Abstract
Objective
We explored the potential of neurofilament light chain (NfL) in serum and cerebrospinal fluid as a biomarker for neurodestruction in status epilepticus.
Methods
In a retrospective analysis, we measured NfL in serum and cerebrospinal fluid samples of patients with status epilepticus using a highly sensitive single-molecule array technique (Simoa). Status epilepticus was diagnosed according to ILAE criteria. Additionally, we employed an alternative classification with more emphasis on the course of status epilepticus. We used data from three large control groups to compare NfL in status epilepticus versus neurologically healthy controls.
Results
We included 28 patients (mean age: 69.4 years, SD: 15 years) with a median status duration of 44 h (IQR: 80 h). Twenty-one patients (75%) suffered from convulsive status epilepticus and seven (25%) from non-convulsive status epilepticus. Six patients died (21%). Cerebrospinal fluid and serum NfL concentrations showed a high correlation (r = 0.73, p < 0.001, Pearson). The main determinant of NfL concentration was the status duration. NfL concentrations did not differ between convulsive status epilepticus and convulsive status epilepticus classified according to the ILAE or to the alternative classification without and with adjusting for status duration and time between status onset and sampling. We found no association of NfL concentration with death, treatment refractoriness, or prognostic scores.
Conclusion
The results suggest that neurodestruction in status epilepticus measured by NfL is mainly determined by status duration, not status type nor therapy refractoriness. Therefore, our results suggest that regarding neurodestruction convulsive and non-convulsive status epilepticus are both neurological emergencies of comparable urgency.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Status epilepticus (SE) is one of the most important and common neurological emergencies. The incidence in adults varies in different geographic regions, studies, and age groups between 1.3 and 73.7/100,000/year adults with subgroups of 80 years and older reaching an incidence over 150/100,000 [18]. Immediate and consistent action is required to terminate the ongoing epileptic activity because mortality and disability in SE are high. Mortality rates range from 16 to 25% in adults and increase with patient age [3]. For refractory status epilepticus (RSE), the reported fatality rates range from 16 to 39% in adults [19]. Novy, Logroscino and Rossetti reported that only 63% of the survivors of SE and 21% of the survivors with RSE recovered completely [25]. Experiments in primates show that prolonged generalized convulsive seizures cause neuronal damage [23]. However, if convulsive features of the generalized seizures are suppressed by muscle relaxation with curare, neuronal damage still occurs but is somewhat milder, suggesting that generalized non-convulsive seizures are also neurotoxic [24]. In animal models of strictly focal SE, bilateral brain pathology and remote neuronal damage are rare [5]. In summary, animal models of epilepsy suggest (I) that severe neuronal damage is found in brain regions with excessive neuronal discharges leading to glutamatergic excitotoxicity, and (II) neuronal damage is aggravated by physiological compromise associated with convulsions [22]. However, several recent studies of the treatment of SE show that in contrast to former studies neurodestruction is comparable between convulsive and non-convulsive SE (NCSE) [15, 27, 29]. Current studies note that the mortality is even higher in NCSE than in convulsive SE [15, 18]. This lead to fundamental discussions about the treatment of convulsive SE and NCSE [29].
Neuron-specific enolase (NSE) is to date the most promising neurodestruction biomarker in SE, but different studies showed equivocal results concerning the correlation with status duration and outcome [7, 8, 36, 37]. However, none of the biomarkers has yet been introduced in clinical practice [9]. The restricted accessibility of cerebrospinal fluid (CSF), especially for multiple follow-up examinations limits the use of CSF biomarkers. A recently developed highly sensitive single-molecule array (Simoa) immunoassay allows reproducible and precise measurements of brain-specific proteins, such as neurofilaments [28]. Neuronal death leads to the release of neurofilaments in CSF and blood. Neurofilament light chain (NfL) has been investigated successfully as a biomarker of neurodestruction in various neurological diseases [42, 43].
Here, we measured NfL in CSF and serum of patients with SE using Simoa technology to explore three hypotheses: First, we asked whether the NfL concentrations in SE are increased in comparison to controls. Second, we investigated if the NfL concentrations differ between patients with convulsive SE and patients with NCSE diagnosed by the ILAE classification and an alternative classification placing more attention on the course of the SE. Third, we explored if NfL predicts prognosis and is associated with established predictive scales (STESS, EMSE-EAC), therapy refractoriness, and death.
Materials and methods
Setting and study design
This study was conducted in an epilepsy center certified by the German Society for Epileptology (DGfE) at the Department of Neurology of the University Medical Center Schleswig–Holstein (UKSH) in northern Germany. The UKSH, campus Kiel serves a catchment area of about 500,000 people. The study was approved by the Ethics Committee of the Medical Faculty of the Christian-Albrechts-University, Kiel, Germany (D 480/20). The study was conducted following the World Medical Association Declaration of Helsinki. Anonymized data will be shared by request with any qualified investigator.
Patients with SE
We identified patients with a diagnosis of SE between the 1st of February 2015 and the 15th of May 2020 through the hospital information system of the UKSH. We included all patients aged above 18 years with SE and severely impaired vigilance with available CSF and serum samples in our biobank. We identified a SE according to the International League Against Epilepsy (ILAE) classification described in the “Report of the ILAE Task Force on Classification of SE” [40] and excluded absence status. In subsequent sections, we refer to “convulsive SE” versus “NCSE” and add “according to ILAE” if necessary. We allowed a purely clinical diagnosis for convulsive SE but NCSE required in addition to severely impaired vigilance one of the following EEG phenomena: spike-and-wave complexes, generalized or lateralized periodic discharges (GPDs/LEDs) with additional characteristics according to Trinka and Leitinger [41]. The patients with NCSE included in this study met the Salzburg criteria for NCSE [16].
In addition, we classified patients according to criteria developed by Leitinger and Trinka which take the evolution of semiology into account [18]. In short, patients with a convulsive semiology throughout the SE are categorized as group A, while patients with convulsive semiology at the beginning and later switching to a non-convulsive semiology or purely non-convulsive semiology are in categories B and C which we summarized as one category. We will refer to these categories “convulsive only” and “NCSE at any point during evolution” and add “alternative classification” if necessary [18]. The SE duration was inferred from the complete information available in the hospital information system including the nursing documentation.
We excluded patients with neurological diseases known to lead to significant increases in NfL concentration, namely: brain tumor [10], ischemic or hemorrhagic strokes, septicemia or posthypoxic encephalopathy within the preceding 12 months, cerebral amyloid angiopathy, head trauma, cerebral autoimmune diseases [14], infectious meningoencephalitis [21], severe peripheral neuropathy [e.g., critical illness polyneuropathy before lumbar puncture (LP)] [4]s, and severe neurodegenerative illness [20, 26, 35], e.g., dementia with Lewy bodies (DLB), corticobasal degeneration (CBD), progressive supranuclear palsy (PSP) and amyotrophic lateral sclerosis (ALS).
Controls
We obtained control measurements of three well-established population-based cohorts. First, we obtained and measured 71 control samples from the Popgen control cohort, a longitudinal study of health in northern Germany [13]. To expand the age range and enlarge the number of control NfL measurements, we obtained 813 additional control values from the population-based arm (cohort 3) of the BiDirect Study [38] and 295 controls values from the MEMO Study, an elderly community sample within the population-based KORA project [32]. We excluded samples with a neurodegenerative disease but not samples with a history of other age-related comorbidities like cardiovascular diseases to avoid the generation of “supercontrols”. The Popgen samples were measured concurrently with the patient samples on the same instrument while BiDirect and MEMO samples were measured on an identical but not the same instrument using the same chemistry.
Clinical data
We extracted the following clinical data from the medical records: age, sex, date and reason of hospital admission, pre-existing diseases, medication during the hospital stay, duration of SE, time from status onset to sampling, semiology and course of SE, the patient’s outcome assessed in prognostic scores and findings in EEG, cranial MRT and CCT investigations.
NfL measurements
We obtained all serum/CSF pairs at the same time point, namely the time of LP. Polypropylene tubes were used for CSF and serum. We used tubes with gel separators for blood collection. The serum was separated by spinning at 2000×g for 10 min after coagulation. All samples were frozen at − 80 °C in polypropylene cryovials within maximal 48 h at 4 °C. Samples were thawed only once immediately before the measurements.
Analysis was performed on a fully automated HD-X platform (Quanterix) based on Simoa assay technique using a commercially available multiplex kit (Neurology 4-plex: NfL, Tau, UCHL-1, and GFAP). For each run, 8 calibrators were measured in duplicates and two controls (low and high, included in the kit) were analyzed in siglicates at the beginning and end of the run for continuous process control. All samples were measured in duplicates. The coefficients of variance between the replicates of 10% for both serum and CSF were tolerated, by values exceeding 10% the measurements were repeated. For analysis, mean concentration of two replicates was estimated. The laboratory personnel was blinded to the clinical history and other laboratory parameters of the study participants. We analyzed only the NfL measurements of the 4-plex Quanterix kit because NfL is already a fairly well-established biomarker [42, 43] with a large number of available control cohorts and shows favorable properties concerning short transfer time to CSF and blood, the long half-life, and well-developed precise assay [28, 34]. Two patients had missing CSF NfL values because samples had been used up completely. The Shapiro–Wilk test for normality showed that the variables time between status onset and lumbar puncture (“status onset time to LP”) and NfL concentration in CSF as well as serum required logarithmic transformation to conform with the assumption of a normal distribution.
Prognostic parameters
To estimate the severity of the SE, we used the following outcome scores: SE Severity Score (STESS), modified STESS (mSTESS), and Epidemiology-Based Mortality Score in SE (EMSE) [17, 30, 31]. The STESS rates the categories consciousness, worst seizure type, age, and history of previous seizures with points: 0–3 points predicting a favorable and 4–6 points predicting an unfavorable outcome. There are different versions of the EMSE: We used the EMSE-EAC rating the patient’s etiology, age, and each comorbidity, which leads to an open upwards point scale with 27 points or more predicting high mortality.
Statistics
We used R/RStudio (version 1.5.46) for all analyses—commands in this paragraph shown in parentheses—and assessed cross-tables using the Fisher exact test (fisher.test). We used the Shapiro–Wilk (shapiro.test) test to assess consistency with the assumption of a normal distribution and calculated mean (mean) and standard deviation (SD) for normally distributed variables. We compared normally distributed variables between two groups with t tests (t.test) or the linear model (lm) command equivalent to a t test. For variables not following a normal distribution, we calculated median (median) and interquartile ranges (IQR) and compared them between groups with the Mann–Whitney U test (wilcox.test). Some variables were log-transformed to achieve consistency with a normal distribution for statistics requiring normally distributed variables, e.g., linear regression (lm) and correlation analysis (cor) using the Pearson method. We calculated receiver operating characteristics using the package pROC (version 1.18.0) and extracted the areas under the curve (AUC). Boxplots display the median as a horizontal line, the hinges indicate the interquartile range (IQR), and the whiskers extend to the smallest/largest value at most 1.5 * IQR of the hinges.
Results
Clinical data of the patients with SE
We included 28 patients (24 women, 4 men) with SE and a mean age of 69.4 years (SD = ± 15)). The median status duration was 44 h (IQR 80 h) and 22 patients (79%) survived SE. Patient characteristics including basic information regarding NfL levels are listed in Table 1. Twenty-one patients (75%) suffered from convulsive SE and seven (25%) from NCSE according to the ILAE classification. All patients showed impaired consciousness. Five patients had epilepsy before the event [unclassified epilepsy n = 2 (7%), structural epilepsy n = 3 (11%)]. Twelve of the remaining patients had a potential structural cause of SE (43%) and the etiology remained unknown for 11 patients (39%) and 6 of them fit the criteria of NORSE (new-onset refractory status epilepticus) [11]. Fifteen patients (54%) had normal standard parameters in the CSF investigation. Ten patients showed mild to moderate brain–blood-barrier dysfunction, two showed an inflammatory CSF, and one of them was due to an isolated elevated cell count. In 21 patients, the SE was refractory to treatment with a benzodiazepine and a classic antiepileptic drug (RSE, 75%) [2]. Burst suppression was used in 4 patients, either by propofol alone (n = 2) or by a combination of propofol and midazolam (n = 2). Six patients (21%) died in the hospital. The reasons for death were respiratory failure, peritonitis, cardiac failure, pneumonia, cardiogenic shock, and aspiration pneumonia.
NfL in status epilepticus versus control samples
We compared the serum NfL values in SE versus a large set of controls (n = 1186, Fig. 1). The control samples covered an age range from 20 to 83 years. Figure 1 shows that the NfL concentration of the Popgen, BiDirect, and MEMO control samples are all in the same range taking age into account. Therefore, we decided to pool all three control samples for analysis. Since NfL did not differ between sexes (p = 0.47, t test), but increases with age, we fitted a linear model with a quadratic term for serum NfL versus age and determined the 95% prediction intervals and extrapolated to the age of the oldest patient with SE (93 years) (Fig. 1). Only four out of 28 patients with SE showed NfL concentrations within the 95% NfL prediction interval for controls (Fig. 1). In most patients with SE the NfL concentrations grossly exceeded those of controls up to an approximately 100-fold increase for the highest NfL value in a patient (2142 pg/ml).
A NfL in patients with serum NfL < 60 pg/ml and controls, B in all patients and controls. Sample: Status epilepticus = SE patients from this study, Popgen = Popgen control sample measured on the same instrument concomitantly with the epilepsy samples, BiDirect, and MEMO: additional control samples measured on an identical but not the same instrument using the same Simoa kit. Blue interrupted line: NfL per age regression line for the joint control sample, gray band: standard error for the mean of the NfL per age regression line. Green interrupted lines: 95% prediction interval for the NfL per age regression, meaning that the linear model predicts that 95% of all population control samples will show NfL concentrations within these limits
Determinants of NfL concentrations in status epilepticus
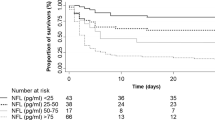
Table 2 shows the basic statistic measures presented in Table 1 stratified by the two different classifications of SE differentiating convulsive SE from NCSE and by survival. CSF and serum NfL concentrations showed a high correlation (r = 0.73, p = < 0.001, Supplementary Fig. 1). Both, NfL in CSF and serum were correlated with “status duration” (CSF: r = 0.58, p = 0.002; serum: r = 0.59, p = 0.001) and “status onset time to LP" (CSF: r = 0.64, p < 0.0004; serum: r = 0.55, p = 0.003, Fig. 2). Almost identical slopes of the regression lines for CSF and serum versus "status duration" and "status onset time to LP" indicate an identical relative increment of NfL concentrations in CSF and serum despite differing absolute values, arguing against a large influence of passage time between CSF and serum. "Status duration” and "status onset time to LP" were correlated with each other (r = 0.66, p = 0.0001, Supplementary Fig. 2), and “status onset time to LP” was always longer or equal to “status duration”. To account for the influence of these covariates (hereinafter referred to only as "covariates") we adjusted for them using linear regression analyses. Without and with adjustment for covariates, NfL concentrations did not differ significantly between patients with convulsive SE and NCSE according to the ILAE classification and the “convulsive only” versus “NCSE at any point during evolution” categories of the alternative classification (Fig. 3). Unadjusted NfL showed a trend towards higher concentrations in treatment RSE (p(CSF) = 0.05, p(serum) = 0.08). The status duration was longer in treatment RSE than in interruptible SE (mean 86.0 vs. 2.4 h).
NfL in convulsive SE and NCSE according to: A ILAE classification, B alternative classification. p p value, LM linear model (equivalent to t test), LMadj linear model adjusted for status duration and status onset time to LP. Serum NfL concentrations were multiplied by the factor 60 to allow plotting them side by side with the CSF NfL concentrations
Association of NfL with death and prognostic scales
Next, we assessed the correlation between NfL, death (Fig. 4a) and the prognostic scales EMSE_EAC (Fig. 4b) and STESS/mSTESS (Supplementary Fig. 3). NfL concentration were not associated with death without adjustment for the covariates (p(CSF) = 0.78, p(Serum) = 0.21) nor with adjustment (p(CSF) = 0.98, p(Serum) = 0.43, Fig. 4a). Next, we estimated the relation between the STESS and EMSE_EAC and death versus survival using Receiver-Operating-Characteristics (ROC) analysis. We obtained Areas-Under-the-Curve of 0.70 for STESS, 0.62 for mSTESS and 0.71 for EMSE-EAC (data not shown). These values point in the expected direction and are roughly comparable to other studies [12, 33]. We assessed the log-transformed EMSE as an interval-scaled normally distributed variable and STESS (7 levels) as well as mSTESS (9 levels) as ordinal categorical variables. EMSE scores were not correlated with NfL (Fig. 4b) neither without covariate adjustment (p(CSF) = 0.89, p(Serum) = 0.16) nor with adjustment (p(CSF) = 0.86, p(Serum) = 0.23).
NfL in A patients who survived or died, B versus EMSE-EAC score. p p value, LM linear model (equivalent to t test), LMadj linear model adjusted for status duration and status onset time to LP. Serum NfL concentrations were multiplied by the factor 60 to allow plotting them side by side with the CSF NfL concentrations
Due to the low number of individuals in some categories, a meaningful statistical analysis of the STESS and mSTESS data was not possible. However, Supplementary Fig. 3 shows that a trend towards higher NfL values in higher STESS or mSTESS categories is not apparent.
Discussion
Unlike most discussions, we start with the evident weaknesses of our study, because these are important not only for the interpretation of the data and results but also for the design of follow-up studies. Apart from the cross-sectional design and the relatively small patient sample, the major weaknesses are the large differences in the time to blood/CSF sampling and the high phenotypic variability of SE concerning the etiology, semiology, and duration. While the time to sampling can be standardized, the large phenotypic variability cannot. If standardization of time to sampling leads to NfL as a clinically useful prognostic marker remains to be shown. However, using a standardized sampling time, some status will be sampled after the end of the status, some during the status, and some status will continue a long time after sampling, continuing to release markers of neurodestruction. Despite its weak points, our study allows several meaningful conclusions. First, we show that Simoa technology is sensitive and reliable enough to measure NfL in serum as a proxy for CSF in SE. This is a finding in line with the other paper on that topic by Giovannini and colleagues [6]. In both studies, even though Giovannini used a different method to measure NfL, the correlation between CSF and serum NfL values in SE patients is very similar.
Second, we conclude from the parallel increase in concentration in serum and CSF that the passage time from CSF to blood must be too short to be meaningful even if the time from seizure to sample ascertainment is as short as ~ 2–6 h. We observe a log/log-linear increase of NfL in CSF and serum with status duration as well as time to sampling. We cannot tease apart with certainty whether status duration or time to sampling is the decisive parameter but we think that status duration has higher importance, at least if the sample was taken later than ~ 24 h (15 samples) because studies of “single hit” events like traumatic brain injury have shown that the NfL levels are rather stable after one day [1]. This observation from our study is relevant for any further use of NfL in clinical settings. Early transfer of CSF into serum means that measurement in serum is meaningful. In addition, the measurement of NfL in serum is easy to perform and repeat. Medically, our most important conclusion is that brain damage as measured by NfL might not differ by a very large extent between convulsive SE and non-convulsive SE, at least in patients with severely impaired consciousness. In our study, this is true for SE classified according to the ILAE classification but also for the alternative classification taking the development of the semiology over time into account. This finding suggests that concerning brain damage only, both convulsive, as well as non-convulsive SE, might turn out to be medical emergencies of comparable urgency if our results are corroborated by future studies. This conclusion is in agreement with animal studies as outlined in the introduction but has not yet been shown in humans [5, 22,23,24]. It is currently a matter of debate if both convulsive- and non-convulsive SE should be treated aggressively considering the adverse effects of sedatives and anticonvulsants [44]. Especially in the elderly, non-convulsive SE is commonly treated less aggressively than convulsive SE [29, 39]. If future studies confirm that non-convulsive SE too leads to significant neurodestruction, intensified treatment might be indicated. Additional findings are that NfL concentrations in most SE patients are higher than the age-adjusted 95th percentile of neurologically healthy controls which is in agreement with the only other study of NfL in SE [6]. Interestingly, the range of NfL concentrations overlapped between SE and controls also in this study [6]. The comparison of NfL between interruptible and treatment RSE pointed towards a possible difference in NfL concentrations just failing to reach statistical significance. However, we think that the main determinant of NfL concentrations is the status duration and not treatment refractoriness because (1) status duration was—as expected—much longer in treatment RSE and (2) status duration was highly linearly correlated on a log–log scale with NfL concentration across the whole range of status durations (Fig. 2). We found no association of NfL levels with scales predicting mortality nor with treatment refractoriness. We cannot exclude that this is caused either by the extreme heterogeneity of SE or the small sample size or our clinical sampling scheme hindering the detection of small differences. This important difference to the study by Giovannini and colleagues might in part be explainable by the fact that we adjusted for the status duration and time to CSF/serum sampling in all analyses while Giovannini and colleagues did not [6]. Larger studies taking the lessons from this study and the study by Giovannini et al. in the study design into account are required to draw firm conclusions. The integration of patients with differential diagnoses of SE is required to explore the diagnostic value of NfL in SE.
Conclusion
In summary, we show that NfL might be a clinically useful biomarker of SE, that neurodestruction, as measured by NfL, does not differ largely between convulsive SE and NCSE, and point out several caveats and suggestions for subsequent studies.
Data availability
Anonymized data will be shared by request from any qualified investigator.
References
Al Nimer F, Thelin E, Nystrom H, Dring AM, Svenningsson A, Piehl F, Nelson DW, Bellander BM (2015) Comparative assessment of the prognostic value of biomarkers in traumatic brain injury reveals an independent role for serum levels of neurofilament light. PLoS ONE 10:e0132177
Bleck TP (2005) Refractory status epilepticus. Curr Opin Crit Care 11:117–120
Boggs JG (2004) Mortality associated with status epilepticus. Epilepsy Curr 4:25–27
Celikbilek A, Tanik N, Sabah S, Borekci E, Akyol L, Ak H, Adam M, Suher M, Yilmaz N (2014) Elevated neurofilament light chain (NFL) mRNA levels in prediabetic peripheral neuropathy. Mol Biol Rep 41:4017–4022
de Curtis M, Rossetti AO, Verde DV, van Vliet EA, Ekdahl CT (2021) Brain pathology in focal status epilepticus: evidence from experimental models. Neurosci Biobehav Rev 131:834–846
Giovannini G, Bedin R, Ferraro D, Vaudano AE, Mandrioli J, Meletti S (2022) Serum neurofilament light as biomarker of seizure-related neuronal injury in status epilepticus. Epilepsia 63:e23–e29
Hanin A, Demeret S, Denis JA, Nguyen-Michel VH, Rohaut B, Marois C, Imbert-Bismut F, Bonnefont-Rousselot D, Levy P, Navarro V, Lambrecq V (2022) Serum neuron-specific enolase: a new tool for seizure risk monitoring after status epilepticus. Eur J Neurol 29:883–889
Hanin A, Denis JA, Frazzini V, Cousyn L, Imbert-Bismut F, Rucheton B, Bonnefont-Rousselot D, Marois C, Lambrecq V, Demeret S, Navarro V (2022) Neuron specific enolase, S100-beta protein and progranulin as diagnostic biomarkers of status epilepticus. J Neurol 269:3752–3760
Hanin A, Lambrecq V, Denis JA, Imbert-Bismut F, Rucheton B, Lamari F, Bonnefont-Rousselot D, Demeret S, Navarro V (2020) Cerebrospinal fluid and blood biomarkers of status epilepticus. Epilepsia 61:6–18
Hepner A, Porter J, Hare F, Nasir SS, Zetterberg H, Blennow K, Martin MG (2019) Serum neurofilament light, glial fibrillary acidic protein and tau are possible serum biomarkers for activity of brain metastases and gliomas. World J Oncol 10:169–175
Hirsch LJ, Gaspard N, van Baalen A, Nabbout R, Demeret S, Loddenkemper T, Navarro V, Specchio N, Lagae L, Rossetti AO, Hocker S, Gofton TE, Abend NS, Gilmore EJ, Hahn C, Khosravani H, Rosenow F, Trinka E (2018) Proposed consensus definitions for new-onset refractory status epilepticus (NORSE), febrile infection-related epilepsy syndrome (FIRES), and related conditions. Epilepsia 59:739–744
Kang BS, Kim DW, Kim KK, Moon HJ, Kim YS, Kim HK, Lee SY, Koo YS, Shin JW, Moon J, Sunwoo JS, Byun JI, Cho YW, Jung KY, Chu K, Lee SK (2016) Prediction of mortality and functional outcome from status epilepticus and independent external validation of STESS and EMSE scores. Crit Care 20:25
Krawczak M, Nikolaus S, von Eberstein H, Croucher PJ, El Mokhtari NE, Schreiber S (2006) PopGen: population-based recruitment of patients and controls for the analysis of complex genotype-phenotype relationships. Community Genet 9:55–61
Kuhle J, Kropshofer H, Haering DA, Kundu U, Meinert R, Barro C, Dahlke F, Tomic D, Leppert D, Kappos L (2019) Blood neurofilament light chain as a biomarker of MS disease activity and treatment response. Neurology 92:e1007–e1015
Lattanzi S, Giovannini G, Brigo F, Orlandi N, Trinka E, Meletti S (2021) Status epilepticus with prominent motor symptoms clusters into distinct electroclinical phenotypes. Eur J Neurol 28:2694–2699
Leitinger M, Beniczky S, Rohracher A, Gardella E, Kalss G, Qerama E, Hofler J, Hess Lindberg-Larsen A, Kuchukhidze G, Dobesberger J, Langthaler PB, Trinka E (2015) Salzburg consensus criteria for non-convulsive status epilepticus–approach to clinical application. Epilepsy Behav 49:158–163
Leitinger M, Holler Y, Kalss G, Rohracher A, Novak HF, Hofler J, Dobesberger J, Kuchukhidze G, Trinka E (2015) Epidemiology-based mortality score in status epilepticus (EMSE). Neurocrit Care 22:273–282
Leitinger M, Trinka E, Giovannini G, Zimmermann G, Florea C, Rohracher A, Kalss G, Neuray C, Kreidenhuber R, Hofler J, Kuchukhidze G, Granbichler C, Dobesberger J, Novak HF, Pilz G, Meletti S, Siebert U (2019) Epidemiology of status epilepticus in adults: a population-based study on incidence, causes, and outcomes. Epilepsia 60:53–62
Leitinger M, Trinka E, Zimmermann G, Granbichler CA, Kobulashvili T, Siebert U (2020) Epidemiology of status epilepticus in adults: apples, pears, and oranges—a critical review. Epilepsy Behav 103:106720
Mattsson N, Andreasson U, Zetterberg H, Blennow K, Alzheimer’s Disease Neuroimaging I (2017) Association of plasma neurofilament light with neurodegeneration in patients with Alzheimer disease. JAMA Neurol 74:557–566
Mattsson N, Bremell D, Anckarsater R, Blennow K, Anckarsater H, Zetterberg H, Hagberg L (2010) Neuroinflammation in Lyme neuroborreliosis affects amyloid metabolism. BMC Neurol 10:51
Meldrum BS (1993) Excitotoxicity and selective neuronal loss in epilepsy. Brain Pathol 3:405–412
Meldrum BS, Brierley JB (1973) Prolonged epileptic seizures in primates. Ischemic cell change and its relation to ictal physiological events. Arch Neurol 28:10–17
Meldrum BS, Vigouroux RA, Brierley JB (1973) Systemic factors and epileptic brain damage. Prolonged seizures in paralyzed, artificially ventilated baboons. Arch Neurol 29:82–87
Novy J, Logroscino G, Rossetti AO (2010) Refractory status epilepticus: a prospective observational study. Epilepsia 51:251–256
Olsson B, Portelius E, Cullen NC, Sandelius A, Zetterberg H, Andreasson U, Hoglund K, Irwin DJ, Grossman M, Weintraub D, Chen-Plotkin A, Wolk D, McCluskey L, Elman L, Shaw LM, Toledo JB, McBride J, Hernandez-Con P, Lee VM, Trojanowski JQ, Blennow K (2019) Association of cerebrospinal fluid neurofilament light protein levels with cognition in patients with dementia, motor neuron disease, and movement disorders. JAMA Neurol 76:318–325
Pollak L, Gandelman-Marton R, Margolin N, Boxer M, Blatt I (2014) Clinical and electroencephalographic findings in acutely ill adults with non-convulsive vs convulsive status epilepticus. Acta Neurol Scand 129:405–411
Rissin DM, Kan CW, Campbell TG, Howes SC, Fournier DR, Song L, Piech T, Patel PP, Chang L, Rivnak AJ, Ferrell EP, Randall JD, Provuncher GK, Walt DR, Duffy DC (2010) Single-molecule enzyme-linked immunosorbent assay detects serum proteins at subfemtomolar concentrations. Nat Biotechnol 28:595–599
Rossetti AO, Hirsch LJ, Drislane FW (2019) Nonconvulsive seizures and nonconvulsive status epilepticus in the neuro ICU should or should not be treated aggressively: a debate. Clin Neurophysiol Pract 4:170–177
Rossetti AO, Logroscino G, Bromfield EB (2006) A clinical score for prognosis of status epilepticus in adults. Neurology 66:1736–1738
Rossetti AO, Logroscino G, Milligan TA, Michaelides C, Ruffieux C, Bromfield EB (2008) Status Epilepticus Severity Score (STESS): a tool to orient early treatment strategy. J Neurol 255:1561–1566
Rubsamen N, Maceski A, Leppert D, Benkert P, Kuhle J, Wiendl H, Peters A, Karch A, Berger K (2021) Serum neurofilament light and tau as prognostic markers for all-cause mortality in the elderly general population-an analysis from the MEMO study. BMC Med 19:38
Sairanen JJ, Kantanen AM, Hyppola HT, Kalviainen RK (2020) Outcome of status epilepticus and the predictive value of the EMSE and STESS scores: a prospective study. Seizure 75:115–120
Shahim P, Politis A, van der Merwe A, Moore B, Ekanayake V, Lippa SM, Chou YY, Pham DL, Butman JA, Diaz-Arrastia R, Zetterberg H, Blennow K, Gill JM, Brody DL, Chan L (2020) Time course and diagnostic utility of NfL, tau, GFAP, and UCH-L1 in subacute and chronic TBI. Neurology 95:e623–e636
Steinacker P, Anderl-Straub S, Diehl-Schmid J, Semler E, Uttner I, von Arnim CAF, Barthel H, Danek A, Fassbender K, Fliessbach K, Foerstl H, Grimmer T, Huppertz HJ, Jahn H, Kassubek J, Kornhuber J, Landwehrmeyer B, Lauer M, Maler JM, Mayer B, Oeckl P, Prudlo J, Schneider A, Volk AE, Wiltfang J, Schroeter ML, Ludolph AC, Otto M (2018) Serum neurofilament light chain in behavioral variant frontotemporal dementia. Neurology 91:e1390–e1401
Suzuki Y, Toribe Y, Goto M, Kato T, Futagi Y (1999) Serum and CSF neuron-specific enolase in patients with West syndrome. Neurology 53:1761–1764
Tanabe T, Suzuki S, Hara K, Shimakawa S, Wakamiya E, Tamai H (2001) Cerebrospinal fluid and serum neuron-specific enolase levels after febrile seizures. Epilepsia 42:504–507
Teismann H, Wersching H, Nagel M, Arolt V, Heindel W, Baune BT, Wellmann J, Hense HW, Berger K (2014) Establishing the bidirectional relationship between depression and subclinical arteriosclerosis–rationale, design, and characteristics of the BiDirect Study. BMC Psychiatry 14:174
Treiman DM, Walker MC (2006) Treatment of seizure emergencies: convulsive and non-convulsive status epilepticus. Epilepsy Res 68(Suppl 1):S77-82
Trinka E, Cock H, Hesdorffer D, Rossetti AO, Scheffer IE, Shinnar S, Shorvon S, Lowenstein DH (2015) A definition and classification of status epilepticus—report of the ILAE task force on classification of status epilepticus. Epilepsia 56:1515–1523
Trinka E, Leitinger M (2015) Which EEG patterns in coma are nonconvulsive status epilepticus? Epilepsy Behav 49:203–222
Varhaug KN, Torkildsen O, Myhr KM, Vedeler CA (2019) Neurofilament light chain as a biomarker in multiple sclerosis. Front Neurol 10:338
Verde F, Otto M, Silani V (2021) Neurofilament light chain as biomarker for amyotrophic lateral sclerosis and frontotemporal dementia. Front Neurosci 15:679199
Walker MC (2007) Treatment of nonconvulsive status epilepticus. Int Rev Neurobiol 81:287–297
Funding
Open Access funding enabled and organized by Projekt DEAL. The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by NGM, JD, ET, FL, WL, AF, KB, JK and GK. The first draft of the manuscript was written by NGM, GK and JD and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
NGM received travel grants from Eisai Pharma and Angelini Pharma. Lecture fees were given by Jazz Pharma and Angelini Pharma. Research support was granted by Jazz Pharma, Angelini Pharma, Eisai Pharma, UCB Pharma, LivaNova and Desitin Pharma. He also receives funding from the Ministry of Health (Schleswig–Holstein) and the German Research Council (DFG). Non-financial interests: none. JD has no relevant financial or non-financial interests to disclose. ET has no relevant financial or non-financial interests to disclose. FL reports having received speaker’s honoraria from Grifols, Biogen, Novartis, Roche, Alexion, serving on advisory boards for Roche and Biogen and receiving research funding from the German Ministry of Education and Research, the German Society for Laboratory Medicine (DGKL) and the German Research Council (DFG). He works for an academic institution offering commercial autoantibody testing. Non-financial interests: none. WL has no relevant financial or non-financial interests to disclose. AF has no relevant financial or non-financial interests to disclose. KB receives for the MEMO Study funding by the German Research Society (Deutsche Forschungsgemeinschaft DFG, grant: BE1996/1-1). The measurement of NfL was done through funds from the Institute of Epidemiology and Social Medicine, University of Muenster. The BiDirect Study is supported by grants of the German Ministry of Research and Education (BMBF) to the University of Muenster (01ER0816 and 01ER1506). Non-financial interests: none. JK received speaker fees, research support, travel support, and/or served on advisory boards by Swiss MS Society, Swiss National Research Foundation (320030_189140/1), University of Basel, Progressive MS Alliance, Bayer, Biogen, Bristol Myers Squibb, Celgene, Merck, Novartis, Octave Bioscience, Roche, Sanofi. Non-financial interests: none. GK receives non-project-related financial support from the BMBF project CONNECT-GENERATE: Genetics of autoimmune encephalitis. Non-financial interests: none. The authors declare that they have no conflict of interest concerning this work.
Ethical standards
The study was approved by the Ethics Committee of the Medical Faculty of the Christian-Albrechts-University, Kiel, Germany (D 480/20). The study was conducted following the World Medical Association Declaration of Helsinki. The use of retrospective data was reviewed and confirmed by the Ethics Committee. The data protection guidelines were also observed. The identity of the subjects is protected.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Margraf, N.G., Dargvainiene, J., Theel, E. et al. Neurofilament light (NfL) as biomarker in serum and CSF in status epilepticus. J Neurol 270, 2128–2138 (2023). https://doi.org/10.1007/s00415-022-11547-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-022-11547-4