Abstract
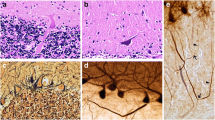
Essential tremor (ET) is among the most prevalent movement disorders, and by some accounts, the most common form of cerebellar degeneration. Over the past 15 years, we have carefully documented a large number of postmortem changes within the cerebellum; these cerebellar changes differ significantly between ET and controls. A recent Consensus Classification of tremor proposed that ET patients with other neurological signs aside from action tremor (e.g., parkinsonism, ataxia, cognitive changes, dystonia) should be segregated off as “ET-plus”. This diagnostic concept has raised considerable controversy and its validity is not yet established. Indeed, “ET-plus” has not been distinguished from ET based on differences in genetics, pathology or prognosis. Here we determine whether ET cases differ from "ET-plus" cases in underlying pathological changes in the postmortem brain. We examined postmortem brains from 50 ET cases (24 ET and 26 ET-plus), using a set of 14 quantitative metrics of cerebellar pathology determined by histologic and immunohistochemical methods. These metrics reflect changes across the Purkinje cell (PC) body (PC counts, empty baskets, heterotopias), PC dendrites (swellings), PC axon (torpedoes and associated axonal changes), basket cell axonal hypertrophy and climbing fiber-PC dendrite synaptic changes. ET and ET-plus were similar with respect to 13 of 14 cerebellar pathologic metrics (p > 0.05). Only one metric, the linear density of thickened PC axon profiles, differed between these groups (ET = 0.529 ± 0.397, ET-plus = 0.777 ± 0.477, p = 0.013), although after correcting for multiple comparisons, there were no differences. If ET-plus were indeed a different entity, then the underlying pathological basis should be distinct from that of ET. This study demonstrated there were no pathological differences in cerebellar cortex between ET versus ET-plus cases. These data do not support the notion that ET and ET-plus represent distinct clinical-pathological entities.


Similar content being viewed by others
References
Louis ED, Ferreira JJ. How common is the most common adult movement disorder? Update on the worldwide prevalence of essential tremor. Mov Disord. 2010;25:534–41.
Louis ED, Faust PL. Essential tremor: the most common form of cereb ellar degeneration? Cerebellum Ataxias. 2020;7:12. https://doi.org/10.1186/s40673-020-00121-1.
Louis ED, Faust PL. Essential Tremor Within the Broader Context of Other Forms of Cerebellar Degeneration. Cerebellum. 2020;19(6):879–96. https://doi.org/10.1007/s12311-020-01160-4.
Filip P, Lungu OV, Manto MU, Bareš M. Linking Essential Tremor to the Cerebellum: Physiological Evidence. Cerebellum. 2016;15(6):774–80. https://doi.org/10.1007/s12311-015-0740-2.
Bhatia KP, Bain P, Bajaj N, et al. Consensus statement on the classification of tremors from the task force on tremor of the International Parkinson and Movement Disorder Society. Mov Disord. 2018;33:75–87.
Pandey S, Bhattad S, Hallett M. The problem of questionable dystonia in the diagnosis of ‘essential tremor-plus’ Tremor Other Hyperkinet Mov (NY). 2020;10:27. https://doi.org/10.5334/tohm.539.
Louis ED. “Essential Tremor Plus”: A Problematic Concept: Implications for Clinical and Epidemiological Studies of Essential Tremor. Neuroepidemiology. 2020;54:180–4.
Louis ED, Bares M, Benito-Leon J, et al. Essential tremor-plus: a controversial new concept. Lancet Neurol. 2020;19:266–70.
Vidailhet M. Essential tremor-plus: a temporary label. Lancet Neurol. 2020;19:202–3.
Louis ED. Essential tremor: “Plus” or “Minus”. Perhaps now is the time to adopt the term “the essential tremors.” Parkinsonism RelatDisord. 2018;56:111–2.
Prasad S, Pal PK. Reclassifying essential tremor: Implications for the future of past research. Mov Disord. 2019;34:437.
Louis ED, Kerridge CA, Chatterjee D, et al. Contextualizing the pathology in the essential tremor cerebellar cortex: a patholog-omics approach. Acta Neuropathol. 2019;138(5):859–76. https://doi.org/10.1007/s00401-019-02043-7.
Louis ED, Faust PL, Vonsattel JP, et al. Neuropathological changes in essential tremor: 33 cases compared with 21 controls. Brain. 2007;130(Pt 12):3297–307. https://doi.org/10.1093/brain/awm266.
Babij R, Lee M, Cortés E, Vonsattel JP, Faust PL, Louis ED. Purkinje cell axonal anatomy: quantifying morphometric changes in essential tremor versus control brains. Brain. 2013;136(Pt 10):3051–61. https://doi.org/10.1093/brain/awt238.
Lee PJ, Kerridge CA, Chatterjee D, Koeppen AH, Faust PL, Louis ED. A Quantitative Study of Empty Baskets in Essential Tremor and Other Motor Neurodegenerative Diseases. J Neuropathol Exp Neurol. 2019;78(2):113–22. https://doi.org/10.1093/jnen/nly114.
Louis ED, Kuo SH, Tate WJ, et al. Heterotopic Purkinje Cells: a Comparative Postmortem Study of Essential Tremor and Spinocerebellar Ataxias 1, 2, 3, and 6. Cerebellum. 2018;17(2):104–10. https://doi.org/10.1007/s12311-017-0876-3.
Choe M, Cortés E, Vonsattel JP, Kuo SH, Faust PL, Louis ED. Purkinje cell loss in essential tremor: Random sampling quantification and nearest neighbor analysis. Mov Disord. 2016;31(3):393–401. https://doi.org/10.1002/mds.26490.
Louis ED, Lee M, Babij R, et al. Reduced Purkinje cell dendritic arborization and loss of dendritic spines in essential tremor. Brain. 2014;137(Pt 12):3142–8. https://doi.org/10.1093/brain/awu314.
Lin CY, Louis ED, Faust PL, Koeppen AH, Vonsattel JP, Kuo SH. Abnormal climbing fibre-Purkinje cell synaptic connections in the essential tremor cerebellum. Brain. 2014;137(Pt 12):3149–59. https://doi.org/10.1093/brain/awu281.
Linn BS, Linn MW, Gurel L. Cumulative illness rating scale. J Am Geriatr Soc. 1968;16(5):622–6. https://doi.org/10.1111/j.1532-5415.1968.tb02103.x.
Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189–98. https://doi.org/10.1016/0022-3956(75)90026-6.
Movement Disorder Society Task Force on Rating Scales for Parkinson’s Disease. The Unified Parkinson’s Disease Rating Scale (UPDRS): status and recommendations. MovDisord. 2003;18(7):738–50. https://doi.org/10.1002/mds.10473.
Louis ED, Hernandez N, Alcalay RN, Tirri DJ, Ottman R, Clark LN. Prevalence and features of unreported dystonia in a family study of “pure” essential tremor. Parkinsonism Relat Disord. 2013;19(3):359–62. https://doi.org/10.1016/j.parkreldis.2012.09.015.
Louis ED, Frucht SJ, Rios E. Intention tremor in essential tremor: Prevalence and association with disease duration. Mov Disord. 2009;24(4):626–7. https://doi.org/10.1002/mds.22370.
Braak H, Braak E. Neuropathological staging of Alzheimer-related changes. Acta Neuropathol. 1991;82(4):239–59. https://doi.org/10.1007/BF00308809.
Mirra SS, Heyman A, McKeel D, et al. The Consortium to Establish a Registry for Alzheimer’s Disease (CERAD). Part II. Standardization of the neuropathologic assessment of Alzheimer’s disease. Neurology. 1991;41(4):479–86. https://doi.org/10.1212/wnl.41.4.479.
Louis ED, Babij R, Ma K, Cortés E, Vonsattel JP. Essential tremor followed by progressive supranuclear palsy: postmortem reports of 11 patients. J Neuropathol Exp Neurol. 2013;72(1):8–17. https://doi.org/10.1097/NEN.0b013e31827ae56e.
Louis ED, Kuo SH, Tate WJ, Kelly GC, Faust PL. Cerebellar pathology in childhood-onset vs. adult-onset essential tremor. NeurosciLett. 2017;659:69–74. https://doi.org/10.1016/j.neulet.2017.08.072.
Kuo SH, Wang J, Tate WJ, Pan MK, Kelly GC, Gutierrez J, Cortes EP, Vonsattel JG, Louis ED, Faust PL. Cerebellar Pathology in Early Onset and Late Onset Essential Tremor. Cerebellum. 2017;16(2):473–82. https://doi.org/10.1007/s12311-016-0826-5.
Louis ED, Kuo SH, Wang J, Tate WJ, Pan MK, Kelly GC, Gutierrez J, Cortes EP, Vonsattel JG, Faust PL. Cerebellar Pathology in Familial vs. Sporadic Essential Tremor Cerebellum. 2017;16(4):786–91. https://doi.org/10.1007/s12311-017-0853-x.
Acknowledgments
This work was supported by the National Institutes of Health (NINDS R01 NS088257).
Author information
Authors and Affiliations
Contributions
The following is a list of all authors and their contributions in the project and the preparation of the manuscript. These include but are not restricted to: (1) research project: A. conception, B. organization, C. execution; (2) statistical analysis: A. design, B. execution, C. review and critique; and (3) manuscript: A. writing of the first draft, B. review and critique.
Mr. Gionco: 1B, 1C, 2B, 2C, 3A, 3B.
Ms. Hartstone: 1C, 3B.
Dr. Martuscello: 1C, 3B.
Dr. Kuo: 1A, 1B, 3B.
Dr. Faust: 1A, 1B, 1C, 2A, 2C, 3B.
Dr. Louis: 1A, 1B, 2A, 2B, 2C, 3B.
Corresponding author
Ethics declarations
Conflicts of Interest
There are no conflicts of interest or competing financial interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gionco, J.T., Hartstone, W.G., Martuscello, R.T. et al. Essential Tremor versus “ET-plus”: A Detailed Postmortem Study of Cerebellar Pathology. Cerebellum 20, 904–912 (2021). https://doi.org/10.1007/s12311-021-01263-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-021-01263-6