Abstract
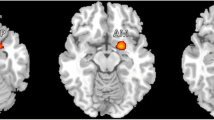
Morphometric MRI studies in adult patients with migraine have consistently demonstrated atrophy of several gray matter (GM) regions involved in pain processing. We explored the regional distribution of GM and white matter (WM) abnormalities in pediatric patients with episodic migraine and their correlations with disease clinical manifestations. Using a 3.0 T scanner, brain T2-weighted and 3D T1-weighted scans were acquired from 12 pediatric migraine patients and 15 age-matched healthy controls. GM and WM volumetric abnormalities were estimated using voxel-based morphometry (p < 0.05, family-wise error corrected). Compared to controls, pediatric migraine patients experienced a significant GM atrophy of several regions of the frontal and temporal lobes which are part of the pain-processing network. They also had an increased volume of the right putamen. The left fusiform gyrus had an increased volume in patients with aura compared to patients without aura and controls, whereas it was significantly atrophied in patients without aura when compared to the other two groups. No abnormalities of WM volume were detected. In migraine patients, regional GM atrophy was not correlated with disease duration and attack frequency, whereas a negative correlation was found between increased volume of the putamen and disease duration (r = −0.95, p < 0.05). These results show that GM morphometric abnormalities do occur in pediatric patients with migraine. The presence of such abnormalities early in the disease course, and the absence of correlation with patient clinical characteristics suggest that they may represent a phenotypic biomarker of this condition.




Similar content being viewed by others
References
Absinta M, Rocca MA, Colombo B, Falini A, Comi G, Filippi M (2012) Selective decreased grey matter volume of the pain-matrix network in cluster headache. Cephalalgia 32:109–115
Ashburner J (2007) A fast diffeomorphic image registration algorithm. Neuroimage 38:95–113
Borsook D, Upadhyay J, Chudler EH, Becerra L (2010) A key role of the basal ganglia in pain and analgesia—insights gained through human functional imaging. Mol Pain 6:27
Burgmer M, Gaubitz M, Konrad C, Wrenger M, Hilgart S, Heuft G, Pfleiderer B (2009) Decreased gray matter volumes in the cingulo-frontal cortex and the amygdala in patients with fibromyalgia. Psychosom Med 71:566–573
Candee MS, McCandless RT, Moore KR, Arrington CB, Minich LL, Bale JF Jr. (2013) White matter lesions in children and adolescents with migraine. Pediatr Neurol 49:393–396
Chudler EH, Dong WK (1995) The role of the basal ganglia in nociception and pain. Pain 60:3–38
Cutrer FM, Huerter K (2007) Migraine aura. Neurologist 13:118–125
Dalgleish T (2004) The emotional brain. Nat Rev Neurosci 5:583–589
DaSilva AF, Granziera C, Snyder J, Hadjikhani N (2007) Thickening in the somatosensory cortex of patients with migraine. Neurology 69:1990–1995
DaSilva AF, Granziera C, Tuch DS, Snyder J, Vincent M, Hadjikhani N (2007) Interictal alterations of the trigeminal somatosensory pathway and periaqueductal gray matter in migraine. NeuroReport 18:301–305
Dinia L, Bonzano L, Albano B, Finocchi C, Del Sette M, Saitta L, Castellan L, Gandolfo C, Roccatagliata L (2013) White matter lesions progression in migraine with aura: a clinical and MRI longitudinal study. J Neuroimaging 23:47–52
Ditchfield JA, McKendrick AM, Badcock DR (2006) Processing of global form and motion in migraineurs. Vision Res 46:141–148
Godinho F, Magnin M, Frot M, Perchet C, Garcia-Larrea L (2006) Emotional modulation of pain: is it the sensation or what we recall? J Neurosci 26:11454–11461
Gonzalez F, Relova JL, Prieto A, Peleteiro M (2005) Evidence of basal temporo-occipital cortex involvement in stereoscopic vision in humans: a study with subdural electrode recordings. Cereb Cortex 15:117–122
Granziera C, DaSilva AF, Snyder J, Tuch DS, Hadjikhani N (2006) Anatomical alterations of the visual motion processing network in migraine with and without aura. PLoS Med 3:e402
Grazzi L, Chiapparini L, Ferraro S, Usai S, Andrasik F, Mandelli ML, Bruzzone MG, Bussone G (2010) Chronic migraine with medication overuse pre-post withdrawal of symptomatic medication: clinical results and FMRI correlations. Headache 50:998–1004
Gwilym SE, Filippini N, Douaud G, Carr AJ, Tracey I (2010) Thalamic atrophy associated with painful osteoarthritis of the hip is reversible after arthroplasty: a longitudinal voxel-based morphometric study. Arthr Rheum 62:2930–2940
Hadjikhani N, Sanchez Del Rio M, Wu O, Schwartz D, Bakker D, Fischl B, Kwong KK, Cutrer FM, Rosen BR, Tootell RB, Sorensen AG, Moskowitz MA (2001) Mechanisms of migraine aura revealed by functional MRI in human visual cortex. Proc Natl Acad Sci USA 98:4687–4692
Hall SD, Barnes GR, Hillebrand A, Furlong PL, Singh KD, Holliday IE (2004) Spatio-temporal imaging of cortical desynchronization in migraine visual aura: a magnetoencephalography case study. Headache 44:204–208
Hamedani AG, Rose KM, Peterlin BL, Mosley TH, Coker LH, Jack CR, Knopman DS, Alonso A, Gottesman RF (2013) Migraine and white matter hyperintensities: the ARIC MRI study. Neurology 81:1308–1313
Headache Classification Subcommittee of the International Headache Society (2004) The international classification of headache disorders: 2nd edition. Cephalalgia 24 Suppl 1:9–160
Kim JH, Suh SI, Seol HY, Oh K, Seo WK, Yu SW, Park KW, Koh SB (2008) Regional grey matter changes in patients with migraine: a voxel-based morphometry study. Cephalalgia 28:598–604
Lakhan SE, Avramut M, Tepper SJ (2013) Structural and functional neuroimaging in migraine: insights from 3 decades of research. Headache 53:46–66
Lee HW, Hong SB, Seo DW, Tae WS, Hong SC (2000) Mapping of functional organization in human visual cortex: electrical cortical stimulation. Neurology 54:849–854
Maizels M, Aurora S, Heinricher M (2012) Beyond neurovascular: migraine as a dysfunctional neurolimbic pain network. Headache 52:1553–1565
Maleki N, Becerra L, Nutile L, Pendse G, Brawn J, Bigal M, Burstein R, Borsook D (2011) Migraine attacks the basal ganglia. Mol Pain 7:71
May A (2008) Chronic pain may change the structure of the brain. Pain 137:7–15
McKendrick AM, Badcock DR (2004) Motion processing deficits in migraine. Cephalalgia 24:363–372
Messina R, Rocca MA, Colombo B, Valsasina P, Horsfield MA, Copetti M, Falini A, Comi G, Filippi M (2013) Cortical abnormalities in patients with migraine: a surface-based analysis. Radiology 268:170–180
Ozge A, Termine C, Antonaci F, Natriashvili S, Guidetti V, Wober-Bingol C (2011) Overview of diagnosis and management of paediatric headache. Part I: diagnosis. J Headache Pain 12:13–23
Palm-Meinders IH, Koppen H, Terwindt GM, Launer LJ, Konishi J, Moonen JM, Bakkers JT, Hofman PA, van Lew B, Middelkoop HA, van Buchem MA, Ferrari MD, Kruit MC (2012) Structural brain changes in migraine. JAMA 308:1889–1897
Rho YI, Chung HJ, Suh ES, Lee KH, Eun BL, Nam SO, Kim WS, Eun SH, Kim YO (2011) The role of neuroimaging in children and adolescents with recurrent headaches—multicenter study. Headache 51:403–408
Rocca MA, Ceccarelli A, Falini A, Colombo B, Tortorella P, Bernasconi L, Comi G, Scotti G, Filippi M (2006) Brain gray matter changes in migraine patients with T2-visible lesions: a 3-T MRI study. Stroke 37:1765–1770
Rocca MA, Pagani E, Colombo B, Tortorella P, Falini A, Comi G, Filippi M (2008) Selective diffusion changes of the visual pathways in patients with migraine: a 3-T tractography study. Cephalalgia 28:1061–1068
Rodriguez-Raecke R, Niemeier A, Ihle K, Ruether W, May A (2009) Brain gray matter decrease in chronic pain is the consequence and not the cause of pain. J Neurosci 29:13746–13750
Schmidt-Wilcke T, Leinisch E, Ganssbauer S, Draganski B, Bogdahn U, Altmeppen J, May A (2006) Affective components and intensity of pain correlate with structural differences in gray matter in chronic back pain patients. Pain 125:89–97
Schmidt-Wilcke T, Leinisch E, Straube A, Kampfe N, Draganski B, Diener HC, Bogdahn U, May A (2005) Gray matter decrease in patients with chronic tension type headache. Neurology 65:1483–1486
Schmidt-Wilcke T, Luerding R, Weigand T, Jurgens T, Schuierer G, Leinisch E, Bogdahn U (2007) Striatal grey matter increase in patients suffering from fibromyalgia—a voxel-based morphometry study. Pain 132(Suppl 1):S109–S116
Schmitz N, Admiraal-Behloul F, Arkink EB, Kruit MC, Schoonman GG, Ferrari MD, van Buchem MA (2008) Attack frequency and disease duration as indicators for brain damage in migraine. Headache 48:1044–1055
Schweinhardt P, Kuchinad A, Pukall CF, Bushnell MC (2008) Increased gray matter density in young women with chronic vulvar pain. Pain 140:411–419
Smith SM, Zhang Y, Jenkinson M, Chen J, Matthews PM, Federico A, De Stefano N (2002) Accurate, robust, and automated longitudinal and cross-sectional brain change analysis. Neuroimage 17:479–489
Stankewitz A, Aderjan D, Eippert F, May A (2011) Trigeminal nociceptive transmission in migraineurs predicts migraine attacks. J Neurosci 31:1937–1943
Wiech K, Ploner M, Tracey I (2008) Neurocognitive aspects of pain perception. Trends Cogn Sci 12:306–313
Yu D, Yuan K, Zhao L, Dong M, Liu P, Wang G, Liu J, Sun J, Zhou G, von Deneen KM, Liang F, Qin W, Tian J (2012) Regional homogeneity abnormalities in patients with interictal migraine without aura: a resting-state study. NMR Biomed 25:806–812
Conflicts of interest
The authors declare that they have no conflict of interest related to the publication of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rocca, M.A., Messina, R., Colombo, B. et al. Structural brain MRI abnormalities in pediatric patients with migraine. J Neurol 261, 350–357 (2014). https://doi.org/10.1007/s00415-013-7201-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-013-7201-y