Abstract
Purpose
Consolidation immunotherapy with the PD-L1 inhibitor durvalumab following concurrent chemoradiotherapy (cCRT) has shown a significant survival improvement and is now a standard of care in patients with unresectable stage III or non-operable non-small cell lung cancer (NSCLC).
Methods
In this early access program cohort, demographic, disease characteristics and safety data were collected for 576 patients from 188 centers, who received durvalumab 10 mg/kg intravenous infusion every 2 weeks, until disease progression or unacceptable toxicity or for a maximum of 12 months following cCRT. Durvalumab exposure data were available for 402 patients.
Results
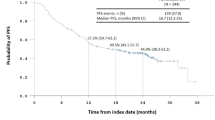
Overall, 576 patients were included, 72.9% were men, median age 64.0 years, 52.3% had a stage IIIB disease. PD-L1 status captured in 445 (77%) patients was positive (48.1%), negative (32.6%), unknown (19.3%). At the end of cCRT, adverse events (AEs) all grade ≤ 2, were reported in 22.7% of patients, mainly esophagitis (6.3%). The main reasons of discontinuation were completion of the planned 12 months of consolidation treatment (42.1% patients), disease progression (28.6%) and adverse events (19.5%). Treatment completion was similar in PDL-1 positive and PDL-1 negative patients groups. 20.7% patients had a SAE drug reaction and 17.7% stopped treatment mainly due to SAE. ADR rate and early treatment discontinuation were higher in patients > 70 years old. Death due to AEs occurred in 7 patients, 2 had interstitial lung disease.
Conclusion
Safety data with durvalumab consolidation after cCRT in a large cohort of patients with stage III NSCLC are reported in this real-life cohort. Consistent data were reported both in the PD-L1 positive and PD-L1 negative NSCLC patients in daily practice.

Similar content being viewed by others
Data Availability
Data and Material available upon request.
Code Availability
Not applicable.
References
Locher C, Debieuvre D, Coëtmeur D et al (2013) Major changes in lung cancer over the last ten years in France: The KBP-CPHG studies. Lung Cancer 81:32–38
Detterbeck FC, Boffa DJ, Kim AW, et al. (2017) The eighth edition lung cancer stage classification. Chest. 151(1):193–203.
Postmus PE, Kerr KM, Oudkerk M et al (2017) Early and locally advanced non-small-cell lung cancer (NSCLC): ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. https://doi.org/10.1093/annonc/mdx222
Ettinger DS, Aisner DL, Wood DE et al (2018) NCCN guidelines insights: non-small cell lung cancer, Version 5.2018. J Natl Compr Canc Netw 16(7):807–821
Goldstraw P, Chansky K, Crowley J et al (2016) J Thorac Oncol 11(1):39–51
Antonia SJ, Villegas A, Daniel D et al (2017) Durvalumab after chemoradiotherapy in stage III non-small-cell lung cancer. N Engl J Med 377(20):1919–1929
Antonia SJ, Villegas A, Daniel D et al (2018) Overall survival with durvalumab after chemoradiotherapy in stage III NSCLC. N Engl J Med 379(24):2342–2350
Hui R, Özgüroğlu M, Villegas A et al (2019) Patient-reported outcomes with durvalumab after chemoradiotherapy in stage III, unresectable non-small-cell lung cancer (PACIFIC): a randomised, controlled, phase 3 study. Lancet Oncol 20(12):1670–1680
Gray JE, Villegas A, Daniel D et al (2020) Three-year overall survival with durvalumab after chemoradiotherapy in stage III NSCLC-update from PACIFIC. J Thorac Oncol 15(2):288–293
Paz-Ares L, Spira A, Raben D et al (2020) Outcomes with durvalumab by tumour PD-L1 expression in unresectable, stage III non-small-cell lung cancer in the PACIFIC trial. Ann Oncol 31(6):798–806
Faivre-Finn C, Vicente D, Kurata T et al (2021) Four-year survival with durvalumab after chemoradiotherapy in stage III NSCLC-an update from the PACIFIC trial. J Thorac Oncol S1556–0864(21):00022–00028
Spigel DR, Faivre-Finn C (2021) Gray JE Five-year survival outcomes with durvalumab after chemoradiotherapy in unresectable stage III NSCLC: An update from the PACIFIC trial. ASCO 39:8511
SmPC Imfinzi®.
Blonde L, Khunti K, Harris SB et al (2018) Interpretation and Impact of Real-World Clinical Data for the Practicing Clinician. Adv Ther 35(11):1763–1774
Maissenhaelter BE, Woolmore AL, Schlag PM (2018) Real-world evidence research based on big data: Motivation-challenges-success factors. Onkologe (Berl) 24(Suppl 2):91–98
Desilets A, Blanc-Durand F, Lau S et al (2021) Durvalumab therapy following chemoradiation compared with a historical cohort treated with chemoradiation alone in patients with stage III non-small cell lung cancer: A real-world multicentre study. Eur J Cancer 142:83–91
Taugner J, Käsmann L, Eze C et al (2021) Durvalumab after chemoradiotherapy for PD-L1 expressing inoperable stage III NSCLC leads to significant improvement of local-regional control and overall survival in the real-world setting. Cancers (Basel) 13(7):1613
Faehling M, Schumann C, Christopoulos P et al (2020) Durvalumab after definitive chemoradiotherapy in locally advanced unresectable non-small cell lung cancer (NSCLC): Real-world data on survival and safety from the German expanded-access program (EAP). Lung Cancer 150:114–122
EPAR (European public assessment report) Imfinzi®.
Girard N, J.M. Smit H, Sibille A et al., et al. PACIFIC-R Real-World Study:
Treatment Duration and Interim Analysis of Progression-Free Survival in Unresectable Stage III NSCLC Patients Treated with Durvalumab After Chemoradiotherapy. Presented at European Lung Cancer Congress 2021 – LBA 1171MO
Brody R, Zhang Y, Ballas M et al (2017) PD-L1 expression in advanced NSCLC: Insights into risk stratification and treatment selection from a systematic literature review. Lung Cancer 112:200–215
Acknowledgements
The authors are grateful to the patients, their families, the physicians who participated in this study and the study project team. Medical writing support and editing was provided by Eltium under the direction of the authors and was funded by Astrazeneca.
Funding
AstraZeneca France.
Author information
Authors and Affiliations
Contributions
All authors whose names appear on the submission. made substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data; or the creation of new software used in the work; drafted the work or revised it critically for important intellectual content; approved the version to be published; and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of interest
Virginie Avrillon: Consultant/Honoraria’s: BMS, Pfizer, Roche; Catherine Daniel: Consultant/ Honoraria’s: Astrazeneca, BMS, Amgen; Pierre Boisselier: Consultant/ Honoraria’s: Astrazeneca, BMS, Merck, MSD; Cécile Le Péchoux: Consultant/Honoraria’s/Grants to institution: Astrazeneca, Roche, Nanobiotix, Eli-Lilly, Medscap, PriMEOncology Amgen; Christos Chouaid: Consultant/ Honoraria’s: BMS, MSD, BI, Roche, AZ, Amgen, Eli-Lilly, Sandoz, Janssen, MundiPhaema, GSK, Sanofi, Bayer, Novartis, Takeda, Pfizer, Research project funding: BMS, BI, Roche, AZ, Amgen, Novartis, Takeda.
Ethical Approval
This early access program cohort was requested by the French Health Authorities (ANSM) and was closely followed by them during the study conduct.
Consent to Participate
All patients have given their consent to participate in this early access program cohort and have received an information note on the collection of their data for this early access program and have given their consent.
Consent for Publication
All patients have received an information note on the publication of the aggregate results and have given their consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Avrillon, V., Daniel, C., Boisselier, P. et al. Nationwide Real-Life Safety and Treatment Exposure Data on Durvalumab After Concurrent Chemoradiotherapy in Unresectable Stage III, Locally Advanced, Non-small Cell Lung Cancer: Analysis of Patients Enrolled in the French Early Access Program. Lung 200, 95–105 (2022). https://doi.org/10.1007/s00408-022-00511-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-022-00511-8