Abstract
Background
Fatigue is one of the most commonly reported subjective symptoms following traumatic brain injury (TBI). The aims were to assess frequency of fatigue over the first 6 months after TBI, and examine whether fatigue changes could be predicted by demographic characteristics, injury severity and comorbidities.
Methods
Patients with acute TBI admitted to 65 trauma centers were enrolled in the study Collaborative European NeuroTrauma Effectiveness Research in TBI (CENTER-TBI). Subjective fatigue was measured by single item on the Rivermead Post-Concussion Symptoms Questionnaire (RPQ), administered at baseline, three and 6 months postinjury. Patients were categorized by clinical care pathway: admitted to an emergency room (ER), a ward (ADM) or an intensive care unit (ICU). Injury severity, preinjury somatic- and psychiatric conditions, depressive and sleep problems were registered at baseline. For prediction of fatigue changes, descriptive statistics and mixed effect logistic regression analysis are reported.
Results
Fatigue was experienced by 47% of patients at baseline, 48% at 3 months and 46% at 6 months. Patients admitted to ICU had a higher probability of experiencing fatigue than those in ER and ADM strata. Females and individuals with lower age, higher education, more severe intracranial injury, preinjury somatic and psychiatric conditions, sleep disturbance and feeling depressed postinjury had a higher probability of fatigue.
Conclusion
A high and stable frequency of fatigue was found during the first 6 months after TBI. Specific socio-demographic factors, comorbidities and injury severity characteristics were predictors of fatigue in this study.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fatigue is defined as "the awareness of a decreased capacity for mental and/or physical activity, because of an imbalance in the availability, utilization or restoration of resources needed to perform activities" [1]. It is one of the most commonly reported subjective symptoms following traumatic brain injury (TBI). Precise estimates of post-TBI fatigue vary greatly (21–73%) [24], but it consistently exceeds the prevalence of fatigue in the general population (10–20%) [21]. The existing evidence shows that self-reported fatigue decreases over time after TBI, but some patients continue to report persisting fatigue or may even report an increase in fatigue over time [27]. A previous study assessing fatigue pathways over the first year after TBI showed an increase of fatigue after severe TBI (sTBI), stable fatigue after moderate TBI and a reduction of fatigue levels over time after mild TBI (mTBI) [4]. Other studies have suggested that longstanding fatigue is not limited to patients with sTBI, and may be exacerbated or caused by emotional and cognitive symptoms, sleep disturbances, and pain across all injury severities [29, 30].
Premorbid variables such as emotional/mental health problems, personality traits, pre-existing fatigue, and other medical comorbidities may contribute additionally to vulnerability for the development of fatigue following TBI [6, 12]. The association between fatigue and personal factors such as age, gender, and education have been assessed to a lesser extent [6, 16, 27]. Gender differences in prevalence and severity of fatigue have been reported after stroke [20]. However, studies after TBI found inconsistent effects of age and gender [7, 12, 16, 27], whereas higher education was associated with higher levels of fatigue [41].
The majority of previous studies have been conducted with patients after mTBI, and at greatly varying time-points postinjury [24]. Despite a growing body of literature on fatigue after TBI, there is a lack of large-scale studies on longitudinal fatigue changes across both acute clinical care pathways, and injury severity. Such studies are important to increase the knowledge concerning which factors contribute the most to the occurrence and persistence of fatigue, as well as aid the development of preventive efforts and targeted fatigue interventions.
Several scales have been developed for the assessment of different aspects of fatigue for different purposes [5, 24, 40]. These scales often contain numerous questions [18], which may present a burden to the patients when other symptoms and aspects after TBI also need to be assessed. The Rivermead Post-Concussion Symptoms Questionnaire (RPQ) is a self-rated questionnaire assessing the presence and severity of common post-concussion symptoms after TBI [17, 39]. Fatigue is the most frequently affirmed symptom reported in the questionnaire, which renders this item useful to evaluate progress or regression of symptom severity [39]. In factor analysis of the RPQ, fatigue loads either on somatic/physiological symptoms [31] or on emotional/somatic or cognitive symptoms [3], and is strongly associated with limitations in daily functioning [35]. Taken together, the single fatigue item in the RPQ seems to provide a good estimate of the subjective experience of general fatigue after TBI. Therefore, we used it in a large sample of patients from the Collaborative European NeuroTrauma Effectiveness Research in Traumatic Brain Injury (CENTER-TBI) observational study [22].
The aims of this study are:
-
1.
To assess frequency and severity of fatigue at baseline (i.e., at time of study inclusion), 3 and 6 months post-TBI across age, gender, patients’ clinical pathways in the acute phase and severity of injury.
-
2.
To investigate whether socio-demographic factors, injury severity characteristics, and pre- and postinjury comorbidities predict fatigue changes across the first 6 months following TBI.
We hypothesize that fatigue presents a significant burden for the majority of patients after TBI regardless of injury severity and time since injury.
Methods
Study design
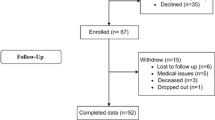
Patients were selected from the core study of the CENTER-TBI project; a multicenter, prospective observational longitudinal cohort study, conducted in Europe and Israel [22], which enrolled patients with all severities of TBI who presented to 65 participating centers between December 19, 2014 and December 17, 2017. Inclusion criteria were a clinical diagnosis of TBI, an indication for CT scanning, presenting to a medical center within 24 h of injury, and obtained informed consent adhering to local and national ethical and legal requirements. Patients were excluded if there was a severe pre-existing neurological disorder that could potentially bias outcome assessments (in this study self-reported fatigue). Three strata were used to prospectively differentiate patients by clinical care pathway: emergency room (ER; patients evaluated in the ER and discharged afterwards), admission (ADM; patients admitted to a hospital ward) and intensive care unit (ICU; patients who were primarily admitted to the ICU). The main descriptive findings of CENTER-TBI have been published elsewhere [34].
Study participants
In total, 4509 participants were enrolled in the CENTER-TBI core study. In the current study, all patients from the ER, ADM and ICU strata who answered the RPQ-fatigue question at least once at either baseline (mean 2.5 days following admission to CENTER-TBI), 3 or 6 months after injury were selected. Thus, 3354 patients (78% of all included in the core study) were included in this study and their baseline characteristics are described in Table 1. Among these, 2286 had answered the RPQ-fatigue question at baseline, 2164 at 3 months after injury, and 2253 at 6 months after injury and were thus further analyzed in this study.
Measurements
Both adults (age group ≥ 16 years) and children and/or their parents (age group < 16 years) were asked to rate the severity of fatigue compared to their preinjury status during the last 24 h. Rating on a 5-point Likert scale was used, from 0 = “not a problem” to 4 = “severe problem”. A study assessing validity showed that RPQ was unbiased for an age range of 6–96 years [19], and parents ratings of fatigue in children with TBI have been applied in research previously [10].
The data were either collected in face-to-face interviews, or per postal or electronic questionnaires at baseline, (mean 2.5 days following study admission, SD ± 12.0), at 3 and at 6 months follow-ups. The cut-off value ≥ 2, corresponding to symptoms rated as mild, moderate and severe, was used as one of the options of evaluation of symptom severity [38]. However, in clinical practice, a sub-group of patients with moderate and/or severe fatigue symptoms may be challenging to treat because of its impact on general functioning and daily activities; thus, a cut-off value ≥ 3, corresponding to symptoms rated as moderate and severe was also applied.
Socio-demographic and injury-related characteristics that were collected at the time of study admission and used as independent variables included gender (female/male), age (continuous, and categorical: 0–18, 19–40, 41–64, > 65 years, and dichotomized at median value) and education (continuous, i.e. in years, and dichotomized at median value).
Preinjury somatic comorbidities were measured by the pre-injury American Society of Anesthesiologists Physical Status Classification System score (ASA-PS) [23].
Preinjury psychiatric conditions comprised anxiety, depression, sleep disorders, schizophrenia, drug abuse or other psychiatric problems as reported by patients retrospectively at follow-up.
Injury-related variables were: injury mechanism (road traffic accident, falls, others); injury severity measured by patient strata, Glasgow Coma Scale (GCS) score/category within the first 24 h after injury [36], presence of intracranial injuries on first CT head, Abbreviated Injury Scale head (AIS head, score ≥ 3 considered as severe intracranial injury) [15], and Injury Severity Score (ISS), where a score > 15 was considered as major overall trauma [2].
Two additional items from RPQ were used to assess sleep disturbances and feeling depressed at baseline, and were applied as determinants of postinjury comorbidities of potential relevance for feeling fatigued. A cut-off score of ≥ 2 (mild, moderate and severe problems) was used.
Statistical analysis
The CENTER-TBI dataset version 2.0 (dataset from May 2019) was analyzed in this manuscript. The frequency of patients experiencing fatigue was assessed per patient strata, age group, gender and GCS severity level.
For descriptive statistics means with standard deviations (SD), medians with interquartile range (IQR), or percentages are presented. Differences in demographic and injury related data between patients’ strata ER, ADM and ICU were tested using a one-way ANOVA or Kruskal–Wallis test for continuous variables. A chi-square test for contingency tables was performed to detect group differences in categorical variables.
To analyze changes in fatigue between the patients’ strata over the entire follow-up period and account for repeated measures by patient, mixed effect logistic regression was performed using fatigue (dichotomized at the value ≥ 2) as the outcome variable. Time and time-by-patient strata interaction were introduced as fixed effects in all models. Based on the mixed effects logistic regression, we estimated risk differences with 95% confidence intervals (CI) from baseline to 6 months using the delta method. For comparison of the effects of different cut-offs, the analysis was replicated using fatigue dichotomized at the value ≥ 3 as the outcome variable.
Further, mixed effect logistic regression analyses were performed to investigate whether changes of fatigue (dichotomized at the value ≥ 2/ ≥ 3) during the follow-up period (baseline, 3, and 6 months) could be predicted by age, gender, patient strata, education, preinjury ASA-PS and psychiatric comorbidities, GCS score, intracranial injury on CT, AIS head, ISS, and RPQ items `feeling depressed`, and `sleep disturbance` (dichotomized at the value of ≥ 2). Time and all predictor variables were treated as fixed effects in the models. Interaction effects between time and fixed factors were verified by introducing product terms. All models included a random intercept. Statistically significant fixed main effects or interaction effects on fatigue ≥ 2 were graphed across each of the three time points. In these figures, if the predictor was continuous a median-split procedure was used to generate separate lines as function of the predictor.
Missing predictor data were handled by multiple imputations with ten imputations applying the Markov Chain Monte Carlo method [32]. Sensitivity analyses were performed to handle missing values in predictor variables. The multiple imputed model was compared with the complete case analyses, and presented in results.
All statistical analyses were performed using IBM SPSS Statistics for Windows version 25 (Armonk, NY: IBM Corp.) and Stata 15 (Stata Corp LLC, College Station, TX).
Results
Table 1 shows demographic and injury characteristics by patient strata; 808 patients were included in the ER stratum, 1351 in ADM, and 1195 in ICU. Median age of the total sample was 49 (IQR 29, 65) years and 65% of the participants were male. Median years of education was 13 (IQR 11, 16) years. Socio-demographics and injury severity characteristics differed significantly between patient strata (Table 1). Severe TBI (GCS 3–8), severe intracranial injury (AIS head ≥ 3) and severe overall trauma (ISS > 15) were observed in 37, 92 and 95% of patients in ICU stratum, respectively.
Furthermore, 2286 patients reported on the fatigue item at baseline and were thus evaluated in this study. Of these, 46.9% reported having fatigue (cut-off score ≥ 2). The frequency was halved when using moderate/severe fatigue cut-off score (≥ 3) (22.8%). The median fatigue score was highest in the patients admitted to ICU (2, IQR 0–3, p = 0.001) where 57.6% reported moderate/severe fatigue. In ADM and ER strata, 48.2 and 39.0% participants experienced moderate/severe fatigue, respectively (Table 2).
eTable 1 in the Supplement presents fatigue scores by age groups and patients’ strata. In the ER stratum, the highest prevalence of moderate/severe fatigue was in the age group 19–40 (22.4%); in the ADM stratum in the age group 0–18 (34.9%). The most frequently reported moderate/severe fatigue was in the ICU stratum in age group 0–18 (48.8%), and age groups 19–40 and 41–65 years (32.4 and 31.4%, respectively).
The frequency of fatigue by 10-year age groups and gender is presented in Fig. 1. Overall, 52.5% of females and 43.6% of males reported fatigue; the frequency was highest in females across all age groups. The highest frequency of moderate/severe fatigue (≥ 3) was found for females aged 50–60 years (38.3%) and males aged 0–10 years (46.4%), and the lowest in females aged 60–70 years (20.3%) and males > 70 years (8.5%).
Changes of fatigue across 6 months follow-up
The estimated proportions of fatigue score ≥ 2 and ≥ 3 by patients strata are reported in Fig. 2a, b.
Overall, there were no statistically significant differences in fatigue proportions between patient strata`s across the first 6 months post injury. However, significant within group differences due to a decrease in fatigue scores ≥ 2 were found in the ER (mean change − 7.2, 95%CI − 12.0 to − 2.4, p = 0.003) and ADM (mean change: − 7.7, 95% CI − 11.5 to − 3.8, p < 0.001) strata from baseline to 6 months, but not for the ICU group (mean change − 2.0, 95%CI − 7.2 to 3.2, p = 0.454). When applying cut-off ≥ 3, representing moderate and severe fatigue, no such reduction was observed, indicating more persistence of severe symptoms compared to mild.
Similar results were found in the modeling of changes of fatigue scores ≥ 2 and the score ≥ 3 by GCS severity categories supporting the notion that the clinical pathways in the acute TBI phase are indicators of injury severity (eFigures 1 a and 1b and eTable 2 in the Supplement).
Predictors of fatigue changes
Two models used in the predictive analyses examined whether changes of fatigue scores ≥ 2 (model 1) and ≥ 3 (model 2) over time could be predicted by demographic variables, injury severity indicators and comorbidities. All statistically significant and non-significant fixed effects from the full model and their coefficients, p-values, and 95% confidence intervals are presented in Table 3.
In model 1, the ICU patient stratum, age, gender, education, preinjury ASA-PS, AIS head, ISS, feeling depressed, and sleep disturbance yielded significant effects on fatigue probability changes. Patients admitted to ICU had a higher probability of experienced fatigue than those admitted to ER and ADM strata. In addition, patients with lower age, higher education, more severe injuries as assessed by AIS head and ISS, with pre-injury somatic and psychiatric diseases and postinjury comorbidity (sleep disturbance and feelings of depression) and females had a higher probability of fatigue.
The significant interaction effect between time and age suggested that the patient group < 49 years tended to report higher fatigue scores initially and then decreased over time, e.g. reported less fatigue, whereas patients ≥ 49 years reported less fatigue symptoms initially and then fatigue slightly increased over time (Fig. 3).
The significant interaction effect between time and education suggested that patients with higher education (≥ 13 years) tended to report higher fatigue scores initially and then decreased over time, whereas those with lower education reported less fatigue initially, and then slightly higher fatigue scores during the first 3 months (Fig. 4).
The significant interaction effect between time and preinjury psychiatric conditions suggested that patients with known psychiatric problems tended to report higher fatigue scores at baseline and then slightly increased scores over time, whereas those without psychiatric conditions reported decreased scores over time (Fig. 5).
The significant interaction effects between time and feeling depressed and sleep disturbance suggested that patients who reported feeling depressed and sleep disturbance (cut-off ≥ 2) tended to report higher fatigue scores initially, then less over the next 3 months and stable levels during the last 3 months. (eFigures 2 and 3 in the Supplement).
In model 2, the same predictors were statistically significant as in model 1 (except the ICU stratum) indicating that the assessed fatigue predictors are of major importance across all fatigue severity levels.
Discussion
This large-scale, observational longitudinal study assessed the frequency of fatigue following TBI, fatigue changes across clinical care pathways, severity of injury, and predictors of fatigue severity levels.
Fatigue is a widespread symptom in the acute and post-acute TBI phase [39]. As expected, we found a high frequency of fatigue throughout the whole sample included in this study: around 47% of patients reported subjective fatigue of any severity (cut-off ≥ 2) at baseline, 48% at 3 months and 46% at 6 months. These frequencies were halved when cut-off ≥ 3 (moderate and severe fatigue) was used. Females and patients of younger age (≤ 40 years) reported higher frequency of fatigue at baseline. The frequency of fatigue was highest in the patients admitted to the ICU, those with moderate and severe TBI, and more severe intracranial injuries and overall trauma. Our results suggest that more severe TBI may increase the risk of fatigue probably due to the neuro-morphological brain damage as discussed later. However, this is in contrast with previous research that reports no increased risk of fatigue in those with more severe TBI [24].
In line with our expectations, level of fatigue stayed quite stable over the first 6 months post-TBI, particularly, the moderate and severe levels (fatigue cut-off ≥ 3). As fatigue has an unfavorable effect on participation in activities of daily life [4], the results indicate that we should identify those with higher levels of fatigue early after the injury, and provide further assessments, timely advices, and targeted rehabilitation programs.
Demographic factors such as age, gender, and education were associated with fatigue levels in this study. As mentioned previously, findings regarding the association between fatigue following TBI and demographic factors are inconsistent in the literature. For example, Cantor et al. [7] did not find any association between age, gender, education and fatigue. In our study, lower age was associated with higher levels of fatigue, probably reflecting the TBI severity in this population (33% of patients in age group ≤ 40 years had severe TBI, in contrast to 20% of patients in age group > 40 years).
We found that females reported greater levels of fatigue compared to males, in line with previous studies [12]. In studies on self-reported symptoms following TBI, women are more likely to report problems across different symptom domains [14]. Furthermore, post-concussion symptoms and especially fatigue is prevalent in the general population as well [37]. However, previous research has suggested that gender differences in socialization and gender-role expectations may change over time and moderate the relationship between gender and outcome measures after TBI [9, 25].
We also found an association between higher levels of education and greater severity of fatigue, which is in line with study by Ziino & Ponsford [41]. This may relate to a trend in the general population where people with higher education report more symptoms, possibly related to them having a better understanding of health problems and health care services utilization [11]. Another possible explanation may be related to the concept of cognitive reserve, i.e. the fact that education seems to contribute to higher levels of cognitive functioning throughout the life-span, which again may result in individuals with higher education coping better with TBI-related cognitive impairments. However, as people with higher levels of education often work in cognitively demanding professions, the subjective experience of fatigue may hamper the use of cognitive reserves, causing fatigue to feel relatively more detrimental to these persons. Given the mixed results in the current literature regarding the association between education and fatigue levels, future studies on the relationship between education, cognitive reserve and fatigue after TBI are needed.
Furthermore, the present results support a relationship between fatigue and more severe TBI and overall trauma. This was indicated by several significant predictors including the ICU stratum, AIS head ≥ 3 and higher ISS score, all affecting the fatigue levels in this study. Some studies have indicated that post-TBI fatigue was positively associated with greater severity of injury [33] whereas others have failed to demonstrate an association between fatigue and injury severity [24, 28, 41]. Methodological differences between studies may explain these discrepancies. Still, it is worth mentioning that previous studies have suggested that intracranial injuries such as traumatic axonal injury (TAI), global and regional thalamic morphometric changes and functional connectivity in the thalamus and middle frontal cortex may contribute to fatigue following TBI [8, 13, 26]. However, there are only few studies on this topic, and further research on the association between neuro-morphological brain injury and fatigue following TBI is needed.
Presence of preinjury (i.e. somatic disease and psychiatric conditions) and postinjury comorbidities (i.e., feeling depressed and sleep disturbance) also predicted fatigue levels. Participants with preinjury psychiatric conditions, those with depressive feelings and sleep problems were at risk of unfavorable fatigue outcomes in this study. Previous TBI studies with mixed severity samples [6, 12] have demonstrated the association between these comorbidities and fatigue. This is of importance to the field of rehabilitation given the impact these symptoms may have on daily activity levels and health-related quality of life. Treating the symptoms that co-occur with and interact with fatigue such as premorbid psychiatric problems, ongoing depression, sleep problems, and pain and finding a balance between rest and activities (i.e., pacing) is currently the best recommendations for fatigue treatment [30].
Overall, the same factors predicted fatigue regardless of the cut-off (≥ 2 or ≥ 3) applied, indicating the reliability of predictors used in the study. Time since injury interacts with a range of predictors, but does not predict changes on its own, whereas injury severity appears to be a robust predictor. The study findings may help health professionals to plan individualized therapy and rehabilitation programs in the early stages of recovery for patients with specific demographic and injury characteristics and comorbidities.
Limitations
These findings may not be generalizable to all European individuals who have sustained a TBI since participants were mainly recruited from trauma referral centers. As such, the findings are not necessarily generalizable to individuals sustaining a minimal TBI or a mild TBI without indication for a CT head. One of the major limitations of this study is the use of a single item operationalization of fatigue; nevertheless, it was the only opportunity to measure fatigue and its changes when using the CENTER-TBI data. The wording of the item asks whether fatigue has been a problem for the past 24 h compared to before the injury. The experience of symptoms, however, can vary, and may be related to the level of activity at the time of assessment. This raises the possibility that the reported ratings of fatigue symptoms are not reflective of the overall experience (i.e., both over- and underreporting possible). Using fatigue assessment instruments with established validity in specific patient groups is recommended [40]; yet, such instruments were not available in this study. Further, usage of specific fatigue tools may not be as achievable in a hectic clinical setting as the broad current use of the RPQ, thus our results may be more easily transferrable to common clinical practice.
Fatigue after TBI has increasingly been conceptualized as a complex condition, with a number of factors that may contribute to its development and persistence [30]. Variables included in our predictive models were selected based on clinical importance and previous studies on TBI. Additionally, other variables such as preinjury fatigue symptoms, neurocognitive function, structural brain abnormalities, potential blood biomarkers, and hormonal imbalance not included in this study should be assessed in future studies. Taken together, translational research is needed to advance a clinical decision-making process and targeted medical treatment of fatigue in the future.
Change history
10 September 2020
The original version of this article unfortunately contained a mistake.
References
Aaronson LS, Teel CS, Cassmeyer V, Neuberger GB, Pallikkathayil L, Pierce J, Press AN, Williams PD, Wingate A (1999) Defining and measuring fatigue. Image J Nurs Sch 31:45–50
Baker SP, O'Neill B, Haddon W Jr, Long WB (1974) The injury severity score: a method for describing patients with multiple injuries and evaluating emergency care. J Trauma 14:187–196
Barker-Collo S, Theadom A, Starkey N, Kahan M, Jones K, Feigin V (2018) Factor structure of the Rivermead post-concussion symptoms questionnaire over the first year following mild traumatic brain injury. Brain Inj 32:453–458
Beaulieu-Bonneau S, Ouellet MC (2017) Fatigue in the first year after traumatic brain injury: course, relationship with injury severity, and correlates. Neuropsychol Rehabil 27:983–1001
Borgaro SR, Baker J, Wethe JV, Prigatano GP, Kwasnica C (2005) Subjective reports of fatigue during early recovery from traumatic brain injury. J Head Trauma Rehabil 20:416–425
Cantor JB, Ashman T, Gordon W, Ginsberg A, Engmann C, Egan M, Spielman L, Dijkers M, Flanagan S (2008) Fatigue after traumatic brain injury and its impact on participation and quality of life. J Head Trauma Rehabil 23:41–51
Cantor JB, Bushnik T, Cicerone K, Dijkers MP, Gordon W, Hammond FM, Kolakowsky-Hayner SA, Lequerica A, Nguyen M, Spielman LA (2012) Insomnia, fatigue, and sleepiness in the first 2 years after traumatic brain injury: an NIDRR TBI model system module study. J Head Trauma Rehabil 27:E1–14
Clark AL, Sorg SF, Holiday K, Bigler ED, Bangen KJ, Evangelista ND, Bondi MW, Schiehser DM, Delano-Wood L (2018) Fatigue is associated with global and regional thalamic morphometry in veterans with a history of mild traumatic brain injury. J Head Trauma Rehabil 33:382–392
Colantonio A, Harris JE, Ratcliff G, Chase S, Ellis K (2010) Gender differences in self reported long term outcomes following moderate to severe traumatic brain injury. BMC Neurol 10:102
Crichton A, Anderson V, Oakley E, Greenham M, Hearps S, Delzoppo C, Beauchamp MH, Hutchison JS, Guerguerian AM, Boutis K, Babl FE (2018) Fatigue following traumatic brain injury in children and adolescents: a longitudinal follow-up 6 to 12 months after injury. J Head Trauma Rehabil 33:200–209
Cutler DM, Lleras-Muney A (2010) Understanding differences in health behaviors by education. J Health Econ 29:1–28
Englander J, Bushnik T, Oggins J, Katznelson L (2010) Fatigue after traumatic brain injury: association with neuroendocrine, sleep, depression and other factors. Brain Inj 24:1379–1388
Esbjörnsson E, Skoglund T, Sunnerhagen KS (2013) Fatigue, psychosocial adaptation and quality of life one year after traumatic brain injury and suspected traumatic axonal injury; evaluations of patients and relatives. A pilot study. J Rehabil Med in press
Farace E, Alves WM (2000) Do women fare worse: a metaanalysis of gender differences in traumatic brain injury outcome. J Neurosurg 93:539–545
Gennarelli TA, Wodzin E (2006) AIS 2005: a contemporary injury scale. Injury 37:1083–1091
Juengst SB, Nabasny A, Terhorst L (2019) Neurobehavioral symptoms in community-dwelling adults with and without chronic traumatic brain injury: differences by age, gender, education, and health condition. Front Neurol 10:1210
King NS, Crawford S, Wenden FJ, Moss NE, Wade DT (1995) The Rivermead post concussion symptoms questionnaire: a measure of symptoms commonly experienced after head injury and its reliability. J Neurol 242(9):587–592
Krupp LB, LaRocca NG, Muir-Nash J, Steinberg AD (1989) The fatigue severity scale. Application to patients with multiple sclerosis and systemic lupus erythematosus. Arch Neurol 46(10):1121–1123
Lannsjo M, Borg J, Bjorklund G, Af Geijerstam JL, Lundgren-Nilsson A (2011) Internal construct validity of the Rivermead post-concussion symptoms questionnaire. J Rehabil Med 43:997–1002
Lerdal A, Bakken LN, Rasmussen EF, Beiermann C, Ryen S, Pynten S, Drefvelin AS, Dahl AM, Rognstad G, Finset A, Lee KA, Kim HS (2011) Physical impairment, depressive symptoms and pre-stroke fatigue are related to fatigue in the acute phase after stroke. Disabil Rehabil 33:334–342
Lerdal A, Wahl A, Rustoen T, Hanestad BR, Moum T (2005) Fatigue in the general population: a translation and test of the psychometric properties of the Norwegian version of the fatigue severity scale. Scand J Public Health 33:123–130
Maas AI, Menon DK, Steyerberg EW, Citerio G, Lecky F, Manley GT, Hill S, Legrand V, Sorgner A (2015) Collaborative European NeuroTrauma Effectiveness Research in Traumatic Brain Injury (CENTER-TBI): a prospective longitudinal observational study. Neurosurgery 76:67–80
Mayhew D, Mendonca V, Murthy BVS (2019) A review of ASA physical status—historical perspectives and modern developments. Anaesthesia 74:373–379
Mollayeva T, Kendzerska T, Mollayeva S, Shapiro CM, Colantonio A, Cassidy JD (2014) A systematic review of fatigue in patients with traumatic brain injury: the course, predictors and consequences. Neurosci Biobehav Rev 47:684–716
Niemeier JP, Perrin PB, Holcomb MG, Rolston CD, Artman LK, Lu J, Nersessova KS (2014) Gender differences in awareness and outcomes during acute traumatic brain injury recovery. J Womens Health (Larchmt ) 23:573–580
Nordin LE, Moller MC, Julin P, Bartfai A, Hashim F, Li TQ (2016) Post mTBI fatigue is associated with abnormal brain functional connectivity. Sci Rep 6:21183
Norrie J, Heitger M, Leathem J, Anderson T, Jones R, Flett R (2010) Mild traumatic brain injury and fatigue: a prospective longitudinal study. Brain Inj 24:1528–1538
Ouellet MC, Morin CM (2006) Fatigue following traumatic brain injury: Frequency, characteristics, and associated factors. Rehabil Psychol 51:140–148
Ponsford J, Schonberger M, Rajaratnam SM (2015) A model of fatigue following traumatic brain injury. J Head Trauma Rehabil 30:277–282
Ponsford JL, Ziino C, Parcell DL, Shekleton JA, Roper M, Redman JR, Phipps-Nelson J, Rajaratnam SM (2012) Fatigue and sleep disturbance following traumatic brain injury–their nature, causes, and potential treatments. J Head Trauma Rehabil 27:224–233
Potter S, Leigh E, Wade D, Fleminger S (2006) The Rivermead post concussion symptoms questionnaire: a confirmatory factor analysis. J Neurol 253:1603–1614
Royston P (2004) Multiple imputation of missing values. Stata J227–241
Schiehser DM, Delano-Wood L, Jak AJ, Hanson KL, Sorg SF, Orff H, Clark AL (2017) Predictors of cognitive and physical fatigue in post-acute mild-moderate traumatic brain injury. Neuropsychol Rehabil 27:1031–1046
Steyerberg EW, Wiegers E, Sewalt C, Buki A, Citerio G, De Keyser V, Ercole A, Kunzmann K, Lanyon L, Lecky F, Lingsma H, Manley G, Nelson D, Peul W, Stocchetti N, Von SN, Vande VT, Verheyden J, Wilson L, Maas AIR, Menon DK (2019) Case-mix, care pathways, and outcomes in patients with traumatic brain injury in CENTER-TBI: a European prospective, multicentre, longitudinal, cohort study. Lancet Neurol 18:923–934
Stulemeijer M, van der Werf S, Bleijenberg G, Biert J, Brauer J, Vos PE (2006) Recovery from mild traumatic brain injury: a focus on fatigue. J Neurol 253(8):1041–1047
Teasdale G, Jennett B (1974) Assessment of coma and impaired consciousness. A practical scale. Lancet 2:81–84
Voormolen DC, Cnossen MC, Polinder S, Gravesteijn BY, von Steinbuechel N, Real RGL, Haagsma JA (2019) Prevalence of post-concussion-like symptoms in the general population in Italy, The Netherlands and the United Kingdom. Brain Inj 33:1078–1086
Voormolen DC, Cnossen MC, Polinder S, von Steinbuechel N, Vos PE, Haagsma JA (2018) Divergent classification methods of post-concussion syndrome after mild traumatic brain injury: prevalence rates, risk factors, and functional outcome. J Neurotrauma 35:1233–1241
Voormolen DC, Haagsma JA, Polinder S, Maas AIR, Steyerberg EW, Vulekovic P, Sewalt CA, Gravesteijn BY, Covic A, Andelic N, Plass AM, von Steinbuechel N (2019) Post-concussion symptoms in complicated vs uncomplicated mild traumatic brain injury patients at three and six months post-injury: results from the CENTER-TBI Study. J Clin Med 8:1921
Whitehead L (2009) The measurement of fatigue in chronic illness: a systematic review of unidimensional and multidimensional fatigue measures. J Pain Symptom Manage 37:107–128
Ziino C, Ponsford J (2005) Measurement and prediction of subjective fatigue following traumatic brain injury. J Int Neuropsychol Soc 11:416–425
Acknowledgement
Open Access funding provided by University of Oslo (incl Oslo University Hospital). CENTER-TBI participants and investigators Cecilia Åkerlund1, Krisztina Amrein2, Nada Andelic3, Lasse Andreassen4, Audny Anke5, Anna Antoni6, Gérard Audibert7, Philippe Azouvi8, Maria Luisa Azzolini9, Ronald Bartels10, Pál Barzó11, Romuald Beauvais12, Ronny Beer13, Bo-Michael Bellander14, Antonio Belli15, Habib Benali16, Maurizio Berardino17, Luigi Beretta9, Morten Blaabjerg18, Peter Bragge19, Alexandra Brazinova20, Vibeke Brinck21, Joanne Brooker22, Camilla Brorsson23, Andras Buki24, Monika Bullinger25, Manuel Cabeleira26, Alessio Caccioppola27, Emiliana Calappi 27, Maria Rosa Calvi9, Peter Cameron28, Guillermo Carbayo Lozano29, Marco Carbonara27, Simona Cavallo17, Giorgio Chevallard30, Arturo Chieregato30, Giuseppe Citerio31, 32, Iris Ceyisakar33, Hans Clusmann34, Mark Coburn35, Jonathan Coles36, Jamie D. Cooper37, Marta Correia38, Amra Čović 39, Nicola Curry40, Endre Czeiter24, Marek Czosnyka26, Claire Dahyot-Fizelier41, Paul Dark42, Helen Dawes43, Véronique De Keyser44, Vincent Degos16, Francesco Della Corte45, Hugo den Boogert10, Bart Depreitere46, Đula Đilvesi 47, Abhishek Dixit48, Emma Donoghue22, Jens Dreier49, Guy-Loup Dulière50, Ari Ercole48, Patrick Esser43, Erzsébet Ezer51, Martin Fabricius52, Valery L. Feigin53, Kelly Foks54, Shirin Frisvold55, Alex Furmanov56, Pablo Gagliardo57, Damien Galanaud16, Dashiell Gantner28, Guoyi Gao58, Pradeep George59, Alexandre Ghuysen60, Lelde Giga61, Ben Glocker62, Jagoš Golubovic47, Pedro A. Gomez 63, Johannes Gratz64, Benjamin Gravesteijn33, Francesca Grossi45, Russell L. Gruen65, Deepak Gupta66, Juanita A. Haagsma33, Iain Haitsma67, Raimund Helbok13, Eirik Helseth68, Lindsay Horton 69, Jilske Huijben33, Peter J. Hutchinson70, Bram Jacobs71, Stefan Jankowski72, Mike Jarrett21, Ji-yao Jiang58, Faye Johnson73, Kelly Jones53, Mladen Karan47, Angelos G. Kolias70, Erwin Kompanje74, Daniel Kondziella52, Evgenios Koraropoulos48, Lars-Owe Koskinen75, Noémi Kovács76, Ana Kowark35, Alfonso Lagares63, Linda Lanyon59, Steven Laureys77, Fiona Lecky78, 79, Didier Ledoux77, Rolf Lefering80, Valerie Legrand81, Aurelie Lejeune82, Leon Levi83, Roger Lightfoot84, Hester Lingsma33, Andrew I.R. Maas44, Ana M. Castaño-León63, Marc Maegele85, Marek Majdan20, Alex Manara86, Geoffrey Manley87, Costanza Martino88, Hugues Maréchal50, Julia Mattern89, Catherine McMahon90, Béla Melegh91, David Menon48, Tomas Menovsky44, Ana Mikolic33, Benoit Misset77, Visakh Muraleedharan59, Lynnette Murray28, Ancuta Negru92, David Nelson1, Virginia Newcombe48, Daan Nieboer33, József Nyirádi2, Otesile Olubukola78, Matej Oresic93, Fabrizio Ortolano27, Aarno Palotie94, 95, 96, Paul M. Parizel97, Jean-François Payen98, Natascha Perera12, Vincent Perlbarg16, Paolo Persona99, Wilco Peul100, Anna Piippo-Karjalainen101, Matti Pirinen94, Horia Ples92, Suzanne Polinder33, Inigo Pomposo29, Jussi P. Posti 102, Louis Puybasset103, Andreea Radoi 104, Arminas Ragauskas105, Rahul Raj101, Malinka Rambadagalla106, Jonathan Rhodes107, Sylvia Richardson108, Sophie Richter48, Samuli Ripatti94, Saulius Rocka105, Cecilie Roe109, Olav Roise110,111, Jonathan Rosand112, Jeffrey V. Rosenfeld113, Christina Rosenlund114, Guy Rosenthal56, Rolf Rossaint35, Sandra Rossi99, Daniel Rueckert62, Martin Rusnák115, Juan Sahuquillo104, Oliver Sakowitz89, 116, Renan Sanchez-Porras116, Janos Sandor117, Nadine Schäfer80, Silke Schmidt118, Herbert Schoechl119, Guus Schoonman120, Rico Frederik Schou121, Elisabeth Schwendenwein6, Charlie Sewalt33, Toril Skandsen122,123, Peter Smielewski26, Abayomi Sorinola124, Emmanuel Stamatakis48, Simon Stanworth40, Robert Stevens125, William Stewart126, Ewout W. Steyerberg33, 127, Nino Stocchetti128, Nina Sundström129, Anneliese Synnot22, 130, Riikka Takala131, Viktória Tamás124, Tomas Tamosuitis132, Mark Steven Taylor20, Braden Te Ao53, Olli Tenovuo102, Alice Theadom53, Matt Thomas86, Dick Tibboel133, Marjolein Timmers74, Christos Tolias134, Tony Trapani28, Cristina Maria Tudora92, Peter Vajkoczy 135, Shirley Vallance28, Egils Valeinis61, Zoltán Vámos51, Mathieu van der Jagt136, Gregory Van der Steen44, Joukje van der Naalt71, Jeroen T.J.M. van Dijck 100, Thomas A. van Essen100, Wim Van Hecke137, Caroline van Heugten138, Dominique Van Praag139, Thijs Vande Vyvere137, Roel P. J. van Wijk100, Alessia Vargiolu32, Emmanuel Vega82, Kimberley Velt33, Jan Verheyden137, Paul M. Vespa140, Anne Vik122, 141, Rimantas Vilcinis132, Victor Volovici67, Nicole von Steinbüchel39, Daphne Voormolen33, Petar Vulekovic47, Kevin K.W. Wang142, Eveline Wiegers33, Guy Williams48, Lindsay Wilson69, Stefan Winzeck48, Stefan Wolf143, Zhihui Yang142, Peter Ylén144, Alexander Younsi89, Frederick A. Zeiler48,145, Veronika Zelinkova20, Agate Ziverte61, Tommaso Zoerle27
1Department of Physiology and Pharmacology, Section of Perioperative Medicine and Intensive Care, Karolinska Institutet, Stockholm, Sweden; 2János Szentágothai Research Centre, University of Pécs, Pécs, Hungary; 3Division of Clinical Neuroscience, Department of Physical Medicine and Rehabilitation, Oslo University Hospital and University of Oslo, Oslo, Norway; 4Department of Neurosurgery, University Hospital Northern Norway, Tromso, Norway; 5Department of Physical Medicine and Rehabilitation, University Hospital Northern Norway, Tromso, Norway; 6Trauma Surgery, Medical University Vienna, Vienna, Austria; 7Department of Anesthesiology & Intensive Care, University Hospital Nancy, Nancy, France; 8Raymond Poincare hospital, Assistance Publique – Hopitaux de Paris, Paris, France; 9Department of Anesthesiology & Intensive Care, S Raffaele University Hospital, Milan, Italy; 10Department of Neurosurgery, Radboud University Medical Center, Nijmegen, The Netherlands; 11Department of Neurosurgery, University of Szeged, Szeged, Hungary; 12International Projects Management, ARTTIC, Munchen, Germany; 13Department of Neurology, Neurological Intensive Care Unit, Medical University of Innsbruck, Innsbruck, Austria; 14Department of Neurosurgery & Anesthesia & intensive care medicine, Karolinska University Hospital, Stockholm, Sweden; 15NIHR Surgical Reconstruction and Microbiology Research Centre, Birmingham, UK; 16Anesthesie-Réanimation, Assistance Publique – Hopitaux de Paris, Paris, France; 17Department of Anesthesia & ICU, AOU Città della Salute e della Scienza di Torino—Orthopedic and Trauma Center, Torino, Italy; 18Department of Neurology, Odense University Hospital, Odense, Denmark; 19BehaviourWorks Australia, Monash Sustainability Institute, Monash University, Victoria, Australia; 20Department of Public Health, Faculty of Health Sciences and Social Work, Trnava University, Trnava, Slovakia; 21 Quesgen Systems Inc., Burlingame, California, USA; 22Australian & New Zealand Intensive Care Research Centre, Department of Epidemiology and Preventive Medicine, School of Public Health and Preventive Medicine, Monash University, Melbourne, Australia; 23Department of Surgery and Perioperative Science, Umeå University, Umeå, Sweden; 24Department of Neurosurgery, Medical School, University of Pécs, Hungary and Neurotrauma Research Group, János Szentágothai Research Centre, University of Pécs, Hungary; 25Department of Medical Psychology, Universitätsklinikum Hamburg-Eppendorf, Hamburg, Germany; 26Brain Physics Lab, Division of Neurosurgery, Dept of Clinical Neurosciences, University of Cambridge, Addenbrooke’s Hospital, Cambridge, UK; 27Neuro ICU, Fondazione IRCCS Cà Granda Ospedale Maggiore Policlinico, Milan, Italy; 28ANZIC Research Centre, Monash University, Department of Epidemiology and Preventive Medicine, Melbourne, Victoria, Australia; 29Department of Neurosurgery, Hospital of Cruces, Bilbao, Spain; 30NeuroIntensive Care, Niguarda Hospital, Milan, Italy; 31School of Medicine and Surgery, Università Milano Bicocca, Milano, Italy; 32NeuroIntensive Care, ASST di Monza, Monza, Italy; 33Department of Public Health, Erasmus Medical Center-University Medical Center, Rotterdam, The Netherlands; 34Department of Neurosurgery, Medical Faculty RWTH Aachen University, Aachen, Germany; 35Department of Anaesthesiology, University Hospital of Aachen, Aachen, Germany; 36Department of Anesthesia & Neurointensive Care, Cambridge University Hospital NHS Foundation Trust, Cambridge, UK; 37School of Public Health & PM, Monash University and The Alfred Hospital, Melbourne, Victoria, Australia; 38Radiology/MRI department, MRC Cognition and Brain Sciences Unit, Cambridge, UK; 39Institute of Medical Psychology and Medical Sociology, Universitätsmedizin Göttingen, Göttingen, Germany; 40Oxford University Hospitals NHS Trust, Oxford, UK; 41Intensive Care Unit, CHU Poitiers, Potiers, France; 42University of Manchester NIHR Biomedical Research Centre, Critical Care Directorate, Salford Royal Hospital NHS Foundation Trust, Salford, UK; 43Movement Science Group, Faculty of Health and Life Sciences, Oxford Brookes University, Oxford, UK; 44Department of Neurosurgery, Antwerp University Hospital and University of Antwerp, Edegem, Belgium; 45Department of Anesthesia & Intensive Care, Maggiore Della Carità Hospital, Novara, Italy; 46Department of Neurosurgery, University Hospitals Leuven, Leuven, Belgium; 47Department of Neurosurgery, Clinical centre of Vojvodina, Faculty of Medicine, University of Novi Sad, Novi Sad, Serbia; 48Division of Anaesthesia, University of Cambridge, Addenbrooke’s Hospital, Cambridge, UK; 49Center for Stroke Research Berlin, Charité – Universitätsmedizin Berlin, corporate member of Freie Universität Berlin, Humboldt-Universität zu Berlin, and Berlin Institute of Health, Berlin, Germany; 50Intensive Care Unit, CHR Citadelle, Liège, Belgium; 51Department of Anaesthesiology and Intensive Therapy, University of Pécs, Pécs, Hungary; 52Departments of Neurology, Clinical Neurophysiology and Neuroanesthesiology, Region Hovedstaden Rigshospitalet, Copenhagen, Denmark; 53National Institute for Stroke and Applied Neurosciences, Faculty of Health and Environmental Studies, Auckland University of Technology, Auckland, New Zealand; 54Department of Neurology, Erasmus MC, Rotterdam, the Netherlands; 55Department of Anesthesiology and Intensive care, University Hospital Northern Norway, Tromso, Norway; 56Department of Neurosurgery, Hadassah-hebrew University Medical center, Jerusalem, Israel; 57Fundación Instituto Valenciano de Neurorrehabilitación (FIVAN), Valencia, Spain; 58Department of Neurosurgery, Shanghai Renji hospital, Shanghai Jiaotong University/school of medicine, Shanghai, China; 59Karolinska Institutet, INCF International Neuroinformatics Coordinating Facility, Stockholm, Sweden; 60Emergency Department, CHU, Liège, Belgium; 61Neurosurgery clinic, Pauls Stradins Clinical University Hospital, Riga, Latvia; 62Department of Computing, Imperial College London, London, UK; 63Department of Neurosurgery, Hospital Universitario 12 de Octubre, Madrid, Spain; 64Department of Anesthesia, Critical Care and Pain Medicine, Medical University of Vienna, Austria; 65College of Health and Medicine, Australian National University, Canberra, Australia; 66Department of Neurosurgery, Neurosciences Centre & JPN Apex trauma centre, All India Institute of Medical Sciences, New Delhi-110029, India; 67Department of Neurosurgery, Erasmus MC, Rotterdam, the Netherlands; 68Department of Neurosurgery, Oslo University Hospital, Oslo, Norway; 69Division of Psychology, University of Stirling, Stirling, UK; 70Division of Neurosurgery, Department of Clinical Neurosciences, Addenbrooke’s Hospital & University of Cambridge, Cambridge, UK; 71Department of Neurology, University of Groningen, University Medical Center Groningen, Groningen, Netherlands; 72Neurointensive Care, Sheffield Teaching Hospitals NHS Foundation Trust, Sheffield, UK; 73Salford Royal Hospital NHS Foundation Trust Acute Research Delivery Team, Salford, UK; 74Department of Intensive Care and Department of Ethics and Philosophy of Medicine, Erasmus Medical Center, Rotterdam, The Netherlands; 75Department of Clinical Neuroscience, Neurosurgery, Umeå University, Umeå, Sweden; 76Hungarian Brain Research Program—Grant No. KTIA_13_NAP-A-II/8, University of Pécs, Pécs, Hungary; 77Cyclotron Research Center, University of Liège, Liège, Belgium; 78Centre for Urgent and Emergency Care Research (CURE), Health Services Research Section, School of Health and Related Research (ScHARR), University of Sheffield, Sheffield, UK; 79Emergency Department, Salford Royal Hospital, Salford UK; 80Institute of Research in Operative Medicine (IFOM), Witten/Herdecke University, Cologne, Germany; 81VP Global Project Management CNS, ICON, Paris, France; 82Department of Anesthesiology-Intensive Care, Lille University Hospital, Lille, France; 83Department of Neurosurgery, Rambam Medical Center, Haifa, Israel; 84Department of Anesthesiology & Intensive Care, University Hospitals Southhampton NHS Trust, Southhampton, UK; 85Cologne-Merheim Medical Center (CMMC), Department of Traumatology, Orthopedic Surgery and Sportmedicine, Witten/Herdecke University, Cologne, Germany; 86Intensive Care Unit, Southmead Hospital, Bristol, Bristol, UK; 87Department of Neurological Surgery, University of California, San Francisco, California, USA; 88Department of Anesthesia & Intensive Care,M. Bufalini Hospital, Cesena, Italy; 89Department of Neurosurgery, University Hospital Heidelberg, Heidelberg, Germany; 90Department of Neurosurgery, The Walton centre NHS Foundation Trust, Liverpool, UK; 91Department of Medical Genetics, University of Pécs, Pécs, Hungary; 92Department of Neurosurgery, Emergency County Hospital Timisoara, Timisoara, Romania; 93School of Medical Sciences, Örebro University, Örebro, Sweden; 94Institute for Molecular Medicine Finland, University of Helsinki, Helsinki, Finland; 95Analytic and Translational Genetics Unit, Department of Medicine; Psychiatric & Neurodevelopmental Genetics Unit, Department of Psychiatry; Department of Neurology, Massachusetts General Hospital, Boston, MA, USA; 96Program in Medical and Population Genetics; The Stanley Center for Psychiatric Research, The Broad Institute of MIT and Harvard, Cambridge, MA, USA; 97Department of Radiology, University of Antwerp, Edegem, Belgium; 98Department of Anesthesiology & Intensive Care, University Hospital of Grenoble, Grenoble, France; 99Department of Anesthesia & Intensive Care, Azienda Ospedaliera Università di Padova, Padova, Italy; 100Dept. of Neurosurgery, Leiden University Medical Center, Leiden, The Netherlands and Dept. of Neurosurgery, Medical Center Haaglanden, The Hague, The Netherlands; 101Department of Neurosurgery, Helsinki University Central Hospital; 102Division of Clinical Neurosciences, Department of Neurosurgery and Turku Brain Injury Centre, Turku University Hospital and University of Turku, Turku, Finland; 103Department of Anesthesiology and Critical Care, Pitié -Salpêtrière Teaching Hospital, Assistance Publique, Hôpitaux de Paris and University Pierre et Marie Curie, Paris, France; 104Neurotraumatology and Neurosurgery Research Unit (UNINN), Vall d'Hebron Research Institute, Barcelona, Spain; 105Department of Neurosurgery, Kaunas University of technology and Vilnius University, Vilnius, Lithuania; 106Department of Neurosurgery, Rezekne Hospital, Latvia; 107Department of Anaesthesia, Critical Care & Pain Medicine NHS Lothian & University of Edinburg, Edinburgh, UK; 108Director, MRC Biostatistics Unit, Cambridge Institute of Public Health, Cambridge, UK; 109Department of Physical Medicine and Rehabilitation, Oslo University Hospital/University of Oslo, Oslo, Norway; 110Division of Orthopedics, Oslo University Hospital, Oslo, Norway; 111Institue of Clinical Medicine, Faculty of Medicine, University of Oslo, Oslo, Norway; 112Broad Institute, Cambridge MA Harvard Medical School, Boston MA, Massachusetts General Hospital, Boston MA, USA; 113National Trauma Research Institute, The Alfred Hospital, Monash University, Melbourne, Victoria, Australia; 114Department of Neurosurgery, Odense University Hospital, Odense, Denmark; 115International Neurotrauma Research Organisation, Vienna, Austria; 116Klinik für Neurochirurgie, Klinikum Ludwigsburg, Ludwigsburg, Germany; 117Division of Biostatistics and Epidemiology, Department of Preventive Medicine, University of Debrecen, Debrecen, Hungary; 118Department Health and Prevention, University Greifswald, Greifswald, Germany; 119Department of Anaesthesiology and Intensive Care, AUVA Trauma Hospital, Salzburg, Austria; 120Department of Neurology, Elisabeth-TweeSteden Ziekenhuis, Tilburg, the Netherlands; 121Department of Neuroanesthesia and Neurointensive Care, Odense University Hospital, Odense, Denmark; 122Department of Neuromedicine and Movement Science, Norwegian University of Science and Technology, NTNU, Trondheim, Norway; 123Department of Physical Medicine and Rehabilitation, St.Olavs Hospital, Trondheim University Hospital, Trondheim, Norway; 124Department of Neurosurgery, University of Pécs, Pécs, Hungary; 125Division of Neuroscience Critical Care, John Hopkins University School of Medicine, Baltimore, USA; 126 Department of Neuropathology, Queen Elizabeth University Hospital and University of Glasgow, Glasgow, UK; 127Dept. of Department of Biomedical Data Sciences, Leiden University Medical Center, Leiden, The Netherlands; 128 Department of Pathophysiology and Transplantation, Milan University, and Neuroscience ICU, Fondazione IRCCS Cà Granda Ospedale Maggiore Policlinico, Milano, Italy; 129Department of Radiation Sciences, Biomedical Engineering, Umeå University, Umeå, Sweden; 130Cochrane Consumers and Communication Review Group, Centre for Health Communication and Participation, School of Psychology and Public Health, La Trobe University, Melbourne, Australia; 131Perioperative Services, Intensive Care Medicine and Pain Management, Turku University Hospital and University of Turku, Turku, Finland; 132Department of Neurosurgery, Kaunas University of Health Sciences, Kaunas, Lithuania; 133Intensive Care and Department of Pediatric Surgery, Erasmus Medical Center, Sophia Children’s Hospital, Rotterdam, The Netherlands; 134Department of Neurosurgery, Kings college London, London, UK; 135Neurologie, Neurochirurgie und Psychiatrie, Charité – Universitätsmedizin Berlin, Berlin, Germany; 136Department of Intensive Care Adults, Erasmus MC– University Medical Center Rotterdam, Rotterdam, the Netherlands; 137icoMetrix NV, Leuven, Belgium; 138Movement Science Group, Faculty of Health and Life Sciences, Oxford Brookes University, Oxford, UK; 139Psychology Department, Antwerp University Hospital, Edegem, Belgium; 140Director of Neurocritical Care, University of California, Los Angeles, USA; 141Department of Neurosurgery, St.Olavs Hospital, Trondheim University Hospital, Trondheim, Norway; 142Department of Emergency Medicine, University of Florida, Gainesville, Florida, USA; 143Department of Neurosurgery, Charité – Universitätsmedizin Berlin, corporate member of Freie Universität Berlin, Humboldt-Universität zu Berlin, and Berlin Institute of Health, Berlin, Germany; 144VTT Technical Research Centre, Tampere, Finland; 145Section of Neurosurgery, Department of Surgery, Rady Faculty of Health Sciences, University of Manitoba, Winnipeg, MB, Canada
Funding
Data used in preparation of this manuscript were obtained in the context of CENTER-TBI, a large collaborative project with the support of the European Union 7th Framework program (EC grant 247 602150). Additional funding was obtained from the Hannelore Kohl Stiftung (Germany), from OneMind (USA) and from Integra LifeSciences Corporation (USA).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical standard
The CENTER-TBI study (EC grant 602150) was conducted in line with relevant local and national ethical guidelines and regulatory requirements for research involving human subjects, as well as with relevant data protection, privacy regulations and informed consent. For a list of recruiting sites, ethical committees, and ethical approval details, see the official Center TBI website (https://www.center-tbi.eu/project/ ethical-approval).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Andelic, N., Røe, C., Brunborg, C. et al. Frequency of fatigue and its changes in the first 6 months after traumatic brain injury: results from the CENTER-TBI study. J Neurol 268, 61–73 (2021). https://doi.org/10.1007/s00415-020-10022-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-020-10022-2