Abstract
Negative symptoms in CHR-P people are generally not responsive to treatments and commonly related to poorer functional outcome. However, less research attention has been dedicated to Persistent Negative Symptoms (PNS), defined as clinically stable negative symptoms of moderate severity evident for at least 6 months. This study aims to (a) determine the prevalence of PNS in a sample of young people at CHR-P; (b) investigate any association of PNS with functioning and clinical features; (c) examine longitudinal course of PNS across 2 years of follow-up and changes in PNS severity levels with specialized treatments. One Hundred Eighty CHR-P participants were recruited and were divided into CHR-P/PNS + and CHR-P/PNS− subgroups. The clinical assessments were based on the PANSS and the GAF and were conducted at baseline and every 12 months during the follow-up. Twenty four participants showed PNS at entry. Of them, 21 concluded the 2-year follow-up period. At baseline, the CHR-P/PNS + participants showed more educational and employment deficits, and more social and functioning impairment. During the follow-up, the CHR-P/PNS + subgroup had a significant longitudinal decrease in negative symptoms, which was specifically related to antidepressant treatment. CHR-P/PNS + subjects also showed a higher incidence of new hospitalization and a lower functional recovery over time. Our findings support that the persistence of negative symptoms in CHR-P people is longitudinally related to worse daily functioning and more severe clinical conditions that are at higher risk of hospitalization and are less responsive to specialized treatments.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
There has been an increasing clinical and research interest in the last few years about Negative Symptoms (NS) in people with First Episode of Psychosis (FEP) and at Clinical High Risk for Psychosis (CHR-P). In this respect, recent evidence in CHR-P populations showed that NS are among the main determinants for psychosis transition [1, 2], as they often appear before other clinical features, are generally not responsive to treatments [3, 4], and correlate with poorer functional outcome [5, 6]. Moreover, continuous, frequent monitoring of NS in people at CHR-P seems also to be justified by another crucial empirical finding, i.e., the initial 6 months of treatment after the enrollment in specialized EIP programs is a key window for improving NS as less improvement is likely afterward [7].
Despite this growing interest in NS in early psychosis, less attention has been dedicated to Persistent Negative Symptoms (PNS), which are usually considered particularly difficult to address and to treat [8]. According to Buchanan (2007) [9], PNS are defined as clinically stable NS of moderate severity evident for an extended period [10]. Specifically, these following criteria are currently required: (a) presence of at least moderate (i.e., a score of ≥ 4 on the PANSS scale) for at least 3 NS or at least moderately severe (i.e., a score of ≥ 5 on the PANSS) for at least 2 NS; (b) persistence of NS for at least 6 months and for an extended period of time prior to the study beginning (e.g., at least 4 weeks); and (c) absence of relevant levels of positive symptoms, depression, and extrapyramidal symptoms as defined thresholds on accepted and validated rating scales [11, 12].
In studies examining the impact of PNS in people with FEP, it was reported that the presence of PNS is specifically related to increased decline in daily functioning [13,14,15,16], poorer quality of life [16], greater cognitive deficits [15], and longer duration of the prodromal phase [17].
Although research on PNS in CHR-P is still limited, increasing evidence recently showed that CHR-P individuals with PNS have poorer premorbid social adaptation compared to those without PNS, together with the presence of deficits in verbal fluency, processing speed, and a greater prevalence of childhood maltreatment (in particular, neglect) [18]. Other interesting investigations also showed more severe impairment in social and role functioning during the follow-ups in the CHR subgroup with PNS in comparison with CHR-P participants without PNS [10, 19]. However, knowledge about PNS in CHR-P individuals and their treatment response remains to be filled [20].
Starting to this background, the aims of this study were to (a) determine the prevalence of PNS in a sample of young people at CHR-P recruited within an Italian “Early Intervention in Psychosis” (EIP) program; (b) investigate any relevant association of PNS with sociodemographic data, functioning, and other relevant clinical features both at baseline and across a 2-year follow-up period; and (c) examine longitudinal course of PNS across the follow-up and any significant relationship of changes in PNS severity levels with the specialized treatment components provided within the EIP program, as well as with specific outcome parameters (e.g., psychosis transition, CHR-P criteria persistence, suicidal thoughts and behavior, new hospitalization, service disengagement, functional and symptomatic remission). We specifically hypothesized that CHR-P participants show significant deficits in functioning and more severe psychopathology, in line with the previous studies, and that PNS are associated with poorer prognosis compared to CHR-P individuals without PNS.
Methods
Sample and setting
CHR-P participants were consecutively recruited within the “Parma At-Risk Mental States” (PARMS) program from January 2016 to December 2021. The PARMS program is a specialized EIP infrastructure diffusely implemented across all adult and adolescent mental health services in the Parma Department of Mental Health (Northern Italy) [21].
Inclusion criteria were as follows: (a) specialist mental healthcare request; (b) age 12–25 years, (c) to meet CHR-P criteria as defined by the “Comprehensive Assessment of At-Risk Mental States” (CAARMS) [22] at the baseline assessment (i.e., Genetic Vulnerability, “Attenuated Psychotic Symptoms” [APS], or “Brief Limited Intermittent Psychotic Symptoms” [BLIPS]).
Exclusion criteria were as follows: (a) previous affective or non-affective psychotic episodes; (b) past exposure to AP medication or current AP intake exceeding 4 weeks in the present episode; (c) known intellectual disability (I.Q. < 70); and (d) neurological or medical disorder with psychiatric manifestations. Previous use of AP drug was considered a proxy for a past psychotic episode, consistently with the original CAARMS criteria for psychosis threshold [23]. A current AP prescription of less than 4 weeks was required to minimize pharmacological interference with baseline psychopathological assessment [24].
All participants and parents (if minors) provided written informed consent for their participation in the study. This research obtained approval from the local ethics committee (AVEN Ethics Committee protocol n. 559/2020/OSS*/AUSLPR) and adhered to the principles outlined in the 1964 Declaration of Helsinki and its later amendments.
Instruments
The psychopathological assessment of this research included the CAARMS, the “Health of the Nation Outcome Scale” (HoNOS) [25], the “Positive and Negative Syndrome Scale” (PANSS) [26], and the “Global Assessment of Functioning” (GAF) scale [27].
The CAARMS is a clinical interview designed to explore various aspects of attenuated psychopathology. Its “Positive Symptoms” subscale was used for defining both CHR-P and psychosis criteria. CAARMS interviews were conducted by trained PARMS team members using the approved Italian version (CAARMS-ITA) [28]. Specifically, PARMS team members were fully trained from the main author of the CAARMS-ITA, who was trained at “Orygen,” the National Centre of Youth Mental Health” in Melbourne (Australia). Regular CAARMS scoring workshops and supervision sessions were implemented to ensure good-to-excellent interrater reliability [29]. Every 12 months in the follow-up, CAARMS interview was re-administered to psychometrically identify psychosis transition and CHR-P criteria persistence.
The HoNOS was developed to evaluate mental health and social functioning of individuals with severe mental illness, including patients with early psychosis [30]. As originally indicated by Wing and colleagues (1999), subscale scores were derived by grouping items into four main domains: (a) “Behavioral Problems,” (b) “Impairment,” (c) “Psychiatric Symptoms,” and (d) “Social Problems” [31]. According to Kortrijk and co-workers [32], we considered a score of ≤ 2 on the HoNOS items 9, 10, and 11 (included in the HoNOS “Social Problems” domain) as index of functional remission.
The PANSS is a widely used clinical interview for assessing psychopathology in psychosis, including young people with early psychosis [19, 33, 34]. PANSS interviews were conducted by trained PARMS team members with a long-term clinical experience (more than 15 years) with patients with early psychosis. Moreover, regular PANSS scoring workshops were repeated across the follow-up to ensure good-to-excellent interrater reliability (Intraclass Correlation Coefficient [ICC] values of > 0.75 for all item and factor scores). As indicated in the meta-analytic model proposed by Shafer and Dazzi [35], after checking its goodness of fit in our CHR-P total sample [11], we considered five principal psychopathological domains: “Disorganization,” “Negative Symptoms,” “Positive Symptoms,” “Resistance/Excitement-Activity,” and “Affect” (“Depression-Anxiety”).
As for the negative dimension, we, however, decided to use the PANSS negative symptom factor recommended by the European Psychiatric Association (EPA) [12], because it has proven to be more coherent with the current conceptualization of negative symptoms [8]. It includes the following 5 PANSS items: N1 “Blunted affect,” N2 “Emotional withdrawal,” N3 “Poor rapport,” N4 “Passive/apathetic social withdrawal,” and N6 “Lack of spontaneity and flow of conversation.” Compared to the EPA configuration, the negative symptom model proposed by Shafer and Dazzi [35] also encompassed PANSS G7 “Motor retardation” and G16 “Active social avoidance” items, which showed to be not really negative symptoms since they were more related to extrapyramidal symptoms, depression, suspiciousness, or social anxiety [12]. Additionally, to support this choice, we conducted a confirmatory factor analysis exploring three competitive models of different PANSS negative symptom factor configurations reported in the current literature on early psychosis (see Supplementary Materials [Table S1] for details). Since the EPA model also showed to have the best fit values in our CHR-P population across the follow-up period, we considered it in the main statistical analysis of the current study. As index of symptomatic remission, we considered a score of ≤ 3 (corresponding to mild severity or less) on the 8 PANSS items indicated by the “Remission in Schizophrenia Working Group’s criteria” [36]. This index was used for defining clinical remission in CHR-P populations along with the absence of persistence of CHR-P criteria at follow-up assessments [37, 38].
In accordance with Buchanan [9], conservative clinical criteria for PNS were used: (a) presence of at least moderate (i.e., score of ≥ 4 on the PANSS) for at least 3 negative symptoms or at least moderately severe (i.e., score of ≥ 5 on the PANSS) for at least 2 negative symptoms; (b) persistence of negative symptoms for at least 6 months and for an extended time period of at least 4 weeks prior to the study beginning; and (c) absence of significant levels of positive symptoms, depression, and extrapyramidal symptoms as defined thresholds on accepted and validated rating scales (i.e., persistent scores of ≤ 3 in all items included in the PANSS “Positive Symptoms” domain and in PANSS “Depression” and “Guilt Feelings” items for at least 6 months in the same time period, together with the absence of extrapyramidal symptoms requiring anticholinergic treatment).
The GAF is a commonly used instrument for evaluating clinical status and socio-occupational functioning in individuals with severe mental disorders, including early psychosis [39]. According to Yang and colleagues [40], we considered a GAF score of > 60 at follow-ups as a current index of functional remission.
Finally, we completed a sociodemographic and clinical chart, including information on employment, “Duration of Untreated Illness” (DUI), new suicide attempt and self-harm behavior, current suicidal ideation, functional recovery, and service disengagement. Specifically, DUI was defined as the time interval between the first psychiatric manifestation and the first pharmacological/psychosocial intervention [41]. Suicide attempt was defined as potentially injurious, self-inflicted behavior without a fatal outcome for which there was (implicit or explicit) evidence of intent to die, while self-harm behavior was intended as acts of deliberate self-harm or intoxication with alcohol or drugs, but where there was no clear intention to die [42]. Service disengagement was defined as a complete lack of contact or untraceable for at least 3 months despite a need of treatment [43]. Current suicidal ideation was detected as a score of ≥ 2 on item 4 (“Suicidality”) of the Brief Psychiatric Rating Scale (BPRS), corresponding at least to occasional suicidal thinking without specific plans [44]. Lastly, as proposed by Silva and Restrepo (2019), functional recovery was simply defined as return to work or school [45].
All assessment tools were administered both at baseline (T0) and every 12 months during the 2-year follow-up period (i.e., at 1- and 2-year assessment time [T1 and T2]).
Procedures
The initial DSM-5 diagnosis was formulated by a minimum of two trained PARMS team members using the “Structured Clinical Interview for DSM-5 mental disorders” (SCID-5) [46]. Based on CAARMS and PANSS interviews, CHR-P participants with PNS were categorized as CHR-P/PNS + subgroup. CHR-P subjects without PNS at entry were included in the CHR-P/PNS− subsample. Specifically, as for the identification of the criterion b of PNS, the “persistence of negative symptoms for at least 6 months” was assessed re-administering the PANSS after 12 months of follow-up. Moreover, information on “the persistence of negative symptoms for an extended time period of at least 4 weeks prior to the study beginning” was collected at baseline from patients’ clinical interviews and/or their medical reports.
Within 3–4 weeks from the PARMS enrollment, CHR-P individuals were assigned to a multi-professional team, including a clinical psychologist, an early rehabilitation case manager, and a psychiatrist. As recommended by current EIP official guidelines [47, 48], AP medication should be prescribed to CHR-P subjects who had (a) a rapid decline in daily functioning, (b) a sudden escalation of overt psychotic symptoms, (c) an immediate risk of suicide or severe violence, or (d) who failed to respond to specialized psychosocial interventions. As first-line pharmacological treatment, low-dose atypical AP drugs were used [49].
Individual psychotherapy was shaped on the model by van der Gaag and co-workers [50] for young people at CHR-P, offering at least 15 sessions per year (each lasting 60 min) [39]. Family psychoeducation was adapted on the model by McFarlane [51] for CHR-P individuals, providing at least 10–12 sessions to each family [52]. Finally, a dedicated case manager was also provided to each subject/family to coordinate all interventions planned, especially those aimed at promoting a recovery-oriented early rehabilitation through at least 24 sessions provided along the 2 years of follow-up (each lasting 60 min) [53].
Information regarding medication (type and dosage), intensity of psychosocial interventions, psychopathology, functioning, and outcome parameters (e.g., service disengagement, psychosis transition, new hospitalization, etc.) was collected both at baseline and each 12 months during the 2 years of follow-up.
Between-group comparisons on sociodemographic, clinical, and treatment measures at baseline were first investigated. Longitudinal changes in PNS severity levels along the 2-year follow-up period was also examined in the CHR-P/PNS + subsample, as well as any relevant association of PNS changes with psychopathological variables and treatment response over time. Lastly, intergroup comparisons on treatment response and specific outcome parameters were investigated.
Statistical analysis
The data analysis was conducted using the Statistical Package for Social Science (SPSS), version 15.0 for Windows [54]. All tests were two-tailed, with a p value significance set at 0.05. In intergroup comparisons, categorical variables were examined using the Chi-square (X2) test, while continuous variables using the Mann–Whitney U test.
Spearman’s rank correlation coefficients were calculated to investigate any significant associations of PANSS “Negative Symptoms” factor scores with sociodemographic and clinical parameters both at baseline and along the 2-year follow-up period (T2) in the CHR-P/PNS + subsample. The Wilcoxon test for repeated measures was also carried out to assess the longitudinal stability of PANSS NS factor scores in the CHR-P/PNS + subgroup across the 2 years of our follow-up.
Additionally, multiple linear regression analyses with PANSS NS factor scores as dependent variables and the specialized PARMS intervention components as independent variables were performed in the CHR-P/PNS + subsample. In our longitudinal examinations, we specifically used the differences (deltas [Δ]) between T0 and T1 or T2 PANSS, GAF, and HoNOS scores as primary clinical parameters to examine overtime. Indeed, according to Ver Hoef [55], the delta scores better describe longitudinal changes and temporal dynamics of psychopathology and the clinical status compared to T0, T1, and T2 single scores.
Furthermore, a mixed-design ANOVA was also carried out to investigate between-group comparisons on longitudinal treatment response for NS. Finally, as for time-to-event outcome parameters (i.e., psychosis transition, new hospitalization, new suicide attempt, new self-harm behavior, and service disengagement), after having previously checked that the proportionality-of-hazards assumption was met, univariate models were fitted for each outcome variable across the 2 years of follow-up using Cox regression analysis [56]. For not time-to-event dependent parameters (such as CHR-P criteria persistence, current suicidal ideation, functional recovery, GAF or HoNOS functional remission, PANSS symptomatic remission, and persistent negative symptoms), binary logistic regression analyses with PNS subgroups as independent variables were performed [57].
Results
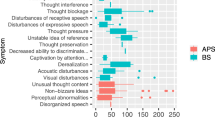
One hundred and eighty CHR-P patients were recruited for this study. Of them, 140 (77.8%) met APS criteria and 30 (16.7%) met BLIPS criteria (Table 1). Twenty-four (13.3%) CHR-P participants showed PNS at entry, with different DSM-5 diagnoses and symptoms (see Supplementary Materials [Figure S1] for details).
Baseline data
As for sociodemographic features, CHR-P/PNS + individuals had a higher prevalence rate of NEET (i.e., participants “Not [engaged] in Education, Employment, or Training”) in comparison with the CHR-P/PNS− group (Table 1).
Compared to CHR-P/PNS−, CHR-P/PNS + subjects showed higher PANSS “Negative Symptoms” and “Disorganization” dimension subscores, as well as higher PANSS total score. They also had a lower GAF score and a higher HoNOS “Social Problems” domain subscore. In the CHR-P/PNS + subgroup at entry (T0), no statistically significant correlations between severity levels in negative symptoms and other clinical and sociodemographic characteristics were found (Table 2).
Longitudinal data in the CHR-P/PNS + group
All 24 CHR-P/PNS + participants reached the 1-year assessment time (T1), while only 21 concluded the 2-year follow-up period (T2) (see Supplementary Materials [Figure S2] for details).
During the 2 years of follow-up, CHR-P/PNS + individuals showed a significant decrease in negative symptom severity levels. As no relevant longitudinal change in negative symptom severity was found during the second year of intervention but exclusively in the first 12 months, this reduction seemed to be primarily attributable to the first year of treatment (i.e., from T0 to T1) (Table 3).
Moreover, CHR-P/PNS + participants had significant positive correlations of the difference (delta) in T0–T2 PANSS “Negative Symptoms” factor subscores with deltas in T0–T2 PANSS “Disorganization” factor subscores and T1 equivalent dose of fluoxetine, as well as a relevant negative correlation with level of education (in years) (Table 4).
In the CHR-P/PNS + group, our linear regression analysis results exclusively showed that the delta in T0–T1 PANSS “Negative Symptoms” factor subscores was significantly predicted by T1 equivalent dose of fluoxetine (Table 5). No other predictive factor for longitudinal changes in negative symptom severity levels was observed.
Our mixed-design ANOVA results confirmed a significant time effect in the CHR-P total sample (“within-subject” effect) with a relevant longitudinal decrease in PANSS “Negative Symptoms” factor subscore (Table 6). However, we found a statistically significant group effect (“between-group” effect) in favor CHR-P/PNS− individuals. Specifically, the detailed examination of estimated marginal means showed significantly higher scores in negative symptom within the CHR/PNS + subgroup compared to the CHR/PNS + one along the entire follow-up (see Supplementary Materials [Figure S3] for profile plots of the two CHR-P subgroups).
As for outcome parameters, at both T1 and T2 assessment, CHR-P/PNS + participants showed higher rates of new hospitalization compared to the CHR-P/PNS− subgroup (Table 7), as well as lower rates of GAF and HoNOS functional remissions (Table 8). Moreover, exclusively at T1, they also showed lower incidence rates of current suicidal ideation and functional recovery.
Discussion
The results of this investigation showed that the baseline prevalence of PNS in the PARMS program was about 13%. This finding is slightly higher than what was reported in similar CHR-P samples, ranging from 6 to 9% [10, 18]. The difference in PNS prevalence may be related to different criteria for defining clinical persistence of negative features and/or different assessment instruments for measuring negative symptoms across studies. Specifically, the “North American Prodrome Longitudinal Study-2” (NAPLS-2) applied a PNS definition exclusively based on having one of the three main negative symptoms of the “Scale of Psychosis-risk Symptoms” (i.e., social anhedonia, avolition, and expression of emotion) scored ≥ 4 (i.e., moderately severe to extreme) for a duration of 1 year [19]. Differently, at the PACE (“Personal Assessment and Clinical Evaluation”) Clinic in Melbourne (Australia) [18], the authors defined PNS as presence of at least one global “Negative symptoms” subscale score (measured with the “Scale for the Assessment of Negative Symptoms” [SANS]) of ≥ 3 at baseline and at follow-up, a combined total score of 6 or less on the BPRS (“Brief Psychiatric Rating Scale”) subscales of depression, guilt, and suicidality (corresponding to an average of “very mild” or less on each item), and a combined total score of 16 or less on the BPRS psychotic subscales of conceptual disorganization, hallucinations, suspiciousness, and unusual thought content (corresponding to an average of “moderate” or less on each item), without using any measure of extrapyramidal symptoms.
As for assessment instruments for measuring negative symptoms, it is important to specify that the PANSS is not strictly in line with the current conceptualization of negative symptoms [12]. Specifically, it includes few negative characteristics and only focuses on the patient’s behavior, failing to evaluate the subject’s internal experience. Moreover, this instrument was developed to be mainly used for adult individuals with full-blown psychotic features, and is probably not enough sensitive to detect the subtle manifestations of negative symptoms in CHR-P individuals. In this respect, second generation scales (such as the “Brief Negative Symptom Scale” [BNSS]) [58] are more in line with current conceptualizations of negative symptoms and have versions more adapted to the intended population. Therefore, future research using second generation scale to investigate PNS is needed.
However, given the high diffusion of the PANSS in clinical populations with early psychosis, our results have the potential to be replicated in similar populations, and this is of great clinical relevance since investigations examining beneficial effects of specialized interventions on PNS are still relative poor. Furthermore, it is important to consider that assessment of PANSS G6 “Depression” and G3 “Guilt feelings” items is not completely adequate to exclude the presence of significant depressive symptoms, as the absence of anticholinergic treatment is not enough to exclude the presence of Parkinsonism. In this respect, both the original definition of PNS [9] and the EPA [12] recommended the use of accepted and validated rating scales, such as the “Calgary Depression Scale” or the “Hamilton Depression Rating Scale” to assess depressive features, and the “Simpson-Angus Extrapyramidal Rating Scale” or the “Extrapyramidal Symptom Rating Scale” to evaluate extrapyramidal symptoms.
In line with what was observed by Yung et al. [18] in the NAPLS-2 population, our CHR-P/PNS + participants showed more educational and employment deficit at baseline compared to the CHR-P/PNS− group, as well as more social and functioning impairment. These findings confirm evidence on PNS in patients with first episode psychosis, presenting statistically significant associations with greater unemployment rates and poorer executive functioning at entry [13, 59]. Moreover, this impairment in daily functioning seemed to be stable over time. Indeed, compared to CHR-P/PNS−, CHR-P/PNS + participants showed lower GAF scores and higher HoNOS “Social Problems” domain subscores also at both T1 and T2 assessment (see Supplementary Materials [Table S2] for details). In this sense, PNS remain a relevant, enduring “stigma” for a satisfying functional and social recovery in this young population [11].
In comparison with CHR-P/PNS−, CHR-P/PNS + individuals also had more severe levels of psychopathology at entry, especially in terms of higher PANSS scores in negative symptoms and disorganization. Additionally, this group-specific greater clinical severity appeared to be persistent over time as confirmed at both T1 and T2 assessment (see Supplementary Materials [Table S2] for details). Overall, these findings confirm that PNS may be considered as enduring psychopathological indicators of clinical severity and poorer clinical recovery.
Across the 2 years of our follow-up, the CHR-P/PNS + subgroup showed a relevant longitudinal decrease in negative symptom severity levels (especially during the first year of intervention). This was specifically correlated with lower educational levels, higher equivalent dose of fluoxetine at T1, and a longitudinal improvement in disorganization (as well as in T0–T2 delta PANSS total scores calculated without negative symptom item subscores [ρ = 0.600; p = 0.009]). These findings confirm a general poor school performance and a privileged psychopathological relationship between negative symptoms and disorganization, in line with the Bleuler’s thinking on “basic” symptoms in schizophrenia [60]. This special link could thus be established before full-blown psychotic symptoms emerge and already characterize the prodromal phase of psychosis, perhaps as direct, early features of a psychotic vulnerability [61].
The results of our multiple linear regression analysis also showed that the longitudinal decrease in PNS severity (specifically in the first year of treatment) was significantly predicted by T1 antidepressant dosage. No association with antipsychotic medication or other psychosocial interventions was found. Moreover, when considering the two-factor structure of negative symptoms described in the literature (i.e., “Expressive” and “Experiential” subdomains) using the PANSS model proposed by Jang and co-workers [62] (i.e., “Expressive” factor including PANSS N1, N3, N6, and G7 items, and “Experiential” factor including PANSS N2, N4, and G16 items), our regression analysis results showed that only longitudinal severity decrease in “Expressive” subdimension (especially during the first year of intervention) was significantly predicted by T1 antidepressant dosage (B = 0.012; p = 0.017), together with a higher session number of family psychoeducation (B = 0.558; p = 0.043) (see Supplementary Materials [Table S3] for details).
Unfortunately, the beneficial effect related to the use of antidepressant drug did not extent into the second year of treatment, and seems to be particularly helpful to improve the diminished expression domain (specifically including blunted affect and alogia) [63], together with the provision of family psychoeducation. This finding seems to be an isolated result in the current literature, especially when independent from improvement in depressive features. Indeed, there is only evidence that a combined therapy with antidepressant in addiction to antipsychotic medication showed beneficial effects on negative symptoms in people with schizophrenia [64, 65]. In this respect, recent research has confirmed that different antidepressants among “Selective Serotonin Re-uptake Inhibitors” (SSRIs) could have different effects [66]. Therefore, we hope to conduct clinical research with more participants in the near future, looking forward to analysis and clinical application of the differences in these pharmacological effects. As for how antidepressant dosage at T1 assessment can predict a negative symptom change between T0 and T1, it is probably related to the pharmacological treatment maintenance across the 1-year follow-up period. Indeed, all CHR-P/PNS + participants who were prescribed with antidepressant drug at baseline, continued to take this medication steadily after one year of intervention (see Supplementary Materials [Table S2] for details).
However, as there were multiple regression models conducted for the three time periods but fluoxetine dosage was only significant in T0–T1 period, this effect seems to be poorly robust and requires further confirmation in both larger CHR-P/PNS + populations and longer prospective research. Moreover, although we excluded clinically relevant severity levels of PANSS “Depression” and “Guilt feelings” scores from the PNS definition, the beneficial effect of fluoxetine in the first year of treatment may also affect other potential secondary negative symptoms (such as blunted affect and avolition). Therefore, future studies using more specific assessment instrument for clinical depression in psychosis are needed. Anyway, our result on fluoxetine effects remarks the importance of an early recognition and a timely intervention focused on expressed negative symptoms.
In this respect, few investigations were prospectively designed to evaluate the effect of pharmacological treatment on PNS, with most reports of decrease in negative symptoms being based on short-term research [67, 68]. Meta-analytic results on randomized, controlled antipsychotic drug trials in patients with schizophrenia reported that low-dose amisulpride (approved for negative symptoms in some European countries) was the only antipsychotic that was superior to placebo in the treatment of predominant negative symptoms. However, a parallel improvement of depression was also observed, making it difficult to evaluate whether the decrease in negative symptoms was due to reduction in clinical depression. Furthermore, a more recent prospective (26 weeks), randomized controlled trial showed that significant differences and clinically relevant improvement in negative symptoms were demonstrated in favor of cariprazine over risperidone [69]. In this study, changes in symptoms from other domains were small, indicating that improvement was specific to negative symptoms and not related to changes in positive, extrapyramidal, and depressive symptoms. Given the considerable unmet medical need associated with negative symptoms [70], drug development is active in this therapeutic area for agents with activity at different receptor systems including NMDA receptors, alpha 7 nicotinic receptors, and 5-HT2A and sigma-2 receptors [71].
Given the limited effective pharmacologic treatments to treat negative symptoms, mental health professionals should be aware of psychosocial interventions that can be used together with antipsychotic medication. In line with our finding on the beneficial effect of family psychoeducation on the expressive subdomain of negative symptoms, some authors suggested that interventions aimed at addressing attitudes, behaviors, and poor psychosocial functioning (including a focus on healthy lifestyles, with emphasis on exercise, sleep, diet, smoking cessation, appropriate alcohol consumption, and social participation) may help individuals gain insight into how their symptoms affect their outlook [72]. In this respect, while social skills training and cognitive remediation therapy showed some evidence for negative symptom improvement [73, 74], the most widely studied psychological intervention is Cognitive-Behavioral Therapy (CBT). Specifically, CBT demonstrated a positive (but moderate) effect on negative symptoms, with a reduction of apathy and improved motivation [75], as well as it seems to support awareness of the link between a patient’s thoughts, behaviors, and feelings in an effort to change symptoms and functioning [4]. Finally, family interventions may help patients and family members cope with the burden of negative symptoms through psychoeducation, communication training, behavioral problem solving, and crisis management [76]. Despite mixed and inconsistent findings, referring patients to psychosocial intervention could be an important way for mental health professionals to support patients and their families as they cope with negative symptoms and attempt to improve outcomes and quality of life [33]. However, future, prospective clinical trials are needed to improve the evidence base for psychosocial interventions in negative symptoms of psychosis (especially in PNS).
Comparing the two CHR/P subgroups, our mixed-design ANOVA results showed a statistically relevant “between-group effect” within global evidence of a significant “time effect” (“within-subjects effect”) in PANSS “Negative Symptoms” score decrease over time (especially in the first year of intervention) (see Supplementary Materials [Table S4] for details). As hypothesized, CHR/PNS− participants showed lower negative symptom severity levels across the entire follow-up period in comparison with the CHR-P/PNS + subgroup, suggesting that the persistence of negative symptoms in CHR-P individuals may negatively interfere not only with the severity of clinical course over time but also with treatment response on negative dimension. In this respect, the longitudinal improvement in negative symptom severity levels found in our CHR-P/PNS− subgroup could also be due to higher beneficial effects of PARMS interventions on decrease in secondary negative symptoms (especially those related to positive symptoms, suspiciousness, and clinical depression).
As for outcome parameters, CHR-P/PNS + subjects showed higher incidence rates of new hospitalization, lower functional remission, and lower functional recovery compared to CHR-P/PNS− ones. All these findings specifically support that the persistence of negative symptoms in people at CHR-P is longitudinally related to worse daily functioning, poorer real-world-performance, and more severe clinical conditions that are at higher risk of hospital admission and are less responsive to the specialized treatments usually provided within EIP services.
Strengths and limitations
This investigation notably examined PNS (defined using conservative clinical criteria) in a clinical sample of young individuals at CHR-P, (i.e., without evident full-blown psychotic features) treated within an EIP service across a 2-year follow-up period. This gave us the opportunity to strictly monitor the progression of negative symptoms over time and to verify their clinical persistence and their treatment response.
However, some limitations should also be considered. A first weakness of our research was related to the small sample size (especially for the CHR-P/PNS + subgroup). This did not allow a reliable generalization of our results and requires replication in future perspective studies on larger CHR-P populations.
Second, although conservative clinical criteria were used [9], the original definition of PNS would require the use of accepted and validated rating scales to evaluate depression and extrapyramidal symptoms (such as the “Calgary Depression Scale” or the “Hamilton Depression Rating Scale,” and the “Simpson-Angus Extrapyramidal Rating Scale” or the “Extrapyramidal Symptom Rating Scale”). Therefore, our assessment of PANSS “Depression” and “Guilt feelings” items may not have been enough to exclude the presence of depressive symptoms, as the absence of anticholinergic treatment could be insufficient to exclude the presence of Parkinsonism. Future research using more specific assessment instruments for extrapyramidal and depressive symptoms is thus needed.
Third, as there was a general reduction in intensity of psychosocial intervention sessions offered within the PARMS program during the second year of treatment [77], this may have contributed to worsening longitudinal outcome, independently to the greater, intrinsic treatment-resistant nature of PNS and their related poorer care response [78]. Thus, maintaining an adequate intensity of EIP treatments also during the second/third year of intervention could consolidate the longitudinal decrease of PNS and more successfully promote functional recovery of this young population. However, in order to more strongly support that the reported longitudinal improvement in PNS severity levels is due to the effect of our specialized treatments, future research with control comparisons is needed. In this respect, other comparable CHR observation studies without interventions found that similar symptom improvements could be seen within the first 6–12 months [79].
Fourth, we examined CHR-P subjects in a “real-world” clinical setting primarily involved in the delivery of evidence-based care pathways within standard community mental healthcare centers. Therefore, our findings should be compared only with similar clinical populations.
Moreover, the current investigation was conducted within a specialized CHR-P program not specifically focused on the evaluation of negative symptoms. In particular, negative symptoms were assessed with the PANSS, a psychometric scale commonly used also in CHR-P populations [2, 11, 80, 81], but poorly articulated and composed of only seven negative symptom items. Therefore, future research using more specific, second generation instruments for negative symptoms (such as the “Brief Negative Symptom Scale” [BNSS] or the “Clinical Assessment Interview for Negative Symptoms” [CAINS]) [58, 82] that were developed to rectify shortcomings of older scales, is needed. Moreover, it is well known that the PANSS was not developed to address negative symptoms in CHR individuals. In this respect, specific scales (such as the “Negative Symptom Inventory” [NSI]) [83] have been developed specifically for the CHR-P population and should be used in future investigations. However, given the high diffusion of the PANSS in populations with early psychosis, our results have the potential to be replicated in similar populations, and this is of great clinical relevance since investigations examining beneficial effects of specialized interventions on PNS are still relative poor.
Additionally, although widely used in CHR-P individuals, also the GAF conflates symptoms and functioning, and cannot be considered an optimal measure of daily functioning, especially in terms of real-world socio-occupational performance. Therefore, future research using more specific instruments to assess social and occupational functioning (such as the “Global functioning social and role scales”) [84] is needed. As for providing more valid statistical results on the association between these potentially overlapping constructs, we also conducted further correlation analyses with the GAF using the EPA negative symptom factor model [85]. However, no statistically significant correlations between baseline GAF score and baseline PANSS “Negative Symptoms” factor subscore suggested by the EPA was observed (ρ = − .179; p = .599), as well as between T0–T2 delta in GAF scores and T0–T2 delta in PANSS “Negative Symptoms” factor subscores (ρ = − .312; p = .380).
Finally, in comparing our results with both existing and future empirical evidence, it is necessary to take into account the PNS criteria used to dichotomize CHR-P individuals in the present research. Indeed, different PNS criteria may create CHR-P subgroups with poorly comparable psychopathological characteristics.
Conclusions
PNS are clinically significant also in a minority of young people at CHR-P treated within specialized EIP programs. Although the results of this investigation show a relevant association between PNS and poorer functional outcomes in young people at CHR-P during a 2-year follow-up period, a longitudinal decrease over time was also observed. Specifically, this reduction seems to be related to specific EIP treatments (such as the T1 antidepressant dosage). In this respect, future research should aim to improve the early identification of CHR people and focus on PNS in a larger portion of population at risk, with a special attention to intensive treatments and rehabilitation programs during the follow-up period.
Data availability
The data that support the findings of this investigation are available from the corresponding author upon reasonable request. The data are not publicly available due to privacy/ethical restrictions.
References
Valmaggia LR, Stahl D, Yung AR, Nelson B, Fusar-Poli P, McGorry PD, McGuire PK (2013) Negative psychotic symptoms and impaired role functioning predict transition outcomes in the at-risk mental state: a latent class cluster analysis study. Psychol Med 43:2311–2325. https://doi.org/10.1017/S0033291713000251
Lim K, Rapisarda A, Keefe RSE, Lee J (2022) Social skills, negative symptoms and real-world functioning in individuals at ultra-high risk of psychosis. Asian J Psychiatr 69:102996. https://doi.org/10.1016/j.ajp.2021.102996
Millan MJ, Fone K, Steckler T, Horan WP (2014) Negative symptoms of schizophrenia: clinical characteristics, pathophysiological substrates, experimental models and prospects for improved treatment. Eur Neuropsychopharmacol 24:645–692. https://doi.org/10.1016/j.euroneuro.2014.03.008
Aleman A, Lincoln T, Bruggeman R, Melle I, Arends J, Arango C, Knegtering H (2017) Treatment of negative symptoms: where do we stand, and where do we go? Schizophr Res 186:55–62. https://doi.org/10.1016/j.schres.2016.05.015
Lin A, Wood SJ, Nelson B, Brewer WJ, Spiliotacopoulos D, Bruxner A, Broussard C, Pantelis C, Yung AR (2011) Neurocognitive predictors of functional outcome two to 13 years after identification as ultra-high risk for psychosis. Schizophr Res 132:1–7. https://doi.org/10.1016/j.schres.2011.06.014
Yung AR, Cotter J, Wood SJ, McGorry P, Thompson AD, Nelson B, Lin A (2015) Childhood maltreatment and transition to psychotic disorder independently predict long term functioning in young people at ultra high risk for psychosis. Psychol Med 45:3453–3465. https://doi.org/10.1017/s003329171500135x
Tran T, Spilka MJ, Raugh IM, Strauss GP, Bearden CE, Cadenhead KS, Cannon TD, Cornblatt BA, Keshavan M, Mathalon DH, McGlashan TH, Perkins DO, Seidman LJ, Stone WS, Tsuang MT, Walker EF, Woods SW, Addington JM (2023) Negative symptom trajectories in individuals at clinical high risk for psychosis: differences based on deficit syndrome, persistence, and transition status. Schizophr Bull Open 4:sgad014. https://doi.org/10.1093/schizbullopen/sgad014
Sauvé G, Brodeur MB, Shah JL, Lepage M (2019) The prevalence of negative symptoms across the stages of the psychosis continuum. Harv Rev Psychiatry 27:15–32. https://doi.org/10.1097/HRP.0000000000000184
Buchanan RW (2007) Persistent negative symptoms in schizophrenia: an overview. Schizophr Bull 33:1013–1022. https://doi.org/10.1093/schbul/sbl057
Devoe LDJ, Lu TD, Cannon KS, Cadenhead BA, Cornblatt TH, McGlashan DO, Perkins LJ, Seidman MT, Tsuang SW, Woods EF, Walker DH, Mathalon CE, Bearden J, Addington J (2021) Persistent negative symptoms in youth at clinical high risk for psychosis: a longitudinal study. Schizophr Res 227:28–37. https://doi.org/10.1016/j.schres.2020.04.004
Pelizza L, Leuci E, Quattrone E, Azzali S, Paulillo G, Pupo S, Pellegrini P, Menchetti M (2023) Longitudinal evaluation on negative symptoms in young people at ultra-high risk (UHR) of psychosis: results from a 2-year follow-up study in a real-world care setting. Eur Arch Psychiatry Clin Neurosci. https://doi.org/10.1007/s00406-023-01600-2
Galderisi S, Mucci A, Dollfus S, Nordentoft M, Falkai P, Kaiser S, Giordano GM, Vandevelde A, Nielsen MØ, Glenthøj LB, Sabé M, Pezzella P, Bitter I, Gaebel W (2021) EPA guidance on assessment of negative symptoms in schizophrenia. Eur Psychiatry 64(1):e23. https://doi.org/10.1192/j.eurpsy.2021.11
Chang W, Hui CL, Tang JY, Wong GH, Lam MM, Chan SK, Chen EY (2011) Persistent negative symptoms in first-episode schizophrenia: a prospective three-year follow-up study. Schizophr Res 133:22–28. https://doi.org/10.1016/j.schres.2011.09.006
Galderisi S, Mucci A, Bitter I, Libiger J, Bucci P, Fleischhacker W, Kahn RS (2013) Persistent negative symptoms in first episode patients with schizophrenia: results from the European first episode schizophrenia trial. Eur Neuropsychopharmacol 23:196–204. https://doi.org/10.1016/j.euroneuro.2012.04.019
Hovington CL, Bodnar M, Joober R, Malla AK, Lepage M (2012) Identifying persistent negative symptoms in first episode psychosis. BMC Psychiatry 12:224. https://doi.org/10.1186/1471-244x-12-224
Puig O, Baeza I, de la Serna E, Cabrera B, Mezquida G, Bioque M, Lobo A, Gonzalez-Pinto A, Parellada M, Corripio I, Vieta E, Bobes J, Usall J, Contreras F, Cuesta MJ, Bernardo M, Castro-Fornieles J (2017) Persistent negative symptoms in first-episode psychosis: early cognitive and social functioning correlates and differences between early and adult onset. J Clin Psychiatry 78:1414–1422. https://doi.org/10.4088/jcp.16m11122
Edwards J, McGorry PD, Waddell FM, Harrigan SM (1999) Enduring negative symptoms in first-episode psychosis: comparison of six methods using follow-up data. Schizophr Res 40:147–158. https://doi.org/10.1016/s0920-9964(99)00043-2
Yung AR, Nelson B, McGorry PD, Wood SJ, Lin A (2018) Persistent negative symptoms in individuals at ultra high risk for psychosis. Schizophr Res 206:355–361. https://doi.org/10.1016/j.schres.2018.10.019
Devoe DJ, Lu L, Cannon TD, Cadenhead KS, Cornblatt BA, Keshavan M, McGlashan TH, Perkins DO, Seidman LJ, Stone WS, Tsuang MT, Woods SW, Walker EF, Mathalon DH, Bearden CE, Addington J (2023) The impact of early factors on persistent negative symptoms in youth at clinical high risk for psychosis. Front Psychiatry 14:1125168. https://doi.org/10.3389/fpsyt.2023.1125168
Pelizza L, Leuci E, Maestri D, Quattrone E, Azzali S, Paulillo G, Pellegrini P (2022) Longitudinal persistence of negative symptoms in young individuals with first episode schizophrenia: a 24-month multi-modal program follow-up. Nord J Psychiatry 76:530–538. https://doi.org/10.1080/08039488.2021.2015431
Pelizza L, Leuci E, Quattrone E, Paulillo G, Pellegrini P (2023) The “parma at-risk mental states” (PARMS) program: general description and process analysis after 5 years of clinical activity. Early Interv Psychiatry 17:625–635. https://doi.org/10.1111/eip.13399
Yung AR, Yuen HP, McGorry PD, Phillips LJ, Kelly D, Dell’Olio M, Francey SM, Cosgrave EM, Killackey E, Stanford C, Godfrey K, Buckby J (2005) Mapping the onset of psychosis: the comprehensive assessment of at-risk mental states. Aust N Z J Psychiatry 39:964–971. https://doi.org/10.1080/j.1440-1614.2005.01714.x
Pelizza L, Azzali S, Paterlini F, Garlassi S, Scazza I, Chiri LR, Poletti M, Pupo S, Raballo A (2021) Anhedonia in the psychosis risk syndrome: state and trait characteristics. Psychiatr Danub 33:36–47. https://doi.org/10.24869/psyd.2021.36
Pelizza L, Poletti M, Azzali S, Garlassi S, Scazza I, Paterlini F, Chiri LR, Pupo S, Raballo A (2021) Subjective experience of social cognition in young people at ultra-high risk of psychosis: a 2-year longitudinal study. Nord J Psychiatry 75:97–108. https://doi.org/10.1080/08039488.2020.1799430
Wing JK, Beevor AS, Curtis RH, Park SB, Hadden S, Burns A (1998) Health of the nation outcome scales (HoNOS): research and development. Br J Psychiatry 172:11–18. https://doi.org/10.1192/bjp.172.1.11
Kay SR, Fiszbein A, Opler LA (1987) The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull 13:261–276. https://doi.org/10.1093/schbul/13.2.261
American Psychiatric Association (APA) (2013) Diagnostic and statistical manual of mental disorders, 5th edition (DSM-5). APA Press, Arlington
Raballo A, Semrov E, Bonner Y, Simmons M (2013) Traduzione e adattamento della CAARMS (comprehensive assessment of at-risk mental states). Centro Stampa della Regione Emilia-Romagna, Bologna
Pelizza L, Paterlini F, Azzali S, Garlassi S, Scazza I, Pupo S, Simmons M, Nelson B, Raballo A (2019) The approved Italian version of the comprehensive assessment of at-risk mental states (CAARMS-ITA): field test and psychometric features. Early Interv Psychiatry 13:810–817. https://doi.org/10.1111/eip.12669
Belvederi Murri M, Ferrara M, Imbesi M, Leuci E, Marchi M, Musella V, Natali A, Neri A, Ragni S, Saponaro A, Tarricone I, Tullini A, Starace F, Early Psychosis Working Group (2022) A public early intervention approach to first-episode psychosis: treated incidence over 7 years in the Emilia-Romagna region. Early Interv Psychiatry 17:724–736. https://doi.org/10.1111/eip.13437
Wing J, Curtis RH, Beevor A (1999) Health of the nation outcome scales (HoNOS): glossary for HoNOS score sheet. Br J Psychiatry 174:432–434. https://doi.org/10.1192/bjp.174.5.432
Kortrijk HE, Mulder CL, Drukker M, Wiersma D, Duivenvoorden HJ (2012) Duration of assertive community treatment and the interpretation of routine outcome data. Aust N Z J Psychiatry 46:240–248. https://doi.org/10.1177/0004867411433214
Leuci E, Pelizza L, Landi G, Quattrone E, Maestri D, Azzali S, Pelosi A, Ceroni P, Soncini C, Daolio MC, Paulillo G, Raballo A, Pellegrini P (2022) Personal health budget in patients with first episode psychosis: a new rehabilitation model based on a community care system in Italy. Early Interv Psychiatry 16:221–230. https://doi.org/10.1111/eip.13145
Scazza I, Pelizza L, Azzali S, Garlassi S, Paterlini F, Chiri LR, Poletti M, Pupo S, Raballo A (2022) Aberrant salience in first-episode psychosis: Longitudinal stability and treatment-response. Early Interv Psychiatry 16:912–919. https://doi.org/10.1111/eip.13243
Shafer A, Dazzi F (2019) Meta-analysis of the positive and negative syndrome scale (PANSS) factor structure. J Psychiatr Res 115:113–120. https://doi.org/10.1016/j.jpsychires.2019.05.008
Andreasen NC, Carpenter WT Jr, Kane JM, Lasser RA, Marder SR, Weinberger DR (2005) Remission in schizophrenia: proposed criteria and rationale for consensus. Am J Psychiatry 162:441–449. https://doi.org/10.1176/appi.ajp.162.3.441
Zhang TH, Li HJ, Woodberry KA, Xu LH, Tang YY, Guo Q, Cui HR, Liu XH, Chow A, Li CB, Jiang KD, Xiao ZP, Seidman LJ, Wang JJ (2017) Two-year follow-up of a Chinese sample at clinical high risk for psychosis: timeline of symptoms, help-seeking and conversion. Epidemiol Psychiatr Sci 26:287–298. https://doi.org/10.1017/S2045796016000184
Pelizza L, Leuci E, Quattrone E, Azzali S, Pupo S, Paulillo G, Menchetti M, Pellegrini P (2023) Adverse outcome analysis in people at clinical high risk for psychosis: results from a 2-year Italian follow-up study. Soc Psychiatry Psychiatr Epidemiol. https://doi.org/10.1007/s00127-023-02597-8
Azzali S, Pelizza L, Scazza I, Paterlini F, Garlassi S, Chiri LR, Poletti M, Pupo S, Raballo A (2022) Examining subjective experience of aberrant salience in young individuals at ultra-high risk (UHR) of psychosis: a 1-year longitudinal study. Schizophr Res 241:52–58. https://doi.org/10.1016/j.schres.2021.12.025
Yang H, Gao S, Li J, Yu H, Xu J, Lin C, Yang H, Teng C, Ma H, Zhang N (2022) Remission of symptoms is not equal to functional recovery: psychosocial functioning impairment in major depression. Front Psychiatry 13:915689. https://doi.org/10.3389/fpsyt.2022.915689
Schultze-Lutter F, Rahman J, Ruhrmann S, Michel C, Schimmelmann BG, Maier W, Klosterkötter J (2015) Duration of unspecific prodromal and clinical high risk states, and early help-seeking in first-admission psychosis patients. Soc Psychiatry Psychiatr Epidemiol 50:1831–1841. https://doi.org/10.1007/s00127-015-1093-3
Silverman MM, Berman AL, Sanddal ND, O’carroll PW, Joiner TE (2007) Rebuilding the tower of Babel: a revised nomenclature for the study of suicide and suicidal behaviors—part 2: suicide-related ideations, communications, and behaviors. Suicide Life Threat Behav 37:264–277. https://doi.org/10.1521/suli.2007.37.3.264
Robson E, Greenwood K (2022) Rates and predictors of disengagement and strength of engagement for people with a first episode of psychosis using early intervention services: a systematic review of predictors and meta-analysis of disengagement rates. Schizophr Bull Open 3:sgac012. https://doi.org/10.1093/schizbullopen/sgac012
Pelizza L, Maestri D, Leuci E, Quattrone E, Azzali S, Paulillo G, Pellegrini P (2022) Individual psychotherapy can reduce suicidal ideation in first episode psychosis: further findings from the 2-year follow-up of the “Parma Early Psychosis” programme. Clin Psychol Psychother 29:982–989. https://doi.org/10.1002/cpp.2678
Silva MA, Restrepo D (2019) Functional recovery in schizophrenia. Rev Colomb Psiquiatr 48:252–260. https://doi.org/10.1016/j.rcp.2017.08.004
First MB, Williams JBW, Karg RS, Spitzer RL (2016) SCID-5-CV. Structured clinical interview for DSM-5 mental disorders, Clinical Version. American Psychiatric Association Publishing, Washington DC
Schmidt SJ, Schultze-Lutter F, Schimmelmann BG, Maric NP, Salokangas RK, Riecher-Rössler A, van der Gaag M, Meneghelli A, Nordentoft M, Marshall M, Morrison A, Raballo A, Klosterkötter J, Ruhrmann S (2015) EPA guidance on the early intervention in clinical high risk states of psychoses. Eur Psychiatry 30:388–404. https://doi.org/10.1016/j.eurpsy.2015.01.013
Regione Emilia-Romagna (RER) (2023) Raccomandazioni regionali per la promozione della salute e del benessere in persone all’esordio psicotico, seconda edizione. Centro Stampa della Regione Emilia-Romagna, Bologna.
Poletti M, Azzali S, Paterlini F, Garlassi S, Scazza I, Chiri LR, Pupo S, Raballo A, Pelizza L (2021) Familiarity for serious mental illness in help-seeking adolescents at clinical high risk of psychosis. Front Psychiatry 11:552282. https://doi.org/10.3389/fpsyt.2020.552282
van der Gaag M, Nieman DH, Rietdijk J, Dragt S, Ising HK, Klaassen RM, Koeter M, Cuijpers P, Wunderink L, Linszen DH (2012) Cognitive behavioral therapy for subjects at ultra-high risk for developing psychosis: a randomized controlled clinical trial. Schizophr Bull 38:1180–1188. https://doi.org/10.1093/schbul/sbs105
McFarlane WR (2016) Family interventions for schizophrenia and the psychoses: a review. Fam Process 55:460–482. https://doi.org/10.1111/famp.12235
Poletti M, Pelizza L, Azzali S, Garlassi S, Scazza I, Paterlini F, Chiri LR, Pupo S, Raballo A (2022) Subjective experience of aberrant salience in young people at ultra-high risk (UHR) for psychosis: a cross-sectional study. Nord J Psychiatry 76:129–137. https://doi.org/10.1080/08039488.2021a
Ficarelli ML, Troisi E, Vignali E, Artoni S, Franzini MC, Montanaro S, Andreoli MV, Marangoni S, Ciampà E, Erlicher D, Pupo S, Pelizza L (2021) Implementing individual and placement support for patients with severe mental illness: findings from the real world. J Psychopathol 27:71–80
SPSS Inc (2010) Statistical Package for Social Science, version 15.0 for Windows. SPSS Press, Chicago (IL)
Ver Hoef JM (2012) Who invented the delta method? Am Statist 66:124–127. https://doi.org/10.1080/00031305.2012.687494
Zhang Z, Reinikainen J, Adeleke KA, Pieterse ME, Groothuis-Oudshoorn CGM (2018) Time-varying covariates and coefficients in Cox regression models. Ann Transl Med 6:121. https://doi.org/10.21037/atm.2018.02.12
Harris JK (2021) Primer on binary logistic regression. Fam Med Community Health 9(Suppl. 1):e001290. https://doi.org/10.1136/fmch-2021-001290
Kirkpatrick B, Strauss GP, Nguyen L, Fischer BA, Daniel DG, Cienfuegos A, Marder SR (2011) The brief negative symptom scale: psychometric properties. Schizophr Bull 37:300–305. https://doi.org/10.1093/schbul/sbq059
Ucok A, Ergul C (2014) Persistent negative symptoms after first episode schizophrenia: a 2-year follow-up study. Schizophr Res 158:241–246. https://doi.org/10.1016/j.schres.2014.07.021
Bleuler E (1911) Dementia Praecox oder Gruppe der Schizophrenien. Verlag, Leipzig
Pelizza L, Poletti M, Azzali S, Garlassi S, Scazza I, Paterlini F, Chiri LR, Pupo S, Raballo A (2020) Subjective experience of social cognition in adolescents at ultra-high risk of psychosis: findings from a 24-month follow-up study. Eur Child Adolesc Psychiatry 29:1645–1657. https://doi.org/10.1007/s00787-020-01482-y
Jang SK, Choi HI, Park S, Jaekal E, Lee GY, Cho YI, Choi KH (2026) A two-factor model better explains heterogeneity in negative symptoms: evidence from the positive and negative syndrome scale. Front Psychol 7:707. https://doi.org/10.3389/fpsyg.2016.00707
Okada H, Hirano D, Taniguchi T (2021) Impact of negative symptom domains and other clinical characteristics on functional outcomes in patients with schizophrenia. Schizophr Res Treatment 2021:8864352. https://doi.org/10.1155/2021/8864352
Barnes TR, Leeson VC, Paton C, Costelloe C, Simon J, Kiss N, Osborn D, Killaspy H, Craig TK, Lewis S, Keown P, Ismail S, Crawford M, Baldwin D, Lewis G, Geddes J, Kumar M, Pathak R, Taylor S (2016) Antidepressant controlled trial for negative symptoms in schizophrenia (ACTIONS): a double-blind, placebo-controlled, randomised clinical trial. Health Technol Assess 20:1–46. https://doi.org/10.3310/hta20290.PMID:27094189;PMCID:PMC4860560
Phan SV, Kreys TJ (2011) Adjunct mirtazapine for negative symptoms of schizophrenia. Pharmacotherapy 31:1017–1030. https://doi.org/10.1592/phco.31.10.1017
Murphy SE, Capitão LP, Giles SLC, Cowen PJ, Stringaris A, Harmer CJ (2021) The knowns and unknowns of SSRI treatment in young people with depression and anxiety: efficacy, predictors, and mechanisms of action. Lancet Psychiatry 8:824–835. https://doi.org/10.1016/S2215-0366(21)00154-1
Leucht S, Leucht C, Huhn M, Chaimani A, Mavridis D, Helfer B, Samara M, Rabaioli M, Bächer S, Cipriani A, Geddes JR, Salanti G, Davis JM (2017) Sixty years of placebo-controlled antipsychotic drug trials in acute schizophrenia: systematic review, bayesian meta-analysis, and meta-regression of efficacy predictors. Am J Psychiatry 174:927–942. https://doi.org/10.1176/appi.ajp.2017.16121358
Correll CU, Schooler NR (2020) Negative symptoms in schizophrenia: a review and clinical guide for recognition, assessment, and treatment. Neuropsychiatr Dis Treat 16:519–534. https://doi.org/10.2147/NDT.S225643
Krause M, Zhu Y, Huhn M, Schneider-Thoma J, Bighelli I, Nikolakopoulou A, Leucht S (2018) Antipsychotic drugs for patients with schizophrenia and predominant or prominent negative symptoms: a systematic review and meta-analysis. Eur Arch Psychiatry Clin Neurosci 268:625–639. https://doi.org/10.1007/s00406-018-0869-3
Veerman SRT, Schulte PFJ, de Haan L (2017) Treatment for negative symptoms in schizophrenia: a comprehensive review. Drugs 77:1423–1459. https://doi.org/10.1007/s40265-017-0789-y
Davidson M, Saoud J, Staner C, Noel N, Luthringer E, Werner S, Reilly J, Schaffhauser JY, Rabinowitz J, Weiser M, Luthringer R (2017) Efficacy and safety of MIN-101: a 12-week randomized, double-blind, placebo-controlled trial of a new drug in development for the treatment of negative symptoms in schizophrenia. Am J Psychiatry 174:1195–1202. https://doi.org/10.1176/appi.ajp.2017.17010122
Beck AT, Grant PM, Huh GA, Perivoliotis D, Chang NA (2013) Dysfunctional attitudes and expectancies in deficit syndrome schizophrenia. Schizophr Bull 39:43–51. https://doi.org/10.1093/schbul/sbr040
Turner DT, van der Gaag M, Karyotaki E, Cuijpers P (2014) Psychological interventions for psychosis: a meta-analysis of comparative outcome studies. Am J Psychiatry 171:523–538. https://doi.org/10.1176/appi.ajp.2013.13081159
Elis O, Caponigro JM, Kring AM (2013) Psychosocial treatments for negative symptoms in schizophrenia: current practices and future directions. Clin Psychol Rev 33:914–928. https://doi.org/10.1016/j.cpr.2013.07.001
Jauhar S, McKenna PJ, Radua J, Fung E, Salvador R, Laws KR (2014) Cognitive-behavioural therapy for the symptoms of schizophrenia: systematic review and meta-analysis with examination of potential bias. Br J Psychiatry 204:20–29. https://doi.org/10.1192/bjp.bp.112.116285
Kaiser S, Lyne J, Agartz I, Clarke M, Mørch-Johnsen L, Faerden A (2017) Individual negative symptoms and domains: relevance for assessment, pathomechanisms and treatment. Schizophr Res 186:39–45. https://doi.org/10.1016/j.schres.2016.07.013
Pelizza L, Leuci E, Quattrone E, Azzali S, Paulillo G, Pupo S, Poletti M, Raballo A, Pellegrini P, Menchetti M (2023) Baseline antipsychotic prescription and short-term outcome indicators in individuals at clinical high-risk for psychosis: findings from the Parma at-risk mental states (PARMS) program. Early Interv Psychiatry. https://doi.org/10.1111/eip.13434
Bucci P, Mucci A, van Rossum IW, Aiello C, Arango C, Baandrup L, Buchanan RW, Dazzan P, Demjaha A, Díaz-Caneja CM, Giordano GM, Glenthøj BY, Leucht S, McGuire P, Rodriguez-Jimenez R, Vignapiano A, Kahn RS, Galderisi S (2020) Persistent negative symptoms in recent-onset psychosis: relationship to treatment response and psychosocial functioning. Eur Neuropsychopharmacol 34:76–86. https://doi.org/10.1016/j.euroneuro.2020.03.010
Strauss GP, Pelletier-Baldelli A, Visser KF, Walker EF, Mittal VA (2020) A review of negative symptom assessment strategies in youth at clinical high-risk for psychosis. Schizophr Res 222:104–112. https://doi.org/10.1016/j.schres.2020.04.019
Rutigliano G, Valmaggia L, Landi P, Frascarelli M, Cappucciati M, Sear V, Rocchetti M, De Micheli A, Jones C, Palombini E, McGuire P, Fusar-Poli P (2016) Persistence or recurrence of non-psychotic comorbid mental disorders associated with 6-year poor functional outcomes in patients at ultra-high risk for psychosis. J Affect Disord 203:101–110. https://doi.org/10.1016/j.jad.2016.05.053
Yang Z, Lim K, Lam M, Keefe R, Lee J (2018) Factor structure of the positive and negative syndrome scale (PANSS) in people at ultra-high risk (UHR) for psychosis. Schizophr Res 201:85–90. https://doi.org/10.1016/j.schres.2018.05.024
Barch DM (2013) The CAINS: theoretical and practical advances in the assessment of negative symptoms in schizophrenia. Am J Psychiatry 170:133–135. https://doi.org/10.1176/appi.ajp.2012.12101329
Strauss GP, Walker EF, Pelletier-Baldelli A, Carter NT, Ellman LM, Schiffman J, Luther L, James SH, Berglund AM, Gupta T, Ristanovic I, Mittal VA (2023) Development and validation of the negative symptom inventory-psychosis risk. Schizophr Bull 49:1205–1216. https://doi.org/10.1093/schbul/sbad038
Carrión RE, Auther AM, McLaughlin D, Olsen R, Addington J, Bearden CE, Cadenhead KS, Cannon TD, Mathalon DH, McGlashan TH, Perkins DO, Seidman LJ, Tsuang MT, Walker EF, Woods SW, Cornblatt BA (2019) The global functioning: social and role scales: further validation in a large sample of adolescents and young adults at clinical high risk for psychosis. Schizophr Bull 45:763–772. https://doi.org/10.1093/schbul/sby126
Galderisi S, Kaiser S, Bitter I, Nordentoft M, Mucci A, Sabé M, Giordano GM, Nielsen MØ, Glenthøj LB, Pezzella P, Falkai P, Dollfus S, Gaebel W (2021) EPA guidance on treatment of negative symptoms in schizophrenia. Eur Psychiatry 64:e21. https://doi.org/10.1192/j.eurpsy.2021.13
Acknowledgements
For their facilitating technical and administrative support in the PARMS program, the authors gratefully acknowledge the “Early Psychosis Facilitators Group” members (Sabrina Adorni, Andrea Affaticati, Anahi Alzapiedi, Paolo Ampollini, Patrizia Caramanico, Maria Teresa Gaggiotti, Tiziana Guglielmetti, Mauro Mozzani, Matteo Rossi, Lucilla Maglio, Matteo Tonna, Fabio Vanni and Matteo Zito) and the “Quality Staff Group” members (Patrizia Ceroni, Stefano Giovanelli, Leonardo Tadonio) of the Parma Department of Mental Health and Pathological Addictions. The authors also wish to thank all the patients and family members who actively participated to the PARMS program.
Funding
Open access funding provided by Alma Mater Studiorum - Università di Bologna within the CRUI-CARE Agreement. This research received no specific grant from any funding agencies in the public, commercial, or not-for-profit sectors. The “Parma At-Risk Mental States” (PARMS) program was partly financed through a special, treatment-oriented regional fund: “Progetto Esordi Psicotici della Regione Emilia Romagna.”
Author information
Authors and Affiliations
Contributions
Lorenzo Pelizza and Emanuela Leuci contributed to study conception and design; Camilla Ricci, Simona Pupo, and Lorenzo Pelizza contributed to literature search and analysis; Emanuela Leuci, Emanuela Quattrone, and Derna Palmisano collected data; Camilla Ricci and Lorenzo Pelizza performed formal analysis and investigation; Camilla Ricci and Lorenzo Pelizza were involved in writing – original draft preparation; all authors were involved in writing – review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent to participate
All individuals and their parents (if minors) gave their written informed consent prior to their inclusion in the study.
Ethical approval
Local ethical approval was obtained for the research (AVEN Ethics Committee protocol n. 559/2020/OSS*/AUSLPR). This investigation was conducted in accordance with the ethical standards of the 1964 Declaration of Helsinki and its later amendments.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ricci, C., Leuci, E., Quattrone, E. et al. Persistent negative symptoms in young people at clinical high risk of psychosis treated with an Italian early intervention program: a longitudinal study. Eur Arch Psychiatry Clin Neurosci (2024). https://doi.org/10.1007/s00406-024-01808-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00406-024-01808-w