Abstract
Background
Imaging studies investigating cerebellar gray matter (GM) in essential tremor (ET) showed conflicting results. Moreover, no large study explored the cerebellum in ET patients with resting tremor (rET), a syndrome showing enhanced blink reflex recovery cycle (BRrc).
Objective
To investigate cerebellar GM in ET and rET patients using voxel-based morphometry (VBM) analysis.
Methods
Seventy ET patients with or without resting tremor and 39 healthy controls were enrolled. All subjects underwent brain 3 T-MRI and BRrc recording. We compared the cerebellar GM volumes between ET (n = 40) and rET (n = 30) patients and controls through a VBM analysis. Moreover, we investigated possible correlations between cerebellar GM volume and R2 component of BRrc.
Results
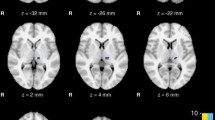
rET and ET patients had similar disease duration. All rET patients and none of ET patients had enhanced BRrc. No differences in the cerebellar volume were found when ET and rET patients were compared to each other or with controls. By considering together the two tremor syndromes in a large patient group, the VBM analysis showed bilateral clusters of reduced GM volumes in Crus II in comparison with controls. The linear regression analysis in rET patients revealed a cluster in the left Crus II where the decrease in GM volume correlated with the R2BRrc increase.
Conclusion
Our study suggests that ET and rET are different tremor syndromes with similar mild cerebellar gray matter involvement. In rET patients, the left Crus II may play a role in modulating the brainstem excitability, encouraging further studies on the role of cerebellum in these patients.

Similar content being viewed by others
Availability of data and materials
The data that support the results of this study are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
References
Agren R, Awad A, Blomstedt P, Fytagoridis A (2021) Voxel-based morphometry of cerebellar lobules in essential tremor. Front Aging Neurosci 13:667854. https://doi.org/10.3389/fnagi.2021.667854
Dyke JP, Cameron E, Hernandez N, Dydak U, Louis ED (2017) Gray matter density loss in essential tremor: a lobule by lobule analysis of the cerebellum. Cerebellum Ataxias 4:10. https://doi.org/10.1186/s40673-017-0069-3
Caligiuri ME, Arabia G, Barbagallo G, Lupo A, Morelli M, Nisticò R, Novellino F, Quattrone A, Salsone M, Vescio B (2017) Structural connectivity differences in essential tremor with and without resting tremor. J Neurol 264:1865–1874
Li JY, Suo XL, Li NN, Lei D, Lu ZJ, Wang L, Peng JX, Duan LR, Jing X, Yi J, Gong QY, Peng R (2021) Altered spontaneous brain activity in essential tremor with and without resting tremor: a resting-state fMRI study. MAGMA 34:201–212. https://doi.org/10.1002/hbm.25425
Louis ED (2016) Linking essential tremor to the cerebellum: neuropathological evidence. Cerebellum 15:235–242. https://doi.org/10.1007/s12311-015-0692-6
Luo R, Pan P, Xu Y, Chen L (2019) No reliable gray matter changes in essential tremor. Neurol Sci 40:2051–2063. https://doi.org/10.1007/s10072-019-03933-0
Nicoletti V, Cecchi P, Frosini D, Pesaresi I, Fabbri S, Diciotti S, Bonuccelli U, Cosottini M, Ceravolo R (2015) Morphometric and functional MRI changes in essential tremor with and without resting tremor. J Neurol 262:719–728. https://doi.org/10.1007/s00415-014-7626-y
Novellino F, Nicoletti G, Cherubini A, Caligiuri ME, Nisticò R, Salsone M, Morelli M, Arabia G, Cavalli SM, Vaccaro MG, Chiriaco C, Quattrone A (2016) Cerebellar involvement in essential tremor with and without resting tremor: a diffusion tensor imaging study. Parkinsonism Relat Disord 27:61–66. https://doi.org/10.1016/j.parkreldis.2016.03.022
Prasad S, Rastogi B, Shah A, Bhalsing KS, Ingalhalikar M, Saini J, Yadav R, Pal PK (2018) DTI in essential tremor with and without rest tremor: two sides of the same coin? Mov Disord 33:1820–1821. https://doi.org/10.1002/mds.27459
Erro R, Sorrentino C, Russo M, Barone P (2022) Essential tremor plus rest tremor: current concepts and controversies. J Neural Transm (Vienna) 129:835–846. https://doi.org/10.1007/s00702-022-02516-2
Bellows S, Jankovic J (2022) Parkinsonism and tremor syndromes. J Neurol Sci 433:120018. https://doi.org/10.1016/j.jns.2021.120018
Cohen O, Pullman S, Jurewicz E, Watner D, Louis ED (2003) Rest tremor in patients with essential tremor: prevalence, clinical correlates, and electrophysiologic characteristics. Arch Neurol 60:405–410. https://doi.org/10.1001/archneur.60.3.405
Rajalingam R, Breen DP, Lang AE, Fasano A (2018) Essential tremor plus is more common than essential tremor: insights from the reclassification of a cohort of patients with lower limb tremor. Parkinsonism Relat Disord 56:109–110. https://doi.org/10.1016/j.parkreldis.2018.06.029
Louis ED, Hernandez N, Michalec M (2015) Prevalence and correlates of rest tremor in essential tremor: cross-sectional survey of 831 patients across four distinct cohorts. Eur J Neurol 22:927–932. https://doi.org/10.1111/ene.12683
Peng J, Li N, Li J, Duan L, Chen C, Zeng Y, Xi J, Jiang Y, Peng R (2022) Reclassification of patients with tremor syndrome and comparisons of essential tremor and essential tremor-plus patients. J Neurol 269:3653–3662. https://doi.org/10.1007/s00415-022-10985-4
Louis ED, Huey ED, Cosentino S (2021) Features of “ET plus” correlate with age and tremor duration: “ET plus” may be a disease stage rather than a subtype of essential tremor. Parkinsonism Relat Disord 91:42–47. https://doi.org/10.1016/j.parkreldis.2021.08.017
Louis ED, Bares M, Benito-Leon J, Fahn S, Frucht SJ, Jankovic J, Ondo WG, Pal PK, Tan EK (2020) Essential tremor-plus: a controversial new concept. Lancet Neurol 19:266–270. https://doi.org/10.1016/S1474-4422(19)30398-9
Lolekha P, Dharmasaroja P, Uransilp N, Sukphulloprat P, Muengtaweepongsa S, Kulkantrakorn K (2022) The differences in clinical characteristics and natural history between essential tremor and essential tremor plus. Sci Rep 12:7669. https://doi.org/10.1038/s41598-022-11775-8
Erro R, Pilotto A, Esposito M, Olivola E, Nicoletti A, Lazzeri G, Magistrelli L, Dallocchio C, Marchese R, Bologna M, Tessitore A, Misceo S, Gigante AF, Terranova C, Moschella V, di Biase L, Di Giacopo R, Morgante F, Valentino F, De Rosa A, Trinchillo A, Malaguti MC, Brusa L, Matinella A, Di Biasio F, Paparella G, De Micco R, Contaldi E, Modugno N, Di Fonzo A, Padovani A, Barone P, TITAN Study Group (2022) The Italian tremor Network (TITAN): rationale, design and preliminary findings. Neurol Sci. https://doi.org/10.1007/s10072-022-06104-w
Bhatia KP, Bain P, Bajaj N, Elble RJ, Hallett M, Louis ED, Raethjen J, Stamelou M, Testa CM, Deuschl G, Tremor Task Force of the International P, Movement Disorder S (2018) Consensus Statement on the classification of tremors. from the task force on tremor of the International Parkinson and Movement Disorder Society. Mov Disord 33:75–87. https://doi.org/10.1002/mds.27121
Nistico R, Pirritano D, Novellino F, Salsone M, Morelli M, Valentino P, Condino F, Arabia G, Quattrone A (2012) Blink reflex recovery cycle in patients with essential tremor associated with resting tremor. Neurology 79:1490–1495. https://doi.org/10.1212/WNL.0b013e31826d5f83
Nistico R, Salsone M, Vescio B, Morelli M, Trotta M, Barbagallo G, Arabia G, Quattrone A (2014) Blink reflex recovery cycle distinguishes essential tremor with resting tremor from de novo Parkinson’s disease: an exploratory study. Parkinsonism Relat Disord 20:153–156. https://doi.org/10.1016/j.parkreldis.2013.10.006
Quattrone A, Nistico R, Morelli M, Arabia G, Crasa M, Vescio B, Mechelli A, Cascini GL, Quattrone A (2021) Rest tremor pattern predicts DaTscan ((123) I-Ioflupane) result in tremulous disorders. Mov Disord 36:2964–2966. https://doi.org/10.1002/mds.28797
Fahn S, Tolosa E, Marín C (1993) Clinical rating scale for tremor. Parkinson’s Dis Movement Disord 2:271–280
Mechelli A, Quattrone A, Nistico R, Crasa M, La Torre D, Vescio B, Quattrone A (2022) Blink reflex recovery cycle distinguishes patients with idiopathic normal pressure hydrocephalus from elderly subjects. J Neurol 269:1007–1012. https://doi.org/10.1007/s00415-021-10687-3
Quattrone A, Morelli M, Quattrone A, Vescio B, Nigro S, Arabia G, Nistico R, Novellino F, Salsone M, Arcuri P, Luca A, Mazzuca A, Alessio C, Rocca F, Caracciolo M (2020) Magnetic Resonance Parkinsonism Index for evaluating disease progression rate in progressive supranuclear palsy: a longitudinal 2-year study. Parkinsonism Relat Disord 72:1–6. https://doi.org/10.1016/j.parkreldis.2020.01.019
Diedrichsen J, Zotow E (2015) Surface-based display of volume-averaged cerebellar imaging data. PLoS One 10:e0133402. https://doi.org/10.1371/journal.pone.0133402
Li JY, Lu ZJ, Suo XL, Li NN, Lei D, Wang L, Peng JX, Duan LR, Xi J, Jiang Y, Gong QY, Peng R (2020) Patterns of intrinsic brain activity in essential tremor with resting tremor and tremor-dominant Parkinson’s disease. Brain Imaging Behav 14:2606–2617. https://doi.org/10.1007/s11682-019-00214-4
Nistico R, Pirritano D, Salsone M, Valentino P, Novellino F, Condino F, Bono F, Quattrone A (2012) Blink reflex recovery cycle in patients with dystonic tremor: a cross-sectional study. Neurology 78:1363–1365. https://doi.org/10.1212/WNL.0b013e3182518316
Louis ED, Asabere N, Agnew A, Moskowitz CB, Lawton A, Cortes E, Faust PL, Vonsattel JP (2011) Rest tremor in advanced essential tremor: a post-mortem study of nine cases. J Neurol Neurosurg Psychiatry 82:261–265. https://doi.org/10.1136/jnnp.2010.215681
Gionco JT, Hartstone WG, Martuscello RT, Kuo SH, Faust PL, Louis ED (2021) Essential tremor versus “ET-plus”: a detailed postmortem study of cerebellar pathology. Cerebellum 20:904–912. https://doi.org/10.1007/s12311-021-01263-6
Pellegrini JJ, Evinger C (1997) Role of cerebellum in adaptive modification of reflex blinks. Learn Mem 4:77–87. https://doi.org/10.1101/lm.4.1.77
Funding
No funding to declare.
Author information
Authors and Affiliations
Contributions
AS, AQ and AQ contributed to the study conception and design. Data collection was performed by AQ, MC, RN, MGV and FC. Statistical analysis was performed by AS, MGB and VG. The first draft of the manuscript was written by AS, AQ and AQ. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical approval
All patients in our study gave their informed consent prior to their inclusion in the study. Approval of our study was obtained from the ethics committee of Magna Graecia University review board, Catanzaro, Italy. The procedures used in this study adhere to the tenets of the Declaration of Helsinki.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent to publication
All patients signed informed consent regarding publishing their data.
Rights and permissions
About this article
Cite this article
Sarica, A., Quattrone, A., Crasà, M. et al. Cerebellar voxel-based morphometry in essential tremor. J Neurol 269, 6029–6035 (2022). https://doi.org/10.1007/s00415-022-11291-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-022-11291-9