switching | 0.196 | 0.904 | 0.855 |
Eigenvalues | 2.484 | 1.800 | |
Variance, % | 49.679 | 35.995 | |
Appendix 3: Neuroimaging correlates of the qualitative fluency measures
Group | Measure | Cluster | T | P FWE-corrected | MNI coordinates | Region | ||
|---|---|---|---|---|---|---|---|---|
x | y | z | ||||||
Total group of phenoconverters | LF | 71,256 | 73.01 | < 0.001 | 33 | 20 | − 4 | Anterior insula R |
851 | 77.34 | < 0.001 | − 33 | 16 | 52 | Middle frontal gyrus L | ||
639 | 49.57 | < 0.001 | − 20 | 42 | 34 | Superior frontal gyrus L | ||
476 | 81.60 | < 0.001 | − 33 | 16 | 54 | Middle frontal gyrus L | ||
438 | 32.65 | < 0.001 | 56 | − 45 | 2 | Middle temporal gyrus R | ||
426 | 26.97 | < 0.001 | 60 | − 26 | 0 | Superior temporal gyrus R | ||
124 | 38.36 | < 0.001 | − 24 | 2 | − 48 | Temporal pole L | ||
117 | 27.64 | 0.002 | − 22 | − 3 | − 46 | Inferior temporal gyrus L | ||
AoA | 98,828 | 88.54 | < 0.001 | − 33 | 16 | 56 | Middle frontal gyrus L | |
1038 | 37.85 | < 0.001 | − 32 | 42 | 30 | Middle frontal gyrus L | ||
442 | 32.71 | < 0.001 | 57 | − 42 | 4 | Middle temporal gyrus R | ||
425 | 28.39 | < 0.001 | 60 | − 26 | 0 | Superior temporal gyrus R | ||
348 | 24.14 | < 0.001 | 54 | − 60 | − 8 | Inferior temporal gyrus R | ||
171 | 31.55 | < 0.001 | − 32 | 56 | 12 | Middle frontal gyrus L | ||
Clusters | 10,400 | 70.87 | < 0.001 | − 33 | − 6 | 9 | Anterior insula L | |
774 | 66.01 | < 0.001 | 63 | − 38 | 30 | Supramarginal gyrus R | ||
401 | 58.95 | < 0.001 | − 34 | 18 | 57 | Middle frontal gyrus L | ||
341 | 24.59 | 0.005 | 54 | − 60 | − 8 | Inferior temporal gyrus R | ||
254 | 40.05 | < 0.001 | − 32 | 42 | 30 | Middle frontal gyrus L | ||
191 | 35.09 | < 0.001 | − 51 | − 48 | 18 | Superior temporal gyrus L | ||
135 | 24.82 | 0.005 | − 4 | 32 | 48 | Superior frontal gyrus L | ||
Cluster size | 80,154 | 89.77 | < 0.001 | − 3 | 9 | 38 | Middle cingulate gyrus L | |
1513 | 37.39 | < 0.002 | 56 | − 45 | 3 | Middle temporal gyrus | ||
714 | 44.30 | < 0.001 | 63 | − 38 | 30 | Supramarginal gyrus R | ||
390 | 24.13 | < 0.001 | 57 | − 63 | − 2 | Inferior temporal gyrus R | ||
265 | 39.38 | 0.002 | − 32 | 42 | 30 | Middle frontal gyrus L | ||
232 | 27.25 | 0.006 | − 21 | 39 | 40 | Superior frontal gyrus L | ||
Switches | 70,083 | 67.98 | < 0.001 | 33 | 18 | − 4 | Anterior insula R | |
1420 | 31.22 | 0.001 | 8 | 16 | 57 | Supplementary motor cortex R | ||
766 | 44.18 | < 0.001 | 63 | − 38 | 28 | Supramarginal gyrus R | ||
502 | 33.31 | < 0.001 | 57 | − 42 | 4 | Middle temporal gyrus R | ||
487 | 58.10 | < 0.001 | − 34 | 18 | 57 | Middle frontal gyrus L | ||
435 | 28.07 | 0.002 | 46 | − 52 | 8 | Inferior temporal gyrus R | ||
373 | 26.80 | 0.003 | 62 | − 18 | − 3 | Superior temporal gyrus R | ||
286 | 50.52 | < 0.001 | 44 | − 51 | 48 | Angular gyrus R | ||
277 | 29.04 | 0.001 | − 50 | − 63 | − 32 | Cerebellum L | ||
276 | 60.77 | < 0.001 | − 33 | 42 | 32 | Middle frontal gyrus L | ||
179 | 28.99 | 0.001 | − 22 | 39 | 42 | Superior frontal gyrus L | ||
GRN phenoconverters | LF | 122 | 114.52 | 0.002 | 3 | − 52 | − 12 | Cerebellum R |
72 | 99.72 | 0.002 | − 30 | 0 | − 18 | Medial temporal lobe L | ||
60 | 114.68 | 0.002 | 27 | − 44 | − 24 | Cerebellum R | ||
32 | 92.53 | 0.002 | 40 | − 57 | − 42 | Cerebellum R | ||
28 | 111.21 | 0.002 | − 20 | − 46 | − 21 | Cerebellum L | ||
25 | 93.84 | 0.002 | − 34 | 10 | − 10 | Anterior insula L | ||
18 | 95.61 | 0.002 | − 39 | − 78 | − 33 | Cerebellum L | ||
AoA | 142 | 113.75 | 0.002 | 0 | − 52 | − 14 | Cerebellum L | |
85 | 116.16 | 0.002 | 24 | − 44 | − 24 | Cerebellum R | ||
55 | 96.63 | 0.002 | − 38 | − 8 | 6 | Anterior insula L | ||
38 | 91.25 | 0.002 | − 28 | 0 | − 16 | Medial temporal lobe L | ||
23 | 106.62 | 0.002 | − 20 | − 46 | − 22 | Cerebellum L | ||
15 | 93.90 | 0.002 | − 57 | − 46 | − 21 | Inferior temporal gyrus L | ||
Clusters | 328 | 133.16 | 0.002 | 18 | − 66 | − 21 | Cerebellum R | |
141 | 127.04 | 0.002 | − 38 | − 9 | − 4 | Posterior insula L | ||
119 | 122.36 | 0.002 | − 2 | − 52 | − 10 | Cerebellum L | ||
106 | 128.52 | 0.002 | − 20 | − 52 | − 20 | Cerebellum L | ||
64 | 109.98 | 0.002 | − 20 | 8 | − 12 | Putamen L | ||
20 | 106.12 | 0.002 | 21 | − 38 | − 26 | Cerebellum R | ||
17 | 113.55 | 0.002 | 33 | 20 | − 4 | Anterior insula R | ||
Cluster size | 150 | 119.60 | 0.002 | 0 | − 52 | − 14 | Cerebellum L | |
89 | 111.33 | 0.002 | − 36 | − 9 | − 4 | Posterior insula L | ||
72 | 117.38 | 0.002 | 24 | − 44 | − 22 | Cerebellum R | ||
43 | 102.41 | 0.002 | − 20 | 8 | − 12 | Putamen L | ||
11 | 98.60 | 0.002 | 33 | 21 | − 4 | Anterior insula R | ||
Switches | 114 | 112.68 | 0.002 | − 3 | − 52 | − 14 | Cerebellum L | |
104 | 113.17 | 0.002 | − 22 | − 50 | − 24 | Cerebellum L | ||
69 | 121.55 | 0.002 | − 22 | 9 | − 6 | Putamen L | ||
57 | 115.68 | 0.002 | − 36 | − 4 | − 6 | Posterior insula L | ||
35 | 103.79 | 0.002 | 5 | − 50 | − 16 | Cerebellum R | ||
33 | 104.91 | 0.002 | 21 | − 39 | − 24 | Cerebellum R | ||
27 | 102.79 | 0.002 | − 24 | − 74 | − 21 | Cerebellum L | ||
MAPT phenoconverters | LF | 25,864 | 143.78 | < 0.001 | 22 | − 84 | − 27 | Cerebellum R |
117 | 18.41 | < 0.001 | 33 | 22 | − 40 | Inferior temporal gyrus R | ||
58 | 23.94 | < 0.001 | 40 | − 4 | − 51 | Temporal pole R | ||
38 | 29.20 | < 0.001 | − 32 | 20 | − 30 | Temporal pole L | ||
35 | 16.10 | < 0.001 | − 39 | 21 | − 38 | Temporal pole L | ||
28 | 16.07 | 0.002 | 51 | 21 | − 21 | Temporal pole R | ||
AoA | 26,431 | 147.06 | < 0.001 | 22 | − 84 | − 27 | Cerebellum R | |
119 | 18.43 | < 0.001 | 33 | 22 | − 40 | Temporal pole R | ||
47 | 23.08 | < 0.001 | 39 | − 6 | − 51 | Inferior temporal gyrus R | ||
27 | 15.77 | < 0.001 | 51 | 21 | − 20 | Temporal pole R | ||
Clusters | 22,010 | 144.32 | < 0.001 | 22 | − 84 | − 28 | Cerebellum R | |
1669 | 68.58 | < 0.001 | − 32 | 44 | 30 | Middle frontal gyrus L | ||
342 | 57.46 | < 0.001 | 45 | 24 | 42 | Middle frontal gyrus R | ||
310 | 24.65 | < 0.001 | 4 | 58 | 32 | Superior frontal gyrus R | ||
224 | 33.39 | < 0.001 | − 4 | 60 | 18 | Superior frontal gyrus L | ||
216 | 56.60 | < 0.001 | 38 | 46 | 28 | Middle frontal gyrus R | ||
201 | 25.60 | < 0.001 | 51 | 42 | 14 | Middle frontal gyrus R | ||
134 | 24.15 | < 0.001 | 36 | 28 | 54 | Middle frontal gyrus R | ||
Cluster size | 21,760 | 146.75 | < 0.001 | 34 | − 42 | − 30 | Cerebellum R | |
1509 | 71.70 | < 0.001 | − 32 | 44 | 28 | Middle frontal gyrus L | ||
382 | 43.23 | < 0.001 | 45 | 24 | 44 | Middle frontal gyrus R | ||
238 | 28.06 | < 0.001 | − 4 | 60 | 20 | Superior frontal gyrus L | ||
236 | 21.36 | < 0.001 | 20 | 63 | − 16 | Frontal pole R | ||
228 | 24.25 | < 0.001 | 4 | 58 | 32 | Superior frontal gyrus R | ||
212 | 43.14 | < 0.001 | − 21 | 44 | 42 | Superior frontal gyrus L | ||
194 | 23.61 | < 0.001 | 50 | 40 | 14 | Middle frontal gyrus R | ||
185 | 25.34 | < 0.001 | − 46 | 52 | − 2 | Middle frontal gyrus L | ||
167 | 44.46 | < 0.001 | 38 | 48 | 28 | Middle frontal gyrus R | ||
103 | 20.98 | < 0.001 | 36 | 28 | 52 | Middle frontal gyrus R | ||
Switches | 22,641 | 152.76 | < 0.001 | 24 | − 82 | − 28 | Cerebellum R | |
297 | 37.51 | < 0.001 | 44 | 26 | 42 | Middle frontal gyrus R | ||
173 | 38.47 | < 0.001 | 26 | 51 | 36 | Superior frontal gyrus R | ||
149 | 25.54 | < 0.001 | 4 | 58 | 32 | Superior frontal gyrus R | ||
144 | 20.55 | < 0.001 | 34 | 21 | − 39 | Temporal pole R | ||
132 | 24.69 | < 0.001 | − 21 | 42 | 45 | Superior frontal gyrus L | ||
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jiskoot, L.C., van den Berg, E., Laenen, S.A.A.M. et al. Longitudinal changes in qualitative aspects of semantic fluency in presymptomatic and prodromal genetic frontotemporal dementia. J Neurol 270, 5418–5435 (2023). https://doi.org/10.1007/s00415-023-11845-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-023-11845-5