Abstract
Major depressive disorder (MDD) and generalized anxiety disorder (GAD) are severe and difficult-to-treat psychiatric illnesses with high rates of comorbidity. Although both disorders are treated with serotonergic based psychotropic agents, little is known on the influence of the serotonergic neurotransmitter system on the occurrence of comorbid GAD when clinically depressed. To investigate this poorly understood clinical question, we examined the involvement of frontolimbic post-synaptic 5-HT2A receptors in 20 medication-resistant depressed (MRD) patients with half of them diagnosed with comorbid GAD with 123I-5-I-R91150 SPECT. To explore whether 5-HT2A receptor-binding indices (BI) associated with comorbid GAD could be related to distinct psychopathological symptoms, all were assessed with the symptom Checklist-90-Revised (SCL-90-R). MRD patients with comorbid GAD displayed significantly higher 5-HT2A receptor BI in the hippocampal–amygdala complex, compared to MRD patients without GAD. Correlation analyses revealed that the 5-HT2A receptor BI in these areas were significantly related to the SCL-90-R subscale hostility (HOS), especially for those MRD patients with comorbid GAD. Comorbid MRD-GAD may be characterized with increased hippocampal–amygdala 5-HT2A receptor BI which could represent enhanced levels in hostility in such kinds of patients. Adapted psychotherapeutic interventions may be warranted.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Major depressive disorder (MDD) and generalized anxiety disorder (GAD) are widespread mental health problems and both illnesses are associated with high recurrence, comorbidity, and they prove to be difficult to treat, so treatment resistance is not uncommon [1, 2]. Pharmacologically managed very similarly with serotonergic based antidepressants [3], it remains, however, unclear how the serotonergic system is involved in the comorbid occurrence of GAD in the depressed state. Better neurobiological insights on the presence of comorbid GAD in MDD could be of considerable interest to fine-tune treatment algorithms in this difficult-to-treat group and certainly in the medication-resistant state.
One possible target for investigation could be the G-protein coupled post-synaptic 5-HT2 receptor, which have been found to be involved in the pathophysiology of mood and anxiety disorders, suicide ideation, and MDD treatment (non)response [4, 5]. Animal and human studies have shown that the 5-HT2A receptor is implicated in the context of anxiety-related behaviors and MDD (see review by [6]). In addition, 5-HT2A receptor dysfunctions of frontolimbic processes are often observed in both GAD [7, 8] and MDD patients [9]. Indeed, functional brain-imaging studies in MDD, as well as in GAD, have also demonstrated deregulated neuronal processing in the frontolimbic areas resulting in enhanced stress sensitivity disturbed emotion regulation processes, rumination, and worrying [10,11,12].
Recently, more attention has been given to the involvement of ‘hostility’ and ‘anger’ in the depressed state [13, 14], which refers to an emotional state varying from mild annoyance to rage [15]. Moreover, elevated levels of multiple dimensions of anger characterize individuals who meet diagnostic criteria for GAD [16]. Here intolerance of uncertainty could be a critical construct underlying the pathophysiology of GAD, and maladaptive behavioral and cognitive reactivity (e.g., biased contextual interpretations of the situation, poor decision-making) may not only increase worry and anxiety [17], but intolerance of uncertainty may also mediate the relationship between GAD symptoms and anger [18]. There is some evidence that supports positive cortical 5-HT2A receptor involvement in aggression, anger, and impulsivity [19]. Surprisingly, no studies have investigated serotonergic differences of comorbid GAD in the treatment-resistant depressed (MRD) state.
Consequently, we collected 20 antidepressant-free (unipolar) melancholic MRD patients (10 with current comorbid GAD) documented to be resistant to several pharmacological interventions, and to examine the influence of the 5-HT2A receptor, we used SPECT and 4-amino-N-[1-[3-(4-fluorophenoxy) propyl]-4-methyl-4-piperidinyl]-5-iodo-2-methoxybenzamide (123I-5-I-R91150) as the radioligand [20] focusing on the frontolimbic areas. We also assessed the symptom Checklist-90-R (SCL-90-R [21]) to further investigate the relationship between SCL-90-R subscales (including a range of psychological measures, besides depression and anxiety, cognitive and conative symptoms such as hostility) and the 5-HT2A receptor BI, to enlarge our insights on the nature of the serotonergic involvement in the co-occurrence of GAD in patients diagnosed with MRD.
Although primary diagnosis was MRD for all patients, we hypothesized that MRD patients with comorbid GAD would display increased frontolimbic 5-HT2A binding BI when compared to those without the GAD comorbidity. Although we explored whether any of the SCL-90-R subscale scores could be related to distinct 5-HT2A receptor BI between MRD patients with or without comorbid GAD, we expected that higher anxiety and the hostility subscale scores would be related to frontolimbic 5-HT2A binding BI.
Methods
Participants
In accordance with the guidelines laid down in the declaration of Helsinki (2004) this study was approved by the Ethical Board of the University Hospital of the Vrije Universiteit Brussel. All patients gave informed consent to participate in the study and for publication. Twenty MRD patients (female: male = 13:7; age = 45.70 years, SD = 9.52) were included using the Dutch version of the Mini-International Neuropsychiatric Interview (M.I.N.I., edition 5.0.0 based on DSM-IV [22]), and all were diagnosed with unipolar melancholic MDD. Depression severity was assessed with the Beck Depression Inventory (BDI-II-NL [23]). Ten patients (50% of the sample) were diagnosed with comorbid GAD. Comorbid psychosis, bipolarity, and substance abuse/dependence were exclusion criteria. Since increased 5-HT2(A) receptor BI are reported in suicide victims [4], suicidal attempts during the current depressive episode were considered as an exclusion criterion. According to the Thase and Rush criteria [24], patients were considered at least stage III treatment resistant: all had at least a minimum of two unsuccessful treatment trials with SSRI/NSRI and one failed clinical trial with a TCA. After a washout period, all patients were free from antidepressant, neuroleptics and mood stabilizers for at least two weeks before scanning. Only benzodiazepines taken on steady dose were allowed. This study was part of a larger investigation examining brain 5-HT2A receptor BI in medication-resistant depression. Given our recent focus on comorbidity in treatment-resistant depression and GAD [25], we re-examined our initial cohort of 21 MRD patients used in an rTMS treatment study [26]. For this study, only the baseline scans were included, before patients underwent the rTMS treatment. Detected retrospectively, one male patient (38 years) diagnosed with MRD and comorbid GAD had an additional opioid addiction (paracetamol codeine for diffuse pain symptoms). Since opioids can increase intrasynaptic levels of serotonin [27], this patient was not included in the current study, nor in another study examining the characterization of 5-HT2A receptor BI in MRD compared to age- and gender-matched drug-naive first episode MDD patients [28].
SCL-90-R
All patients were assessed with the Dutch version of the Symptom Checklist-90-R (SCL-90-R [21]). This 90-item self-report symptom inventory is designed to reflect psychological symptom patterns of psychiatric and medical patients [29]. On a five-point Likert scale, participants rate the degree to which they have experienced symptoms during the past week. The symptom dimensions in the Dutch version are labeled as somatization (SOM), insufficiency in thought and behavior (IN), interpersonal sensitivity (SEN), depression (DEP), anxiety (ANX), hostility (HOS), agoraphobia (AGO), sleeping problems (SLA), and rest symptoms not otherwise specified (REST). The mean value of all separate items is referred to as the Global Severity Index (GSI), an estimate of general psychological distress in psychiatric populations. Given that we were primarily interested in the first eight SCL-90-R subscales (and not the non-specified rest category), we only focused on SOM, IN, SEN, DEP, ANX, HOS, AGO, and SLA.
SPECT brain imaging
Static SPECT-imaging was performed with a Siemens MultiSPECT triple-headed gamma camera, equipped with parallel-hole medium-energy collimators. For thyroid blockage all patients received oral Lugol’s solution containing 400 mg of potassium iodide 15 min prior to injection of an average dose of 150 MBq 123I-5-I-R91150. SPECT acquisition was performed at a minimum of 120 min after administration of the tracer. Data were collected from 96 angular positions over 360° in a 128 × 128 matrix, with a total acquisition time of 32 min. Reconstruction of the acquired projection images was performed using an iterative reconstruction algorithm (Ordered Subset Expectation Maximization, OSEM, 8 iterations, 8 subsets) and filtered with a 3D Gaussian using 15 mm full width at half-maximum.
SPECT scans were automatically co-registered to a template image placed in a predefined stereotactic (image) space (BRASS; Nuclear Diagnostics Ltd., Sweden) (See also [30]). The sequential acquisition of transmission and emission images was further used to anatomically standardize the emission image using the same linear parameters as those used for the transmission image [31]. All images were visually double-checked to ensure correct anatomical positioning of the predefined VOIs [32].
Radioactivity estimates in the volumes of interest (VOI) were assumed to represent total ligand binding (specific plus nonspecific binding plus free ligand) [32]. Since very few 5-HT2A receptors are present in the cerebellum, this region was chosen to represent nonspecific activity [20]. Calculation of relative indices of specific BI was performed by VOI normalization to the activity per volume element in the cerebellum. Under these pseudo-equilibrium circumstances, BI is directly related to the in vivo receptor density (Bmax) and affinity (Kd). BI was defined as (target activity-background activity in the brain)/(background activity) which was operationally estimated as (counts/pixel in VOI-counts/pixel in the cerebellum)/(counts/pixel in the cerebellum) [31].
Given the relatively small study sample, for the analysis we limited our number of VOIs to six: the brainstem, the dorsolateral prefrontal cortex (left + right DLPFC), orbitofrontal cortex (left + right OFC), ventrolateral prefrontal cortex (left + right VLPFC), anterior cingulate cortex (left + right ACC), hippocampal–amygdala complex (left + right HippAM).
Statistical methods
All statistical analyses were performed with SPSS 25 (Statistical Package for the Social Sciences, IBM, Chicago, USA). Where necessary, we applied the Greenhouse–Geisser correction to ensure the assumption of sphericity. The significance level was set at p < 0.05, two-tailed, except for the SCL-90-R. Since the SCL-90-R was introduced for exploratory purposes and given that we were primarily interested in the first eight subscales, labeled SOM, IN, SEN, DEP, ANX, HOS, AGO, and SLA, for these eight subscale analyses with VOI 5-HT2A receptor-binding indices, here the p value, two-tailed was Bonferroni corrected for the number of significant VOIs derived and eight subscales, was set at p < 0.00625, two-tailed (Bonferroni correction).
Behavioral analysis
Concerning the behavioral outcome results, we compared demographics and SCL-90-R questionnaire scores between MRD patients with and without comorbid GAD with X2 or t tests. See for details Table 1.
SPECT analysis
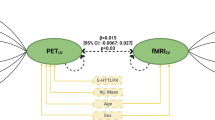
To test our hypothesis on 5-HT2A receptor BI differences between depressed patients with and without comorbid GAD, first, in a MANCOVA analysis the six VOIs were analyzed as multiple dependent variables and MRD group (GAD versus no GAD) was the fixed between subjects’ factor. Given that age-dependent reductions in 5-HT2A receptor-binding indices have consistently been reported [33, 34], we added age as covariate. Significant outcomes of the omnibus MANCOVA were followed up by univariate analyses. Second, to control whether 5-HT2A receptor BI in these significant VOIs could be influenced by the individual daily dose intake of benzodiazepines (mg/day), the duration of the current depressive episode (years), suicide risk (MINI), or the individual depression severity scores (BDI-II), we performed an exploratory (partial) correlation analysis (corrected for age) for the entire sample and for each MRD group (GAD versus no GAD) separately. Third, to evaluate whether one of the eight symptom dimensions of the Dutch version of the SCL-90-R was related to any of the significant VOI 5-HT2A receptor BI, again we performed an (partial) correlation analysis (corrected for age) between the significant VOIs and the eight SCL-90-R subscales for each MRD group separately (GAD versus no GAD), Bonferroni corrected. See Fig. 1.
Sagittal glass brain view depicting the predefined volumes of interest (VOIs). The selected VOIS include brainstem, the dorsolateral prefrontal cortex (DLPFC), the ventrolateral prefrontal cortex (VLPFC), the orbital prefrontal cortex (OFC), the anterior cingulate cortex (ACC), and the hippocampal–amygdala complex (HippAM)
Results
Demographics
MRD patients with (n = 10) or without GAD (n = 10) were not significantly different in age, gender distribution, benzodiazepine use, depression severity, or current episode duration. See Table 1. Of note, for one patient (in the MRD group with comorbid GAD), the SCL-90-R questionnaire went missing, so to evaluate the effects of SCL-90-R subscales only 19 patients were included. Here, we found that MRD patients with GAD scored significantly higher on insufficiency in thought and behavior (IN: p < 0.009) and nearly reached significance on Hostility (HOS; p = 0.052).
Brain imaging
First, the MANCOVA analysis, with the different cortical VOIs (DLPFC, VMPFC, OFC, ACC, HippAM, and brainstem) as dependent variables, MRD group (GAD versus no GAD) as the between group fixed factor, and age as covariate, revealed a significant main effect of group [Wilks’ Lambda: F(6,12) = 3.48, p = 0.03, ηp2 = 0.64], and age [Wilks’ Lambda: F(6,12) = 3.03, p = 0.048, ηp2 = 0.60]. The individual univariate tests showed no significant age influences on 5-HT2A receptor BI, although only for the hippocampal–amygdala complex this nearly reached significance [F(1,17) = 4.35, p = 0.052, ηp2 = 0.20]. The univariate analysis for the other VOIs were not significant (p’s > 0.05). Furthermore, only for the hippocampal–amygdala complex, the individual univariate analysis showed a significant MRD group difference with a significant higher the 5-HT2A receptor BI in patients with comorbid GAD (mean = 301.48, sd = 136.64) compared to those without comorbid GAD (mean = 232.62, sd = 68.80) [F(1,17) = 5.08, p = 0.038, ηp2 = 0.23]. The other univariate analysis evaluating the other VOIs were not significant (p’s > 0.05). See also Table 2.
Second, partial correlation analysis (controlled for age) showed no significant association between HippAM 5-HT2A receptor BI and the use of benzodiazepines, depression severity, suicide risk, and the duration of the current depressive episode for the entire sample, as well for the two groups (GAD vs. no GAD) separately (p’s > 0.01).
Third, because group differences were found to be significant only for the HippAM VOI, for the (partial) correlation analysis with the SCL-90-R subscales, here, the p value was set at p < 0.00625, two-tailed (Bonferroni correction for eight SCL-90-R subscales). To evaluate whether any SCL-90-R questionnaire scales were associated with HippAM 5-HT2A receptor BI (mean = 266.82, sd = 114.09), the partial correlation analysis (controlled for age: mean = 45.74, sd = 9.78) including all MRD patients showed a nearly significant positive correlation with the SCL-90-R subscale HOS only [(mean = 11.42, sd = 4.30), r(16) = 0.64, p = 0.007]. The results of the zero order correlation showed that there was a significant positive correlation between the HippAM 5-HT2A receptor BI and hostility (HOS) only, r(17) = 0.59, p = 0.004, indicating that controlling for age had little effect on the strength of the relationship between the two variables. Separate explorative correlation analyses (not Bonferroni corrected) were separately performed in MRD patients with GAD and without GAD to further investigate whether the HippAM 5-HT2A receptor BI and HOS correlation were any different between the two groups. Pearson partial correlation analysis showed that there was no significant correlation between both variables [r(8) = − 0.27, p = 0.23] in MRD patients without comorbid GAD. However, in MRD patients diagnosed with comorbid GAD, Pearson partial correlation analysis revealed a positive correlation between HippAM 5-HT2A receptor BI and the individual HOS scores [r(7) = 0.71, p = 0.02]. See also Fig. 2. In addition, to assess the significance of the difference between the two correlation coefficients using the Fisher r-to-z transformation, which showed a significant difference (z = − 2.18, p = 0.03).
Scatter plots between the individual scores on hostility (HOS) (x-axis) and Hippocampal amygdala 5-HT2A receptor BI complex (y-axis) for the entire medication-resistant depressed (MRD) sample (straight thin full line), and plotted separately for MRD patients with generalized anxiety disorder (GAD) (thick full line and full bullets) and without comorbid GAD (dashed thick line and open bullets). Of note, the lines representing the least-squares fit to the data are not corrected for age
Finally, given the relatively small sample size and the potential influence of age differences between the two groups, as an additional manipulation check, we removed the oldest patient (female, 61 years, no GAD), and repeated all analyses. Besides that indeed there were no significant age differences between the two groups [t(17) = 1.52, p = 0.15], this did not affect any of the former outcome results, indicating that age was not a major influencer between the group analyses.
Discussion
MRD patients with a comorbid diagnosis of GAD scored higher on insufficiency in thought and behavior (IN), and on hostility (HOS). Both behaviors have been reported when clinically depressed and/or when diagnosed with GAD [3, 15]. Whereas the IN dimension focuses on feelings of personal inadequacy and inferiority in comparisons with others—including self-deprecation, uneasiness, and discomfort during interpersonal interactions—higher scores on HOS indicates thoughts, feelings, or actions characteristic of the negative affect state of anger reflecting qualities such as aggression, irritability, rage and resentment [29]. Besides that, the demographics in both groups were similar (see Table 1), depression and anxiety SCL-90-R subscale scores did also not differ between the two groups, which suggests that the levels of depression and anxiety symptoms were not influenced by the occurrence of comorbid GAD in our MRD sample.
Concerning our SPECT results, the lack of frontal cortical group 5-HT2A receptor BI differences in our sample indicates that serotonergic influences may not differ in MRD with or without comorbid GAD, suggesting no top–down cognitive different influences from the frontal cortical areas [5]. Of note, findings concerning the frontal cortical 5-HT2A receptor in the depressed state are not univocal, with some authors demonstrating 5-HT2A receptor increases, and others demonstrating decreases, or no differences in receptor ligand binding at all [28]. On the other hand, the observed increased (hippocampal) amygdala 5-HT2A receptor BI agrees with its modulating capacity of anxiety-states and anxiety-related behavior and are probably modulated by GABAergic interneurons in the (basolateral) amygdalae [35]. Of interest, in patients with Urbach–Wiethe disease, displaying severe bilateral amygdala damage, this syndrome has been associated with decreases in amygdala 5-HT2A receptor density and related to decreased fear and anxiety behavioral manifestations [36]. In addition, because of its involvement in the hypothalamic–pituitary–adrenal (HPA)-stress system, the role of the hippocampus in the glucocorticoid regulation of stress reactions is well known [37]. Here, the HPA-system is thought to be regulated by serotonin in hippocampal neurons [38, 39], and especially by hippocampal 5-HT2A receptor activation [40].
It is of interest to note that in patients suffering from Borderline Personality Disorder—an emotional unstable and stress-sensitive personality disorder characterized amongst other symptoms with separation anxiety, anger and impulse control disturbances—only hippocampal 5-HT2A receptor binding was increased, independent of depressed mood [41]. Our (partial) correlation analysis showed that increases in HippAM 5-HT2A receptor BI were related to higher scores on the SCL-90-R subscale score hostility (HOS) in the MRD state. Common with BPS symptoms, our exploratory partial correlation analysis also showed that increased HippAM 5-HT2A receptor BI in relation to increased hostility could me especially apparent in MRD patients with comorbid GAD. In addition, GAD may lead to emotional dysregulation, including unsuppressed anger and low tolerance to frustration [42]. In addition, higher levels of anger and hostility contributed and predicted GAD symptom severity and elevated levels of multiple dimensions of anger characterized individuals who met the diagnostic criteria for GAD [16].
In preclinical studies, the involvement of the 5-HT2A receptor in anxious behavior has been observed in dogs [43], and disinhibited aggressive behavior associates with an increased cortical uptake of the 5-HT2A receptor radioligand 123I-5-IR91150 [44]. Of interest, successful SSRI treatment in impulsive-aggressive dogs downregulated the cortical 5-HT2A receptor BI also in the temporal cortical areas, in canine species regarded as part of the hippocampal complex [45]. In depressed adolescents, a single dose of fluoxetine also immediately reduced neural amygdala–hippocampal activity in response to angry facial expressions [46]. Furthermore, among outpatients with MDD early clinical improvement in anger/hostility symptoms may predict response to antidepressant treatment with fluoxetine [47]. It may of interest to note that, although we focused on 5-HT2A receptor BI in the frontolimbic regions, with inclusion of the brainstem, the investigated VOIs are structurally connected to the medial forebrain bundle, primarily mediated by the dopaminergic system as being part of the reward pathway targeting similar brain regions also selected for our current 5-HT2A post-synaptic receptor study (MFB; [48]). In TRD patients, these (superolateral) MFB has been recently the target areas for deep-brain stimulation (DBS; [49, 50]) offering depressed patients unresponsive to several pharmacotherapeutic interventions an option to relief their mental burden. It would be of interest to verify in future DBS studies in depression whether clinical improvement also differentially attenuates hostility thoughts and actions in TRD patients diagnosed with or without GAD.
Patients diagnosed with GAD tend to avoid intense negative emotions by engaging in worry or behavioral avoidance, which may lead to an increase in emotional distress [51], displaying an elevated intolerance of uncertainty (IU) and anger [52]. Importantly, individual and group psychotherapeutic sessions targeting this IU construct in GAD patients may result not only in decreases in IU, but also on worrying and on angriness [18, 53]. One could speculate that MRD patients with comorbid GAD may benefit from such specific additional psychotherapeutic interventions, impacting elevated HippAM 5-HT2A receptor BI.
Besides the obvious limitation, the relatively small sample size, all interpretations should be limited to melancholic MRD patients. Since we have not contrasted our findings to a non-depressed group, a GAD group with no comorbid (treatment resistant) depressed episode, the absence of cortical group 5-HT2A receptor BI only implies that in the MRD cohort frontal 5-HT2A receptor densities are not significantly different with co-occurrence of GAD, and that the hippocampal–amygdala serotonergic findings are specific for the comorbid existence of MRD and GAD. A healthy control group could also have been informative to examine whether 5-HT2A receptor VOI-binding indices would be any different between the healthy and the depressed state, and to evaluate whether in healthy samples hostility symptoms would also be related to higher HippAM 5-HT2A receptor densities. Although MDD was the primary diagnosis, it remains possible that for some the diagnoses of GAD preceded the MDD episode, and not vice versa (e.g., [54]). We used the MINI as a diagnostic tool, but the severity of GAD symptoms was not examined with disease specific questionnaires. The MINI does also not include any personality diagnostic measurement. Although the amygdala and hippocampus may serve different neuronal and behavioral functions, the limited resolution of the SPECT scan prohibited clear disentanglement of 5-HT2A receptor BI between the two anatomical structures [55]. Future PET ligand studies, more capable to delineate between hippocampus and amygdala could examine potential 5-HT2A receptor BI regional differences. Of note, given the sample size of only 20 MRD patients, due to potential insufficient statistical power, the negative findings of the other VOIs may be a false negative. Since left and right VOIs were pooled due to the relatively small sample size, we cannot comment on possible lateralization influences.
In conclusion, the increased hippocampal–amygdala 5-HT2A receptor BI found in depressed patients documented to be resistant to several antidepressant interventions and with the comorbid diagnosis of generalized anxiety could represent a serotonergic biomarker for this comorbid occurrence. Furthermore, the enhanced levels in hostility related to hippocampal–amygdala 5-HT2A receptor BI in such kinds of patients displaying this comorbidity could indicate that adapted psychotherapeutic interventions may be warranted. It remains without saying that these preliminary findings should be replicated in larger clinical samples to substantiate our assumptions.
Availability of data and material
(Data transparency). Data will be made available on reasonable request.
Code availability
(Software application or custom code). Data will be made available on reasonable request.
References
Ferrari AJ, Somerville AJ, Baxter AJ, Norman R, Patten SB, Vos T, Whiteford HA (2013) Global variation in the prevalence and incidence of major depressive disorder: a systematic review of the epidemiological literature. Psychol Med 43:471–481. https://doi.org/10.1017/S0033291712001511
Ferreira-Garcia R, Mochcovitch M, do Cabo MC, Nardi AE, Freire RC (2017) Predictors of pharmacotherapy response in generalized anxiety disorder: a systematic review. Harv Rev Psychiatry 25:65–79. https://doi.org/10.1097/HRP.0000000000000127
Tiller JW (2013) Depression and anxiety. Med J Aust 199:S28-31
van Heeringen C, Audenaert K, Van Laere K, Dumont F, Slegers G, Mertens J, Dierckx RA (2003) Prefrontal 5-HT2a receptor binding index, hopelessness and personality characteristics in attempted suicide. J Affect Disord 74:149–158. https://doi.org/10.1016/s0165-0327(01)00482-7
Stein DJ, Hemmings S, Moolman-Smook H, Audenaert K (2007) 5-HT2A: its role in frontally mediated executive function and related psychopathology. CNS Spectr 12:512–516
Fisher PM, Hariri AR (2013) Identifying serotonergic mechanisms underlying the corticolimbic response to threat in humans. Philos Trans R Soc Lond B Biol Sci 368:20120192. https://doi.org/10.1098/rstb.2012.0192
Fonzo GA, Etkin A (2017) Affective neuroimaging in generalized anxiety disorder: an integrated review. Dialogues Clin Neurosci 19:169–179
Makovac E, Meeten F, Watson DR, Herman A, Garfinkel SND, Critchley H, Ottaviani C (2016) Alterations in amygdala-prefrontal functional connectivity account for excessive worry and autonomic dysregulation in generalized anxiety disorder. Biol Psychiatry 80:786–795. https://doi.org/10.1016/j.biopsych.2015.10.013
Gray JP, Müller VI, Eickhoff SB, Fox PT (2020) Multimodal abnormalities of brain structure and function in major depressive disorder: a meta-analysis of neuroimaging studies. Am J Psychiatry 177:422–434. https://doi.org/10.1176/appi.ajp.2019.19050560
Disner SG, Beever CG, Haigh EA, Beck AT (2011) Neural mechanisms of the cognitive model of depression. Nat Rev Neurosci 12:467–477. https://doi.org/10.1038/nrn3027
Fitzgerald JM, Klumpp H, Langenecker S, Phan KL (2019) Transdiagnostic neural correlates of volitional emotion regulation in anxiety and depression. Depress Anxiety 36:453–464. https://doi.org/10.1002/da.22859
Mochcovitch MD, da Freire RCR, Garcia RF, Nardi AE (2014) A systematic review of fMRI studies in generalized anxiety disorder: evaluating its neural and cognitive basis. J Affect Disord 167:336–342. https://doi.org/10.1016/j.jad.2014.06.041
Jeon HJ, Peng D, Chua HC, Srisurapanont M, Fava M, Bae JN, Chang SM, Hong JP (2013) Melancholic features and hostility are associated with suicidality risk in Asian patients with major depressive disorder. J Affect Disord 148:368–374. https://doi.org/10.1016/j.jad.2013.01.001
Salem T, Winer ES, Jordan DG, Nadorff MR, Fanning JR, Bryant J, Berman ME, Veilleux JC (2018) Anhedonia and the relationship between other depressive symptoms and aggressive behavior. J Interpers Violence. https://doi.org/10.1177/0886260518770646
Riley WT, Treiber FA, Woods MG (1989) Anger and hostility in depression. J Nerv Ment Dis 177:668–674
Deschênes SS, Dugas MJ, Fracalanza K, Koerner N (2012) The role of anger in generalized anxiety disorder. Cognit Behav Ther 41:261–271. https://doi.org/10.1080/16506073.2012.666564
Bomyea J, Ramsawh H, Ball TM, Taylor CT, Paulus MP, Lang AJ, Stein MB (2015) Intolerance of uncertainty as a mediator of reductions in worry in a cognitive behavioral treatment program for generalized anxiety disorder. J Affect Disord 33:90–94. https://doi.org/10.1016/j.janxdis.2015.05.004
Laposa JM, Fracalanza K (2019) Does intolerance of uncertainty mediate improvement in anger during group CBT for GAD? A preliminary investigation. Behav Cognit Psychother 47:585–593. https://doi.org/10.1017/S1352465819000249
da Cunha-Bang S, Stenbæk DS, Holst K, Licht CL, Jensen PS, Frokjaer VG, Mortensen EL, Knudsen GM (2013) Trait aggression and trait impulsivity are not related to frontal cortex 5-HT2A receptor binding in healthy individuals. Psychiatry Res 212:125–131. https://doi.org/10.1016/j.pscychresns.2012.09.007
Terriere D, Janssen PM, Gommeren W, Gysemans M, Mertens JJ, Leysen JE (1995) Evaluation of radioiodo-4-amino-N-[1-[3-(4-fluorophenoxy)-propyl]-4- methyl-4-piperidinyl]-5-iodo-2-methoxybenzamide as a potential 5HT2 receptor tracer for SPE(C)T. Nucl Med Biol 22:1005–1015. https://doi.org/10.1016/0969-8051(95)02023-3
Arrindell WA, Ettema JHM (1986) SCL-90: Handleiding bij een multidimensionele pathologie-indicator. Swets, Test Publishers, Lisse
Overbeek T, Schruers K, Griez E (1999) Mini international neuropsychiatric interview. University of Maastricht, Maastricht
Beck AT, Steer RA, Ball R, Ranieri WF (1996) Comparison of beck depression inventories-IA and -II in psychiatric outpatients. J Pers Assess 67:588–597
Rush AJ, Thase ME, Dubé S (2003) Research issues in the study of difficult-to-treat depression. Biol Psychiatry 53:743–753. https://doi.org/10.1016/s0006-3223(03)00088-x
Baeken C, Wu GR, De Raedt R (2018) Dorsomedial frontal cortical metabolic differences of comorbid generalized anxiety disorder in refractory major depression: a [18F] FDG PET brain imaging study. J Affect Disord 227:550–553. https://doi.org/10.1016/j.jad.2017.11.066
Baeken C, De Raedt R, Bossuyt A, Van Hove C, Mertens J, Dobbeleir A, Blanckaert P, Goethals I (2011) The impact of HF-rTMS treatment on serotonin(2A) receptors in unipolar melancholic depression. Brain Stimul 4:104–111. https://doi.org/10.1016/j.brs.2010.09.002
Baldo BA (2018) Opioid analgesic drugs and serotonin toxicity (syndrome): mechanisms, animal models, and links to clinical effects. Arch Toxicol 92:2457–2473. https://doi.org/10.1007/s00204-018-2244-6
Baeken C, De Raedt R, Bossuyt A (2012) Is treatment-resistance in unipolar melancholic depression characterized by decreased serotonin2A receptors in the dorsal prefrontal—anterior cingulate cortex? Neuropharmacology 62:340–346. https://doi.org/10.1016/j.neuropharm.2011.07.043
Derogatis LR, Rickels K, Rock AF (1976) The SCL-90 and the MMPI: a step in the validation of a new self-report scale. Br J Psychiatry 128:280–289
Van Laere K, Koole M, D’Asseler Y, Versijpt J, Audenaert K, Dumont F, Dierckx R (2001) Automated stereotactic standardization of brain SPECT receptor data using single-photon transmission images. J Nucl Med 42:361–375
Audenaert K, Van Laere K, Dumont F, Vervaet M, Goethals I, Slegers G, Merten J, van Heeringen C, Dierckx RA (2003) Decreased 5-HT2a receptor binding in patients with anorexia nervosa. J Nucl Med 44:163–169
Goethals I, Vervaet M, Audenaert K, Van de Wiele C, Ham H, Vandecapelle M, Slegers G, Dierckx R, van Heeringen C (2004) Comparison of cortical 5-HT2A receptor binding in bulimia nervosa patients and healthy volunteers. Am J Psychiatry 161:1916–1918. https://doi.org/10.1176/ajp.161.10.1916
Baeken C, D’haenen H, Flamen P, Mertens J, Terriere D, Chavatte K, Boumon R, Bossuyt A (1998) 123I–5-I-R91150, a new single-photon emission tomography ligand for 5-HT2A receptors: influence of age and gender in healthy subjects. Eur J Nucl Med 25:1617–1622. https://doi.org/10.1007/s002590050339
Meltzer CC, Smith G, Price JC, Reynolds CF 3rd, Mathis CA, Greer P, Lopresti B, Mintun MA, Pollock BG, Ben-Eliezer D, Cantwell MN, Kaye W, DeKosky ST (1998) Reduced binding of [18F]altanserin to serotonin type 2A receptors in aging: persistence of effect after partial volume correction. Brain Res 813:167–171. https://doi.org/10.1016/s0006-8993(98)00909-3
Hale MW, Johnson PL, Westerman AM, Abrams JK, Shekhar A, Lowry CA (2010) Multiple anxiogenic drugs recruit a parvalbumin-containing subpopulation of GABAergic interneurons in the basolateral amygdala. Prog Neuro Psychopharmacol Biol Psychiatry 34:1285–1293. https://doi.org/10.1016/j.pnpbp.2010.07.012
Hurlemann R, Schlaepfer TE, Matusch A, Reich H, Shah NJ, Zilles K, Maier W, Bauer A (2009) Reduced 5-HT(2A) receptor signaling following selective bilateral amygdala damage. Soc Cognit Affect Neurosci 4:79–84. https://doi.org/10.1093/scan/nsn039
Schmidt HD, Duman RS (2007) The role of neurotrophic factors in adult hippocampal neurogenesis, antidepressant treatments and animal models of depressive-like behavior. Behav Pharmacol 18:391–418. https://doi.org/10.1097/FBP.0b013e3282ee2aa8
Keeney A, Jessop DS, Harbuz MS, Marsden CA, Hogg S, Blackburn-Munro RE (2006) Differential effects of acute and chronic social defeat stress on hypothalamic-pituitary-adrenal axis function and hippocampal serotonin release in mice. J Neuroendocrinol 18:330–338
Lai M, McCormick JA, Chapman KE, Kelly PA, Seckl JR, Yau JL (2003) Differential regulation of corticosteroid receptors by monoamine neurotransmitters and antidepressant drugs in primary hippocampal culture. Neuroscience 118:975–984
Umeda Y, Amano M, Suemaru K, Yamaguchi T, Kitamura Y, Gomita Y, Kawasaki H, Araki H (2007) The Influence of hyperactivity of the hypothalamic-pituitary-adrenal axis and hyperglycemia on the 5-HT2A receptor-mediated wet-dog shake responses in rats. Acta Med Okayama 61:311–317. https://doi.org/10.18926/AMO/32879
Soloff PH, Price JC, Meltzer CC, Fabio A, Frank GK, Kaye WH (2007) 5HT2A receptor binding is increased in borderline personality disorder. Biol Psychiatry 62:580–587. https://doi.org/10.1016/j.biopsych.2006.10.022
Madonna D, Delvecchio G, Soares JC, Brambilla P (2019) Structural and functional neuroimaging studies in generalized anxiety disorder: a systematic review. Braz J Psychiatry 41:336–362. https://doi.org/10.1590/1516-4446-2018-0108
Vermeire ST, Audenaert KR, Dobbeleir AA, De Meester RH, De Vos FJ, Peremans KY (2009) Evaluation of the brain 5-HT2A receptor binding index in dogs with anxiety disorders, measured with 123I–5I-R91150 and SPECT. J Nucl Med 50:284–289. https://doi.org/10.2967/jnumed.108.055731
Peremans K, Audenaert K, Coopman F, Blanckaert P, Jacobs F, Otte A, Verschooten F, van Bree H, van Heeringen K, Mertens J, Slegers G, Dierckx R (2003) Estimates of regional cerebral blood flow and 5-HT2A receptor density in impulsive, aggressive dogs with 99mTc-ECD and 123I–5-I-R91150. Eur J Nucl Med Mol Imaging 30:1538–1546. https://doi.org/10.1007/s00259-003-1250-x
Peremans K, Audenaert K, Hoybergs Y, Otte A, Goethals I, Gielen I, Blankaert P, Vervaet M, van Heeringen C, Dierckx R (2005) The effect of citalopram hydrobromide on 5-HT2A receptors in the impulsive-aggressive dog, as measured with 123I–5-I-R91150 SPECT. Eur J Nucl Med Mol Imaging 32:708–716. https://doi.org/10.1007/s00259-005-1772-5
Capitão LP, Chapman R, Murphy SE, Harvey CJ, James A, Cowen PJ, Harmer CJ (2019) A single dose of fluoxetine reduces neural limbic responses to anger in depressed adolescents. Transl Psychiatry 9:30. https://doi.org/10.1038/s41398-018-0332-2
Farabaugh A, Sonawalla S, Johnson DP, Witte J, Papakostas GI, Goodness T, Clain A, Baer L, Mischoulon D, Fava M, Harley R (2010) Early improvements in anxiety, depression, and anger/hostility symptoms and response to antidepressant treatment. Ann Clin Psychiatry 22:166–171
Bracht T, Linden D, Keedwell P (2015) A review of white matter microstructure alterations of pathways of the reward circuit in depression. J Affect Disord 187:45–53
Fenoy AJ, Schulz PE, Selvaraj S, Burrows CL, Zunta-Soares G, Durkin K, Zanotti-Fregonara P, Quevedo J, Soares JC (2018) A longitudinal study on deep brain stimulation of the medial forebrain bundle for treatment-resistant depression. Transl Psychiatry 8:111
Döbrössy MD, Ramanathan C, Ashouri Vajari D, Tong Y, Schlaepfer T, Coenen VA (2021) Neuromodulation in psychiatric disorders: experimental and clinical evidence for reward and motivation network deep brain stimulation: focus on the medial forebrain bundle. Eur J Neurosci 53:89–113
Hawkins A, Cougle JR (2011) Anger problems across the anxiety disorders: findings from a population-based study. Depress Anxiety 28:145–152. https://doi.org/10.1002/da.20764
Fracalanza K, Koerner N, Deschênes SS, Dugas MJ (2014) Intolerance of uncertainty mediates the relation between generalized anxiety disorder symptoms and anger. Cognit Behav Ther 43:122–132. https://doi.org/10.1080/16506073.2014.888754
Hui C, Zhihui Y (2017) Group cognitive behavioral therapy targeting intolerance of uncertainty: a randomized trial for older Chinese adults with generalized anxiety disorder. Aging Ment Health 21:1294–1302. https://doi.org/10.1080/13607863.2016.1222349
Moffitt TE, Harrington H, Caspi A, Kim-Cohen J, Goldberg D, Gregory AM, Poulton R (2007) Depression and generalized anxiety disorder: cumulative and sequential comorbidity in a birth cohort followed prospectively to age 32 years. Arch Gen Psychiatry 64:651–660. https://doi.org/10.1001/archpsyc.64.6.651
Bombardi C (2014) Neuronal localization of the 5-HT2 receptor family in the amygdaloid complex. Front Pharmacol 5:68. https://doi.org/10.3389/fphar.2014.00068
Acknowledgements
This research was supported by the National Natural Science Foundation of China (Grant no. 61876156), a grant from the Scientific Fund W. Gepts UZBrussel (C.B), and a BOF16/GOA/017 grant for a Concerted Research Action of Ghent University (R.D.R & C.B).
Funding
(Information that explains whether and by whom the research was supported). This research was supported by the National Natural Science Foundation of China (Grant No. 61876156), a grant from the Scientific Fund W. Gepts UZBrussel (C.B), and a BOF16/GOA/017 grant for a Concerted Research Action of Ghent University (R.D.R & C.B).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
(Include appropriate disclosures). None to report.
Ethics approval
(Include appropriate approvals or waivers). In accordance with the guidelines laid down in the declaration of Helsinki (2004), this study was approved by the Ethical Board of the University Hospital of the Vrije Universiteit Brussel.
Consent to participate
(Include appropriate statements). All the patients gave informed consent.
Consent for publication
(Include appropriate statements). All the patients gave informed consent to participate in the study and for publication.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Baeken, C., Xu, Y., Wu, GR. et al. Hostility in medication-resistant major depression and comorbid generalized anxiety disorder is related to increased hippocampal–amygdala 5-HT2A receptor density. Eur Arch Psychiatry Clin Neurosci 271, 1369–1378 (2021). https://doi.org/10.1007/s00406-021-01243-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00406-021-01243-1