Abstract
Purpose
Low serum creatinine/cystatin C ratio (CCR) is associated with unfavorable characteristics in patients with chronic obstructive pulmonary disease (COPD); however, the relationship between CCR and in-hospital mortality of patients with acute exacerbation of COPD (AECOPD) is unexplored. Our objective was to assess the value of CCR for predicting in-hospital mortality of patients hospitalized with AECOPD.
Methods
Patients with AECOPD (n = 597) were retrospectively enrolled. Patient’s clinical characteristics and laboratory tests, including serum cystatin C and creatinine, were reviewed. The prediction value of CCR was evaluated using area under the receiver operating characteristic curve (AUC) values. Factors potentially impacting in-hospital mortality were investigated using univariate and multivariate logistic regression analyses.
Results
Mortality rate during hospitalization was 10.05%. CCR was lower in non-surviving vs. survived patients (41.67 vs. 61.52, P < 0.001). AUC value for CCR for in-hospital mortality prediction was 0.79 [95% confidence interval (CI) 0.73–0.85]. On multivariate logistic regression analysis, in-hospital mortality was strongly associated with CCR < 52.27 [odds ratio (OR) 6.23, 95% CI (3.00–12.92), P < 0.001], age ≥ 81 years [OR 2.97, 95% CI (1.20–7.37), P = 0.019], oxygenation index < 300 [OR 3.28, 95% CI (1.27–8.44), P = 0.014], CRP > 8 mg/L [OR 1.84, 95% CI (1.15–2.95), P = 0.012], and D-dimer > 500 ng/L [OR 5.19, 95% CI (1.51–17.79), P = 0.009].
Conclusions
CCR was significantly lower, and is a potential prognostic indicator, in patients with AECOPD who died during hospitalization.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chronic obstructive pulmonary disease (COPD) is an airway disease characterized by incompletely reversible airflow limitation that causes high mortality and morbidity worldwide [1]. Acute exacerbation (AE) is the most common reason for hospitalization among patients with COPD, and is associated with a rapid decline of respiratory function, impaired quality of life, and poor prognosis [2, 3]. In addition, frequent exacerbation is recognized as a significant risk factor contributing to increased mortality in patients with COPD [4]. Thus, early and accurate diagnosis of AE, risk stratification of patients, and prediction of treatment response warrant increased research attention.
Malnutrition and sarcopenia are common among patients with COPD, particularly when the disease progresses to an advanced stage [5, 6], and is associated with adverse effects on exercise, muscle function, lung function, and increased rates of exacerbation, hospitalization, and mortality, as well as higher costs [7,8,9]. Therefore, accurate assessment of malnutrition and sarcopenia in patients with COPD is of great importance [10]. Currently, the most commonly used methods to evaluate nutritional status in patients with COPD in the clinic are as follows: ultrasonography, including diaphragm thickness and echo intensity of rectus femoris; computed tomography (CT) scan, including cross-sectional areas of the midthigh and erector spinae muscles; and screening and assessment tools, including body mass index (BMI), the Mini Nutrition Assessment, Nutrition Risk Screening 2002, and Subjective Global Assessment [11]. However, these approaches have limitations in terms of specificity, sensitivity, and convenience [12, 13]; thus, there is an urgent need to find alternative evaluation methods. The serum creatinine (SCr)/Cystatin C (Cys C) ratio (CCR) has emerged as a promising surrogate marker of muscle wasting and dysfunction [14,15,16] independent of renal function [17] since its first proposal by Kashani et al. [18]. Further, CCR is a marker of poor prognosis in patients with coronary artery disease [19] and serious neurological disease [20], as well as in patients receiving intensive care and continuous kidney replacement therapy [21].
In the context of COPD, CCR is a valuable marker for evaluating skeletal muscle mass, disease severity, and prognosis. Specifically, CCR is a useful indicator of muscle strength decline in male patients with AECOPD [22], and is significantly associated with forced vital capacity and forced expiratory volume in 1 s (FEV1) values, particularly in former smokers [23]. Further, low CCR values are associated with longer duration of hospital admission, increased dyspnea, greater functional impairment in patients admitted for AECOPD [24], and is also an independent predictor of hospitalization in the following year for outpatients with stable COPD [25]. Nevertheless, to the best of our knowledge, there has been no published study that has assessed the relationship between CCR value and in-hospital mortality of patients hospitalized with AECOPD. Therefore, in this study, we aimed to clarify the potential value of the CCR for predicting in-hospital mortality of patients hospitalized with AECOPD.
Materials and Methods
Study Design and Subjects
Patients with AECOPD hospitalized at the Department of Respiratory and Critical Care Medicine of Wuhu Hospital of Traditional Chinese Medicine, China were enrolled in this retrospective observational study between January 2016 and February 2022. The primary outcome was in-hospital mortality. Diagnosis of COPD was based on the Global Initiative for Chronic Obstructive Lung Disease (GOLD 2018) criteria [1]. Inclusion criteria were hospitalized patients with AECOPD, defined as an acute deterioration in respiratory symptoms of sufficient severity to require admission. Exclusion criteria were as follows: (1) age < 40 years; (2) end-stage renal disease (ESRD), defined as estimated GFR < 15 mL/min/1.73 m2, or already receiving renal replacement therapy; (3) acute renal injury; (4) receiving palliative care; or (5) missing data required for the study. For patients with multiple hospital admissions during the study period, only the first admission record was used. As a retrospective analysis, the study was not registered.
Data Collection
Clinical data were extracted from the electronic medical records system. Data collected included general information (age, gender, and number of hospitalizations due to AE in the previous year); underlying disease (hypertension, diabetes mellitus, coronary heart disease, lung cancer, and heart failure); laboratory tests [platelet count (PLT), SCr, Cys C, C-reactive protein (CRP), D-dimer, oxygenation index (OI) (SpO2/FiO2), partial pressure of carbon dioxide (pCO2), and blood lactate (Lac)]; and treatment outcomes (non-invasive ventilation (NIV), invasive mechanical ventilation (IMV), ICU admission, length of hospital stay (LOS), and prognosis (survival/death)). Hypercapnia was defined as a pCO2 > 45 mmHg. CCR was calculated as CCR = SCr/Cys C. Data were collected from laboratory tests conducted at the first examination on admission.
Statistical Analysis
Quantitative variables are expressed as median with interquartile range (IQR), and qualitative variables as frequencies or percentages. Statistical comparisons of variables between the survived and non-survived groups were performed using the chi-square test for categorical variables and the Mann–Whitney U test for continuous variables. Receiver operating characteristic (ROC) curves were used to evaluate the ability of CCR to predict in-hospital mortality, and Youden’s index was used to determine the optimal cut-off point. The predictive accuracy of CCR was determined using the area under the ROC curve (AUC). Subsequently, patients were classified into two groups based on the CCR cut-off value. Risk factors potentially influencing in-hospital mortality were analyzed using univariable and multivariable logistic regressions. The goodness of fit of models was assessed using calibration curves. The cut-off values for OI, CRP, and D-dimer were determined based on the clinical reference intervals used in our hospital laboratory. Age was categorized based on tertile values for the cohort. All statistical analyses were performed using SPSS 26.0 (IBM, SPSS) and GraphPad Prism 8.0 (GraphPad Software).
Results
Comparison of Patient Characteristics Between Non-survival and Survival Groups
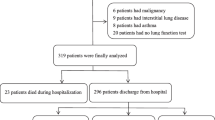
A flow chart showing patient inclusion in this study is presented as Fig. 1. In total, 597 patients were ultimately included in this study and 60 (10.1%) died during hospitalization. Participant baseline characteristics and comparisons between non-survival and survival groups are summarized in Table 1. Median (IQR) CCR values in total, the non-survived, and survived patients were 59.7 (48.4–72.4), 41.7 (34.7–52.1), and 61.5 (50.8–73.8), respectively; the difference between survived and non-survived patients was significant (P < 0.001). However, SCr did not differ significantly between the two groups (P = 0.590). Compared to survived patients, those who died were older, had more frequent hospitalization due to AE in the previous year, longer LOS, and included higher proportions with heart failure, lung cancer, and coronary heart disease. CRP, Cys C, D-dimer levels, proportion with hypercapnia, and patients requiring NIV, IMV, and ICU admission were higher in the non-survivor group. Further, OI and Hb were significantly lower in non-survived than survived patients, whereas there were no significant differences in PLT, PDW, and Lac between the two groups.
Relationships Between CCR Levels and Clinically Relevant Results
ROC curves to evaluate the ability of CCR and Cys C predicting in-hospital mortality are shown in Fig. 2. The AUC values of CCR and Cys C were 0.79 (95% CI 0.73–0.85) and 0.73 (95% CI 0.66–0.81), respectively. The optimal cut-off CCR value was 52.27, corresponding to sensitivity and specificity values of 76.7% and 71.5%, respectively. Next, we divided participants into low (CCR < 52.27, N = 199, 33.3%) and high (CCR ≥ 52.27, N = 398, 66.7%) CCR groups, according to the CCR cut-off value and found significant differences between the two groups (Table 2). Compared with the high CCR group, patients in the low CCR group were more likely to be older, have more frequent hospitalization due to AE in the previous year, and have a longer LOS, and included higher proportions with coexisting lung cancer and heart failure, ICU admission, and elevated D-dimer. Conversely, Hb levels were significantly lower in the low CCR group than those in the high group. Further, there were significantly more male patients in the high CCR group. No significant differences in OI, PLT, PDW, or CRP were observed between the groups.
Univariate and Multivariate Regression Analyses of Factors Associated with In-hospital Mortality
The results of univariate logistic regression analysis of risk factors associated with in-hospital mortality are presented in Table 3. The in-hospital mortality of patients with low CCR levels was 23.1%, which was significantly higher than that in patients with high CCR levels (3.5%) [OR 8.2, 95% CI (4.4–15.4)]. Further, age, number of hospitalizations due to AE, hypercapnia, heart failure, OI, CRP, and D-dimer were also significant predictors of in-hospital mortality. Based on the literature, previous clinical experience, and the results of univariate analysis, we selected CCR value, age, number of hospitalizations due to AE in the past year, coexisting heart failure, OI, hypercapnia, CRP, and D-dimer for inclusion in multivariate logistic regression analysis (Fig. 3), which indicated that CCR < 52.27 [OR 6.23, 95% CI (3.00–12.92), P < 0.001], age ≥ 81 years, number of hospitalizations due to AE in the previous year, OI < 300, CRP > 8 mg/L, and D-dimer > 500 ng/L were significant predictors of in-hospital mortality for patients hospitalized with AECOPD. A calibration curve for the multivariable logistic regression model demonstrated that the model has sufficient goodness of fit (Hosmer‒Lemeshow goodness of fit test, P = 0.693).
Discussion
In the present study, the value of CCR for predicting in-hospital mortality in patients hospitalized with AECOPD was assessed. We found that CCR was significantly lower in the non-survival group than in the survival group among patients hospitalized for AECOPD. The AUC of the ROC curve for CCR prediction of in-hospital mortality was 0.79 (95% CI 0.73–0.85), and the optimal cut-off CCR value was 52.27, with maximum sensitivity and specificity of 76.7% and 71.5%, respectively. More importantly, multivariate logistic regression analysis confirmed that CCR < 52.27 was an independent risk factor for in-hospital mortality after adjusting for confounding factors, including hypercapnia and history of heart failure. These results clearly indicate that the CCR value has potential to be an excellent predictor of short-term prognosis for patients with AECOPD during hospitalization. We also found that age ≥ 81 years, number of hospitalizations due to AE in the previous year, OI < 300, CRP > 8 mg/L, and D-dimer > 500 ng/L were strongly associated with increased hospital mortality.
Plasma Cys C has been used to predict the prognosis of diseases [26,27,28], including COPD. Hu et al. [29] reported that Cys C was a strong and independent risk factor for hospital mortality secondary to AECOPD. They reported an AUC value for prediction of death using Cys C of 0.77 (95% CI 0.70–0.84), which is generally consistent with the results of our study; however, importantly, the AUC for CCR prediction of in-hospital mortality in the current study was 0.79 (95% CI 0.73–0.85), which was higher than that for Cys C alone (0.73, 95% CI 0.66–0.81), indicating that CCR has better short-term predictive value.
In this study, patients in the high CCR group tended to be younger and more often male than those in the CCR low group. One possible explanation for this is that CCR value is strongly associated with muscle mass [30], and low CCR can be considered an independent predictor of sarcopenia [31]. In general, females have less muscle mass than males, and aging is also associated with decreased muscle mass [32]. Hence, CCR values tend to be lower in older adult and female populations [33]. We also found that patients in the low CCR group had more frequent hospitalization due to AE in the previous year than those in the CCR high group. This conclusion is supported by a similar finding reported by Hirai et al. [34], in which low CCR was a predictor of severe exacerbation, independent of age, COPD Assessment Test score, and %FEV1 (P < 0.01). Lu et al. [19] investigated the association between CCR and the prevalence of major adverse cardiovascular events in patients with coronary artery disease and found that those in the low CCR group were more likely to have congestive heart failure (P < 0.05). Jung et al. [35] found that patients with CCR values in the highest quartile had shorter lengths of ICU and hospital stay (both P < 0.001) in patients with cancer. These results are all concordant with our findings; however, no significant differences in the requirement for, or duration of, mechanical ventilation were identified between the high and low CCR groups in our study, inconsistent with the results of a previous investigation [20]; further studies are needed to explain this discrepancy.
One of the most important findings of our study is that CCR < 52.27 is significantly associated with in-hospital mortality for patients hospitalized with AECOPD [OR 6.23, 95% CI (3.00–12.92), P < 0.001], independently of hypercapnia and history of heart failure. This may be attributable to reduced muscle mass, which can be accurately assessed by a low CCR value [14, 34, 36], and is directly correlated with poor prognosis in various diseases, including short-term and long-term mortality. Jaitovich et al. [37] performed a prospective observational study to investigate the association of skeletal muscle and fat mass at ICU admission with survival and disability at hospital discharge. They concluded that pectoralis muscle area, determined by CT scan at the time of ICU admission, was associated with 6-month and hospital survival, as well as ICU-free days, despite not being a good predictor of regaining an independent lifestyle after discharge. Zheng et al. [38] demonstrated that lower CCR could be used to identify sarcopenia in patients with esophageal cancer and was associated with shorter overall survival. Further, they found the all-cause mortality risk gradually decreased with increased CCR. In addition to CCR < 52.27, we found that age ≥ 81 years, frequency of hospitalization due to AE in the previous year, OI < 300, CRP > 8 mg/L, and D-dimer > 500 ng/L were closely associated with in-hospital mortality. These findings are in line with the conclusions of earlier research. Crisafulli et al. [39] found that age and acute severe hypoxemia at admission were significant independent predictors of mortality in patients hospitalized with AECOPD. Soler-Cataluña et al. [40] showed that patients with three or more acute COPD exacerbations had the greatest mortality risk. Hu et al. [41] found that D-dimer was a strong and independent risk factor associated with in-hospital and 1-year death of patients with AECOPD requiring hospitalization. Sneh et al. [42] reported that high CRP levels at baseline, and persistently elevated CRP may predict mortality in patients with AE-COPD requiring mechanical ventilation.
Inevitably, this study has some inherent limitations. First, there may have been selection bias and the results are less reliable than those of a prospective study would be, because of the single-center retrospective design. Second, although patients enrolled in this study had confirmed COPD diagnosis, most had disease too severe to have lung function examined during hospitalization. Thus, we could not consider the impact of lung function indices. Third, the cut-off value of CCR was determined by the Youden index, which might not be a practical value in clinical practice to some extent. If the aim is to design a scoring tool, other practical techniques should be considered, as described in these studies [43, 44]. Finally, we did not include other indicators used to assess nutritional status, such as BMI, geriatric nutritional risk index, controlling nutritional status score, and prognostic nutritional index; therefore, we could not compare the predictive efficacy of these different indicators for in-hospital mortality.
Conclusions
CCR was significantly lower among patients hospitalized with AECOPD who died during admission than those who survived; therefore, it could be a powerful predictor of mortality by incorporating it within clinical risk scores.
References
Mirza S, Clay RD, Koslow MA et al (2018) COPD guidelines: a review of the 2018 GOLD report. Mayo Clin Proc 93:1488–1502. https://doi.org/10.1016/j.mayocp.2018.05.026
Kim V, Aaron SD (2018) What is a COPD exacerbation? Current definitions, pitfalls, challenges and opportunities for improvement. Eur Respir J. https://doi.org/10.1183/13993003.01261-2018
Dransfield MT, Kunisaki KM, Strand MJ et al (2017) Acute exacerbations and lung function loss in smokers with and without chronic obstructive pulmonary disease. Am J Respir Crit Care Med 195:324–330. https://doi.org/10.1164/rccm.201605-1014OC
Hillas G, Perlikos F, Tzanakis N (2016) Acute exacerbation of COPD: is it the “stroke of the lungs”? Int J Chron Obstruct Pulmon Dis 11:1579–1586. https://doi.org/10.2147/COPD.S106160
Itoh M, Tsuji T, Nemoto K et al (2013) Undernutrition in patients with COPD and its treatment. Nutrients 5:1316–1335. https://doi.org/10.3390/nu5041316
Sepulveda-Loyola W, Osadnik C, Phu S et al (2020) Diagnosis, prevalence, and clinical impact of sarcopenia in COPD: a systematic review and meta-analysis. J Cachexia Sarcopenia Muscle 11:1164–1176. https://doi.org/10.1002/jcsm.12600
Keogh E, Mark Williams E (2021) Managing malnutrition in COPD: a review. Respir Med 176:106248. https://doi.org/10.1016/j.rmed.2020.106248
Mete B, Pehlivan E, Gulbas G et al (2018) Prevalence of malnutrition in COPD and its relationship with the parameters related to disease severity. Int J Chron Obstruct Pulmon Dis 13:3307–3312. https://doi.org/10.2147/COPD.S179609
Cavailles A, Brinchault-Rabin G, Dixmier A et al (2013) Comorbidities of COPD. Eur Respir Rev 22:454–475. https://doi.org/10.1183/09059180.00008612
van Beers M, Rutten-van Molken M, van de Bool C et al (2020) Clinical outcome and cost-effectiveness of a 1-year nutritional intervention programme in COPD patients with low muscle mass: the randomized controlled NUTRAIN trial. Clin Nutr 39:405–413. https://doi.org/10.1016/j.clnu.2019.03.001
Raad S, Smith C, Allen K (2019) Nutrition status and chronic obstructive pulmonary disease: can we move beyond the body mass index? Nutr Clin Pract 34:330–339. https://doi.org/10.1002/ncp.10306
Buckinx F, Landi F, Cesari M et al (2018) Pitfalls in the measurement of muscle mass: a need for a reference standard. J Cachexia Sarcopenia Muscle 9:269–278. https://doi.org/10.1002/jcsm.12268
Masanes F, Rojano ILX, Salva A et al (2017) Cut-off points for muscle mass—not grip strength or gait speed—determine variations in sarcopenia prevalence. J Nutr Health Aging 21:825–829. https://doi.org/10.1007/s12603-016-0844-5
Tabara Y, Kohara K, Okada Y et al (2020) Creatinine-to-cystatin C ratio as a marker of skeletal muscle mass in older adults: J-SHIPP study. Clin Nutr 39:1857–1862. https://doi.org/10.1016/j.clnu.2019.07.027
Fujita K, Ohkubo H, Nakano A et al (2022) Serum creatinine/cystatin C ratio is a surrogate marker for sarcopenia in patients with idiopathic pulmonary fibrosis. BMC Pulm Med 22:203. https://doi.org/10.1186/s12890-022-02000-3
An JN, Kim JK, Lee HS et al (2022) Serum cystatin C to creatinine ratio is associated with sarcopenia in non-dialysis-dependent chronic kidney disease. Kidney Res Clin Pract. https://doi.org/10.23876/j.krcp.21.214
Lin YL, Chen SY, Lai YH et al (2020) Serum creatinine to cystatin C ratio predicts skeletal muscle mass and strength in patients with non-dialysis chronic kidney disease. Clin Nutr 39:2435–2441. https://doi.org/10.1016/j.clnu.2019.10.027
Kashani KB, Frazee EN, Kukralova L et al (2017) Evaluating muscle mass by using markers of kidney function: development of the sarcopenia index. Crit Care Med 45:e23–e29. https://doi.org/10.1097/CCM.0000000000002013
Lu YW, Tsai YL, Chou RH et al (2021) Serum creatinine to cystatin C ratio is associated with major adverse cardiovascular events in patients with obstructive coronary artery disease. Nutr Metab Cardiovasc Dis 31:1509–1515. https://doi.org/10.1016/j.numecd.2021.01.024
Wang S, Xie L, Xu J et al (2019) Predictive value of serum creatinine/cystatin C in neurocritically ill patients. Brain Behav 9:e01462. https://doi.org/10.1002/brb3.1462
Jung CY, Joo YS, Kim HW et al (2021) Creatinine-cystatin C ratio and mortality in patients receiving intensive care and continuous kidney replacement therapy: a retrospective cohort study. Am J Kidney Dis 77:509-516.e1. https://doi.org/10.1053/j.ajkd.2020.08.014
Huang D, Xie C, Sun C et al (2022) Serum creatinine to cystatin C ratio is an effective indicator for muscle strength decline in men with acute exacerbation of chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis 17:781–789. https://doi.org/10.2147/COPD.S356314
Nishiki K, Nojiri M, Kato R et al (2021) Serum creatinine/cystatin C ratio associated with cross-sectional area of erector spinae muscles and pulmonary function in patients with chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis 16:3513–3524. https://doi.org/10.2147/COPD.S339243
Warnken-Miralles MD, López-García F, Zamora-Molina L et al (2021) Sarcopenia index in hospitalized patients with chronic obstructive pulmonary disease exacerbation. Medicina 81:323–328
Amado CA, Garcia-Unzueta MT, Lavin BA et al (2019) The ratio serum creatinine/serum cystatin c (a surrogate marker of muscle mass) as a predictor of hospitalization in chronic obstructive pulmonary disease outpatients. Respiration 97:302–309. https://doi.org/10.1159/000494296
Wu X, Xu G, Zhang S (2020) Association between cystatin C and cardiac function and long-term prognosis in patients with chronic heart failure. Med Sci Monit 26:e919422. https://doi.org/10.12659/MSM.919422
Ravn B, Prowle JR, Martensson J et al (2017) Superiority of serum cystatin C over creatinine in prediction of long-term prognosis at discharge from ICU. Crit Care Med 45:e932–e940. https://doi.org/10.1097/CCM.0000000000002537
Sun Y, Lu Q, Cheng B et al (2021) Prognostic value of cystatin C in patients with acute coronary syndrome: a systematic review and meta-analysis. Eur J Clin Invest 51:e13440. https://doi.org/10.1111/eci.13440
Hu G, Wu Y, Zhou Y et al (2016) Cystatin C as a predictor of in-hospital mortality after exacerbation of COPD. Respir Care 61:950–957. https://doi.org/10.4187/respcare.04034
Rizk JG, Streja E, Wenziger C et al (2021) Serum creatinine-to-cystatin-C ratio as a potential muscle mass surrogate and racial differences in mortality. J Ren Nutr. https://doi.org/10.1053/j.jrn.2021.11.005
Shin JY (2022) Low serum creatinine to cystatin C ratio is independently associated with sarcopenia and high carotid plaque score in patients with type 2 diabetes. Nutr Metab Cardiovasc Dis 32:1454–1462. https://doi.org/10.1016/j.numecd.2022.02.005
Thongprayoon C, Cheungpasitporn W, Kashani K (2016) Serum creatinine level, a surrogate of muscle mass, predicts mortality in critically ill patients. J Thorac Dis 8:E305–E311. https://doi.org/10.21037/jtd.2016.03.62
Lee HS, Park KW, Kang J et al (2020) Sarcopenia index as a predictor of clinical outcomes in older patients with coronary artery disease. J Clin Med. https://doi.org/10.3390/jcm9103121
Hirai K, Tanaka A, Homma T et al (2021) Serum creatinine/cystatin C ratio as a surrogate marker for sarcopenia in patients with chronic obstructive pulmonary disease. Clin Nutr 40:1274–1280. https://doi.org/10.1016/j.clnu.2020.08.010
Jung CY, Kim HW, Han SH et al (2022) Creatinine-cystatin C ratio and mortality in cancer patients: a retrospective cohort study. J Cachexia Sarcopenia Muscle. https://doi.org/10.1002/jcsm.13006
Tang T, Xie L, Hu S et al (2022) Serum creatinine and cystatin C-based diagnostic indices for sarcopenia in advanced non-small cell lung cancer. J Cachexia Sarcopenia Muscle 13:1800–1810. https://doi.org/10.1002/jcsm.12977
Jaitovich A, Khan M, Itty R et al (2019) ICU admission muscle and fat mass, survival, and disability at discharge: a prospective cohort study. Chest 155:322–330. https://doi.org/10.1016/j.chest.2018.10.023
Zheng C, Wang E, Li JS et al (2022) Serum creatinine/cystatin C ratio as a screening tool for sarcopenia and prognostic indicator for patients with esophageal cancer. BMC Geriatr 22:207. https://doi.org/10.1186/s12877-022-02925-8
Crisafulli E, Manco A, Guerrero M et al (2021) Age is a determinant of short-term mortality in patients hospitalized for an acute exacerbation of COPD. Intern Emerg Med 16:401–408. https://doi.org/10.1007/s11739-020-02420-1
Soler-Cataluña JJ, Martínez-García MA, Román Sánchez P et al (2005) Severe acute exacerbations and mortality in patients with chronic obstructive pulmonary disease. Thorax 60:925–931. https://doi.org/10.1136/thx.2005.040527
Hu G, Wu Y, Zhou Y et al (2016) Prognostic role of D-dimer for in-hospital and 1-year mortality in exacerbations of COPD. Int J Chron Obstruct Pulmon Dis 11:2729–2736. https://doi.org/10.2147/copd.S112882
Sneh A, Pawan T, Randeep G et al (2020) Acute phase proteins as predictors of survival in patients with acute exacerbation of chronic obstructive pulmonary disease requiring mechanical ventilation. COPD 17:22–28. https://doi.org/10.1080/15412555.2019.1698019
Habibzadeh F, Habibzadeh P, Yadollahie M (2016) On determining the most appropriate test cut-off value: the case of tests with continuous results. Biochem Med (Zagreb) 26:297–307. https://doi.org/10.11613/BM.2016.034
Hajian-Tilaki K (2018) The choice of methods in determining the optimal cut-off value for quantitative diagnostic test evaluation. Stat Methods Med Res 27:2374–2383. https://doi.org/10.1177/0962280216680383
Funding
This study was supported by the High-level Talents Project of the Wuhu Health Commission (2020-132, Zhixiang Chen, and 2021-134, Lei Zha).
Author information
Authors and Affiliations
Contributions
ZC and LZ conceived the idea for this report and are co-first authors. ZC, FY, WL, and WW collected data for the study. ZC and LZ performed the statistical analyses and prepared figures and tables. ZC, XM, JX, DH, WW, LC, FY, WL, and WW wrote the first draft of the manuscript. ZC and LZ reviewed, rated, and revised the manuscript. All authors approved the final version and are accountable for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest for this work.
Consent to Participate
Informed consent was waived because our study was conducted solely by collecting retrospective anonymized patient data from electronic medical records, with no influence on patient treatment.
Consent to Publish
Not applicable.
Ethical approval
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The present study was approved by the Medical Ethics Committee of Wuhu Hospital of Traditional Chinese Medicine (LW 2022-10; May 13, 2022).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, Z., Zha, L., Ma, X. et al. Serum Creatinine/Cystatin C Ratio as a Predictor of In-hospital Mortality in Patients Hospitalized with Acute Exacerbation of Chronic Obstructive Pulmonary Disease. Lung 200, 609–617 (2022). https://doi.org/10.1007/s00408-022-00568-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-022-00568-5