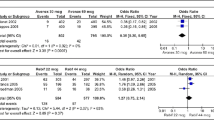
Abstract.
A proportion of people with multiple sclerosis (MS) treated with interferon (IFN) â develop neutralising anti-IFN β antibodies (NABs). The immunogenicity of the available commercial compounds relates to the genetic structure of the IFN β molecule, its mode of production, glycosylation status, aggregate formation, commercial formulation, potency, dose, frequency and, possibly, route of administration. At present, it is not possible to predict who will develop NABs usually appear within the first 2 years of starting therapy. In patients treated with IFN β in whom NABs persist for a significant period of time, their presence is associated with a reduction in both the biological effects and clinical efficacy. Approximately one third of NAB-positive patients with a titre > 20 NU/mL will revert to NAB-negative status with long-term follow-up. The persistence of NABs appears to be linked to the type of IFN β treatment as well as the titre of antibodies. The overall efficacy of IFN β and, hence, of any biological disease-modifying treatment (DMT) would be substantially improved if the development of NABs could be prevented or reversed. Although the overall efficacy of IFN β in MS is relatively modest, the efficacy in individuals who remain NAB-negative is considerably better than in those who become persistently NAB-positive. One could argue that when comparing the ‘true’ clinical efficacy of different IFN β products, the comparisons should be limited to the cohorts that remain NAB-negative. As a corollary, the therapeutic efficacy of IFN β could be maximised if patients who tolerate higher-dose preparations could be prevented from developing persistent NABs. Strategies employed to prevent or reverse the development of NABs with other biological compounds (e. g. insulin, factor VIII, IFN β, recombinant human erythropoietin) include improvements in the manufacturing process, immunosuppression, induction of tolerance and deimmunisation, and these should be considered in relation to biological DMT therapy as part of future clinical studies.
Similar content being viewed by others
References
Schellekens H (2002) Bioequivalence and the immunogenicity of biopharmaceuticals. Nat Rev Drug Discov 1:457–462
Giovannoni G, Munschauer FE 3rd, Deisenhammer F (2002) Neutralising antibodies to interferon beta during the treatment of multiple sclerosis. J Neurol Neurosurg Psychiatry 73:465–469
PRISMS Study Group and the University of British Columbia MS/MRI Analysis Group (2001) PRISMS-4: long-term efficacy of interferon-beta-1a in relapsing MS. Neurology 56:1628–1636
The IFNB Multiple Sclerosis Study Group (1993) Interferon beta-1b is effective in relapsing-remitting multiple sclerosis. I. Clinical results of a multicenter, randomized, double-blind, placebo-controlled trial. Neurology 43:655–661
The IFNB Multiple Sclerosis Study Group and The University of British Columbia MS/MRI Analysis Group (1995) Interferon beta-1b in the treatment of multiple sclerosis: final outcome of the randomized controlled trial. Neurology 45:1277–1285
The IFNB Multiple Sclerosis Study Group and the University of British Columbia MS/MRI Analysis Group (1996) Neutralizing antibodies during treatment of multiple sclerosis with interferon beta-1b: experience during the first three years. Neurology 47:889–894
Ross C, Clemmesen KM, Svenson M, Sorensen PS, Koch-Henriksen N, Skovgaard GL, Bendtzen K and the Danish Multiple Sclerosis Study Group (2000) Immunogenicity of interferon-beta in multiple sclerosis patients: influence of preparation, dosage, dose frequency, and route of administration. Ann Neurol 48:706–712
Miller DH, Khan OA, Sheremata WA, Blumhardt LD, Rice GP, Libonati MA, Willmer-Hulme AJ, Dalton CM, Miszkiel KA, O’Connor PW and the International Natalizumab Multiple Sclerosis Trial Group (2003) A controlled trial of natalizumab for relapsing multiple sclerosis. N Engl J Med 348:15–23
Salama HH, Hong J, Zang YC, El-Mongui A, Zhang J (2003) Blocking effects of serum reactive antibodies induced by glatiramer acetate treatment in multiple sclerosis. Brain 126:2638–2647
Pungor E Jr, Files JG, Gabe JD, Do LT, Foley WP,Gray JL, Nelson JW, Nestaas E, Taylor JL, Grossberg SE (1998) A novel bioassay for the determination of neutralizing antibodies to IFN-beta1b. J Interferon Cytokine Res 18:1025–1030
WHO Expert Committee on Biological Standardisation (1985) Thirty-fifth report.WHO Technical Report Series 725. World Health Organisation, Geneva
Sorensen PS, Ross C, Clemmesen KM, Bendtzen K, Frederiksen JL, Jensen K, Kristensen O, Petersen T, Rasmussen S, Ravnborg M, Stenager E, Koch-Henriksen N and the Danish Multiple Sclerosis Study Group (2003) Clinical importance of neutralising antibodies against interferon beta in patients with relapsing-remitting multiple sclerosis. Lancet 362:1184–1191
Bellomi F, Scagnolari C, Tomassini V, Gasperini C, Paolillo A, Pozzilli C, Antonelli G (2003) Fate of neutralizing and binding antibodies to IFN beta in MS patients treated with IFN beta for 6 years. J Neurol Sci 215:3–8
Gneiss C, Reindl M, Lutterotti A, Ehling R, Egg R, Khalil M, Berger T, Deisenhammer F (2003) Interferon-beta: the neutralizing antibody (NAb) titer predicts reversion to NAb negativity. Mult Scler 9(Suppl 9):P482. S119
Dubois BD, Keenan E, Porter BE, Kapoor R, Rudge P, Thompson AJ, Miller DH, Giovannoni G (2003) Interferon beta in multiple sclerosis: experience in a British specialist multiple sclerosis centre. J Neurol Neurosurg Psychiatry 74:946–949
van Boxel-Dezaire AH, van Trigt-Hoff SC, Killestein J, Schrijver HM, van Houwelingen JC, Polman CH, Nagelkerken L (2000) Contrasting responses to interferon beta-1b treatment in relapsing-remitting multiple sclerosis: does baseline interleukin-12p35 messenger RNA predict the efficacy of treatment? Ann Neurol 48:313–322
Petereit HF, Nolden S, Schoppe S, Bamborschke S, Pukrop R, Heiss WD (2002) Low interferon gamma producers are better treatment responders: a two-year follow-up of interferon beta-treated multiple sclerosis patients. Mult Scler 8:492–494
Wandinger KP, Lunemann JD, Wengert O, Bellmann-Strobl J, Aktas O, Weber A, Grundstrom E, Ehrlich S, Wernecke KD, Volk HD, Zipp F (2003) TNF-related apoptosis inducing ligand (TRAIL) as a potential response marker for interferon-beta treatment in multiple sclerosis. Lancet 361:2036–2043
Waubant E, Vukusic S, Gignoux L, Dubief FD, Achiti I, Blanc S, Renoux C, Confavreux C (2003) Clinical characteristics of responders to interferon therapy for relapsing MS. Neurology 61:184–189
Villoslada P, Barcellos LF, Rio J, Begovich AB, Tintore M, Sastre-Garriga J, Baranzini SE, Casquero P, Hauser SL, Montalban X, Oksenberg JR (2002) The HLA locus and multiple sclerosis in Spain. Role in disease susceptibility, clinical course and response to interferon-beta. J Neuroimmunol 130:194–201
Panitch H, Goodin DS, Francis G, Chang P, Coyle PK, O’Connor P, Monaghan E, Li D, Weinshenker B, the EVIDENCE (EVidence of Interferon Doseresponse: European North American Comparative Efficacy) Study Group and the University of British Columbia MS/MRI Research Group (2002) Randomized, comparative study of interferon beta-1a treatment regimens in MS: the EVIDENCE trial. Neurology 59:1496–1506
Durelli L,Verdun E, Barbero P, Bergui M, Versino E, Ghezzi A, Montanari E, Zaffaroni M and the Independent Comparison of Interferon (INCOMIN) Trial Study Group (2002) Every-otherday interferon beta-1b versus once-weekly interferon beta-1a for multiple sclerosis: results of a 2-year prospective randomised multicentre study (INCOMIN). Lancet 359:1453–1460
Eckardt KU, Casadevall N (2003) Pure red-cell aplasia due to anti-erythropoietin antibodies. Nephrol Dial Transplant 18:865–869
Casadevall N (2002) Antibodies against rHuEPO: native and recombinant. Nephrol Dial Transplant 17 (Suppl 5):42–47
Bertolotto A, Malucchi S, Sala A, Orefice G, Carrieri PB, Capobianco M, Milano E, Melis F, Giordana MT (2002) Differential effects of three interferon betas on neutralising antibodies in patients with multiple sclerosis: a follow up study in an independent laboratory. J Neurol Neurosurg Psychiatry 73:148–153
Baert F, Noman M, Vermeire S, Van Assche G, D’Haens G, Carbonez A, Rutgeerts P (2003) Influence of immunogenicity on the long-term efficacy of infliximab in Crohn’s disease. N Engl J Med 348:601–608
Ingerslev J (2000) Hemophilia. Strategies for the treatment of inhibitor patients. Haematologica 85(10 Suppl):15–20
Pozzilli C, Antonini G, Bagnato F, Mainero C, Tomassini V, Onesti E, Fantozzi R, Galgani S, Pasqualetti P, Millefiorini E, Spadaro M, Dahlke F, Gasperini C (2002) Monthly corticosteroids decrease neutralizing antibodies to IFN beta1b: a randomized trial in multiple sclerosis. J Neurol 249:50–56
Herndon RM, Jacobs LD, Coats ME, Goodkin DE, Mass MK, Rickert JR, Rudick RA, Wauhant EL, Weinstock-Guttman B, Scaramucci JO, Burnett BK, Jones WE, Simonian NA (1999). Results of an ongoing, open-label, safety-extension study of interferon beta-1a (Avonex) treatment in multiple sclerosis. Int J MS Care 2:1–6
Perini P, Facchinetti A, Bulian P, Massaro AR, Pascalis DD, Bertolotto A, Biasi G, Gallo P (2001) Interferon-beta (IFN-beta) antibodies in interferon-beta1a-and interferon-beta1b-treated multiple sclerosis patients. Prevalence, kinetics, cross-reactivity, and factors enhancing interferon-beta immunogenicity in vivo. Eur Cytokine Netw 12:56–61
Giovannoni G (2003) Strategies to treat and prevent the development of neutralizing anti-interferon-beta antibodies. Neurology 61(9 Suppl 5):S13–S17
Ure DR, Rodriguez M (2002) Polyreactive antibodies to glatiramer acetate promote myelin repair in murine model of demyelinating disease. FASEB J 16:1260–1262
Teitelbaum D, Brenner T, Abramsky O, Aharoni R, Sela M, Arnon R (2003) Antibodies to glatiramer acetate do not interfere with its biological functions and therapeutic efficacy. Mult Scler 9:592–599
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Giovannoni, G. Optimising MS disease-modifying therapies: antibodies in perspective. J Neurol 251 (Suppl 5), v30–v35 (2004). https://doi.org/10.1007/s00415-004-1505-x
Issue Date:
DOI: https://doi.org/10.1007/s00415-004-1505-x