Abstract
Purpose
Identifying outcome measurements instruments (OMIs) to evaluate treatment efficacy in patients with vocal fold atrophy and/or sulcus.
Methods
Systematic review of records published before March 2021 by searching Pubmed and EMBASE. Included studies reported on adults (> 18 year) with dysphonia caused by glottic insufficiency due to vocal fold atrophy with or without sulcus, who were enrolled into a randomized controlled trial, a non-randomized controlled trial, a case-controlled study or a cohort study. All included studies described an intervention with at least one outcome measurement.
Results
A total of 5456 studies were identified. After removing duplicates, screening title and abstract and full text screening of selected records, 34 publications were included in final analysis. From these 50 separate OMIs were recorded and categorized according to the ELS protocol by DeJonckere et al. (Eur Arch Otorhinolaryngol 258: 77–82, 2001). With most OMIs being used in multiple studies the total number of OMIs reported was 265. Nineteen (19) individual OMIs accounted for 80% of reports. The most frequently used OMIs according to category were: VHI and VHI-10 (subjective evaluation); G of GRBAS (perceptual evaluation); F0, Jitter and Shimmer (acoustic evaluation); MPT and MFR (aerodynamic evaluation) and glottic closure and mucosal wave (endoscopic evaluation). Of these OMIs VHI had a high percentage of significance of 90%.
Conclusion
This systematic review identifies the most used OMIs in patients with glottic incompetency due to vocal fold atrophy and/or sulcus as a step toward defining a Core Outcome Set (COS) for this population.
PROSPERO registration
238274.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Vocal fold atrophy, with or without sulcus, can lead to both reduced vocal fold closure and reduced vibration during phonation. There is however a great heterogeneity in presentation among patients, varying from mild to severe dysphonia with a similarly large variation in patient’s disease burden and findings during laryngostroboscopy. There is also a wide variation in treatment options for the glottic incompetence caused by these entities varying from speech language therapy (SLT) to different forms of laryngeal surgery. Vocal fold injection (VFI) with different injection material or laryngeal frame surgery (LFS) are often performed to improve glottic closure and for patients with sulcus there is also the option of microphonosurgery of the upper vibratory layers of the vocal fold using cold steel, lasers or tissue engineering techniques [2, 3]. Finally, there is also variation in outcome measurement instruments (OMIs) used to assess the severity of the condition and/or treatment outcome. These OMIs can be divided into subjective (self-assessment), perceptual, aerodynamic and acoustic measurements in addition to videolaryngostroboscopic findings according to the guidelines on voice quality assessment published and recently updated by the European Laryngological Society [1, 4].
Taking all of the above into account, evaluating treatment outcome in this patient group in a consistent way is challenging. To reliably evaluate and compare treatment outcomes it is important to formulate a core outcome set (COS). A COS is a consensus-based agreed minimum set of outcomes that should be evaluated and reported in clinical trials in a specific disease or population. For unilateral vocal fold paralysis (UVFP) such a COS has been formulated by Desuter et al., but up until now this is lacking for patients with non-paralytic glottic insufficiency [5, 6]. The protocol for developing a COS in an evidenced-based multi-step process has been described in the COSMIN guidelines (consensus-based standards for the selection of health measurement instruments) [7,8,9]. In the first step, a definition for the construct to be measured is created. In the second step, the existing OMIs for the defined construct are determined through a systematic review of the literature. In the third step, quality assessments of the included studies and the OMIs are performed. Finally, in the fourth step minimal outcome measures to be included in the COS are selected, often in a Delphi type procedure [10].
The definition of the construct of voice (step 1) has already been achieved through the work of the European Laryngological Society guidelines described earlier [1]. The aim of this review is to establish a systematic overview of the frequency and type of OMIs used in literature for patients with non-paralytic glottic insufficiency caused by vocal fold atrophy with or without sulcus as a further step towards formulating a COS for this patient population.
Materials and methods
The design of this study was modeled on earlier studies such as by Desuter et al. in patients with vocal fold paralysis [5]. The construct to be measured in this review was determined to be “treatment effect in patients with dysphonia caused by vocal fold atrophy and/or sulcus”. A systematic review was conducted following the preferred reporting items for systematic reviews and meta-analyses (PRISMA) statement [11]. With assistance of a clinical librarian a search was performed in two databases Pubmed and EMBASE.
The search in Pubmed was constructed with following terms:
("Glottis"[MeSH Terms] OR "Glottis"[Title/Abstract] OR "glottic"[Title/Abstract] OR "glottal"[Title/Abstract] OR "vocal fold"[Title/Abstract] OR "vocal folds"[Title/Abstract] OR "vocal cord"[Title/Abstract] OR "vocal cords"[Title/Abstract]) AND ("incompetence"[Title/Abstract] OR "incompetency"[Title/Abstract] OR "insufficience"[Title/Abstract] OR "insufficiency"[Title/Abstract] OR "Atrophy"[MeSH Terms:noexp] OR "Atrophy"[Title/Abstract] OR "atrophic"[Title/Abstract] OR "Cicatrix"[MeSH Terms] OR "scar"[Title/Abstract] OR "scars"[Title/Abstract] OR "scarring"[Title/Abstract] OR "scarred"[Title/Abstract] OR "sulcus"[Title/Abstract] OR "paresis"[Title/Abstract] OR "pareses"[Title/Abstract] OR "hypomobil*"[Title/Abstract] OR "hypo mobil*"[Title/Abstract]).
The search in EMBASE was constructed with the following terms:
('glottis'/exp OR 'glottis' OR 'glottis':ti,ab,kw OR 'glottic':ti,ab,kw OR 'glottal':ti,ab,kw OR 'vocal fold':ti,ab,kw OR 'vocal folds':ti,ab,kw OR 'vocal cord':ti,ab,kw OR 'vocal cords':ti,ab,kw) AND ('incompetence':ti,ab,kw OR 'incompetency':ti,ab,kw OR 'insufficience':ti,ab,kw OR 'insufficiency':ti,ab,kw OR 'atrophy'/exp OR 'atrophy' OR 'atrophy':ti,ab,kw OR 'atrophic':ti,ab,kw OR 'scar'/exp OR 'scar':ti,ab,kw OR 'scars':ti,ab,kw OR 'scarring':ti,ab,kw OR 'scarred':ti,ab,kw OR 'sulcus':ti,ab,kw OR 'paresis':ti,ab,kw OR 'pareses':ti,ab,kw OR 'hypomobil*':ti,ab,kw OR 'hypo mobil*':ti,ab,kw).
Records published before March 2021 were identified. Included studies reported on adults (> 18 year) with dysphonia caused by glottic insufficiency due to vocal fold atrophy with or without sulcus, who were enrolled into a randomized controlled trial, a non-randomized controlled trial, a case-controlled study or a cohort study. All included studies described an intervention with at least one outcome measurement. Studies including other etiologies of glottic incompetence, with vocal fold atrophy and/or sulcus included as a subgroup, were not included. Pre-clinical studies, including animal and laboratory studies, were also excluded, as were case reports.
Title and abstract of the identified studies were screened by two independent reviewers (EB and SM). This was followed by full-text evaluation by one reviewer (EB). Baseline characteristics of the included studies were extracted (publication date, study type, diagnosis, number of patients, gender, mean age, treatment, follow up). Treatment was categorized in three groups: speech language therapy (SLT), surgery and regenerative therapy. The surgery group included microlaryngeal surgery (MLS) with cold steel instruments—labelled as “direct”- and with the use of laser—labelled as “laser”- and medialization technique vocal fold injection (VFI) and laryngeal framework surgery (LFS). All reported OMIs were extracted and listed according to frequency of use. Cumulative percentages were calculated to identify the OMIs accounting for 80% of the total of reported OMIs and outcomes were displayed in a Pareto diagram. Subsequently these OMIs were also subdivided in categories based on the ELS protocol for functional assessment of dysphonia [1] consisting of: subjective parameters, perceptual parameters, acoustic and aerodynamic parameters and videolaryngostroboscopic findings (henceforth endoscopic findings). In this last category we included the assessment of glottic closure and mucosal wave in accordance with a recent review on vocal fold scar [3]. OMIs on respiratory function were separately collected. OMIs that did not fit this category, nor the categories of the ELS protocol were reported as “additional” parameters. Finally, the percentage of significance for the most frequently used OMIs, defined as the number of studies with a significant post-treatment improvement divided by the total number of studies using this OMI, was calculated [5].
Results
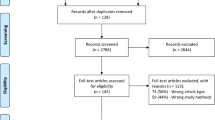
A total of 173 articles were full text assessed. Fifty-eight (58) studies were excluded for the following reasons: population not clearly described (n = 11), no atrophy and/or sulcus included (n = 12), no therapy (n = 2), no relevant outcome (n = 2, complication, duration), no pre- and posttreatment outcome (n = 9), language other than English (n = 10, 1 Slovenic, 1 Polish, 1 Serbian, 2 Japanese, 2 Chinese, 3 Portuguese), abstract only (n = 10), duplicate (n = 2). Another 81 studies were excluded, because they included a mixed study populations of glottic insufficiencies including atrophy and/or sulcus but also scar, paresis, hypomobility, paralysis or other causes. Thirty-four (34) studies, only including atrophy and/or sulcus, were included in final qualitative synthesis (Fig. 1).
Table 1 shows the baseline characteristics and OMIs of the included studies [2, 12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44] .
Two studies were randomized clinical trials [13, 16]; of which one was a double blind randomized controlled trial (RCT)[16]. All other reports were cohort studies of which 7 were prospective, 25 were retrospective; 11 studies included atrophy patients, 14 included sulcus, and 9 studies atrophy and sulcus. Two studies included a control group; one study with sulcus in a prospective cohort and one study with atrophy and sulcus in a RCT, mentioned above [16, 33].
In 3 studies treatment consisted of SLT, in 3 studies SLT or surgery, in 24 surgical treatments and in 4 regenerative therapy. The studies with surgical treatment were divided into “direct” microlaryngeal surgery (MLS), including microphonosurgical cold steel procedures invading subepithelial space with or without grafting (n = 6), MLS with laser surgery, including KTP, PDL and CO2 laser (n = 2), different types of medialization both VFI with different fillers (hyaluronic acid (HA), calciumhydroxylapatite (CaHA), carboxymethylcellulose (CMC)) and LFS (n = 10) or a combination of above described technics (n = 6). Follow-up varied from 1 to 36 months (median of 8 months). In Table 2 an overview is presented of the included studies and their OMIs.
A total of 50 different OMIs were reported in the 34 selected studies. With most OMIs being used in multiple studies the total number of OMIs reported was 265. The frequencies of the different OMIs are shown in Table 3 and additionally as a Pareto diagram in Fig. 2. Nineteen (19) OMIs accounted for 80% of reports. Table 4 shows an overview of these top 19 OMIs identified from the Pareto diagram divided into the ELS subcategories, together with the percentage of studies that find a significant impact after treatment. Five of these OMIs show a significant change after treatment in more than half of the studies that reported on them.
Pareto diagram of OMIs. MV mucosal wave, F0 fundamental frequency, MPT maximum phonation time, Shim shimmer, GRBAS grade roughness breathiness asthenic strain, Jitt jitter, GC glottic closure, NHR noise to harmonic tatio, VHI voice handicap index, MFR mean flow rate, MR melodic range, HNR harmonic to noise ratio, APQ amplitude pertubation quotient, DR dynamic range, PPQ pitch perturbation quotient, SPI soft phonation index, PT phonation time, Psub subglottal pressure, SPL sound pressure level, CAPE-V consensus auditory-perceptual evaluation of voice, vF0 fundamental frequency coefficient variation, vAm peak-to-peak amplitude, GFI glottal function index, Jita absolute jitter, RAP relative average perturbation, sPPQ smoothed pitch perturbation quotient, sAPQ smoothed amplitude pertubation quotient, VRQOL voice related quality of life, CPIB communicative participation item bank, VAS visual analog scale, ShdB absolute shimmer, VTI voice turbulence index, NNE normalized noise energy, CPPS smoothed cepstral peak prominence, CPP cepstral peak prominence, CSID cepstral spectral index of dysphonia, PQ phonation quotient, AR aerodynamic resistance
Discussion
In this review we identified the OMIs most used to evaluate treatment effect in patients with non-paralytic glottic insufficiency caused by vocal fold atrophy with and without sulcus. A total of 50 different OMIs were identified with 19 of these accounting for 80% of total reported OMIs. Of these 19 OMIs, five showed a significant change after treatment in more than half of the studies where they were used.
Interestingly, of the top ten most used parameters most were acoustic, aerodymamic or stroboscopic. Only one patients’ self-evaluation parameter was included in the top ten which was the VHI-30 ranked as 9th most used while self-evaluation is one of the most clinically relevant tools for measuring treatment outcome in daily practice. Additionally, several studies have proposed that it is the one most reliable tool for evaluating treatment response in this patient population [2, 45, 46]. This review shows it has a much higher percentage of significance than the acoustic or endoscopic parameters.
It is well known that assessing voice outcome after treatment is complex and that multidimensional evaluation is necessary. With the large body of OMIs available, choosing representative and reliable parameters is challenging and much evidence points to the fact that disease specific core outcome sets of OMIs are needed [10]. Before formulating such a COS a basic overview of parameters used in literature is required. It is important to emphasize that the parameters found to be the most frequently used for patients with non-paralytic glottic insufficiency caused by vocal fold atrophy with or without sulcus in this review may not necessarily be the most appropriate for this cohort. To properly assess the usefulness of an OMI, not only frequency of use, but also clinical relevance, applicability and psychometric validity are important factors to consider [10].
However, as a starting point, it is valuable to have insight into the choices that are currently being made by clinicians. The findings of this review can guide further initiatives on the route to a COS by indicating which parameters should be prioritized going forwards. The top OMIs revealed by this review as well as the factors for determining the ultimate relevance of an OMI are discussed below.
Subjective OMIs
The VHI-30 was the most frequently used subjective OMI (n = 10, 9th rank) and had a very high percentage of significance at 90%. The VHI-10 was the second most used (n = 7, 12th rank) with a lower percentage of significance of 57%. Around 75% of the studies in this review (26 out of 34) used a form of subjective rating. Subjective evaluation is one of the most clinically relevant tools in the communication with patients. The VHI is a robust questionnaire with translations and validations in many languages and it has the most sufficient psychometric construct based on COSMIN taxonomy [47, 48]. One may argue that VHI is designed for dysphonic patients in general and not specifically for patients with glottic insufficiency and that a questionnaire especially designed for glottic insufficiency may be preferred above VHI. More focused questionnaires for a future COS could be the glottal function index (GFI)[49], vocal fatigue index (VFI)[50] or vocal fatigue handicap questionnaire (VFHQ)[51] with the GFI having the advantage above the other disease specific questionnaires being the only OMI with moderate positive rating on psychometric ratings [48].
However, it is also important to consider that instead of incorporating ever more detailed disease specific PROMs (Patient-reported Outcome Measurement), there is also a countercurrent in literature supporting the development and use of generic PROMs focusing on general health aspects such as physical, mental and social health including quality of sleep or ability to work. An initiative to develop and measure generic PROMs is PROMIS (Patient-reported Outcomes Measurement Information System) which is an innovative, intelligent system for measuring generic PROMs to be used for different health problems and diseases (www.healthmeasures.net). A generic, non-disease specific health survey may be also of interest as a quality of life measurement instrument which can be used for cost-utility analysis by measuring quality-adjusted life years (QALYs) such as the EQ-5d (EuroQol 5D) [52].
Perceptual OMIs
The GRBAS was the most frequently used perceptual OMI (n = 18, 5th rank), with a percentage of significance of 72%. GRBAS is a widely used perception scale. The G, general grade, has a satisfactory inter- and intra-rater reliability and is therefore suitable as a single OMI. In the latest ELS proposal the use of complete GRBAS scale is preferred [4]. A main disadvantage of using perceptual OMIs in patients with non-paralytic glottic insufficiency is that structural defects, such as sulcus, are not always addressed in treatments, such as medialization procedures, where the primary goal of treatment is to improve endurance and not perceptual quality of the voice [17, 22].
Acoustic OMIs
Interestingly, our review showed that studies relied heavily on acoustic OMIs such as fundamental frequency (F0) (n = 20, 2nd rank), shimmer (n = 19, 4th rank), jitter (n = 18, 6th rank) and noise to harmonic ratio (NHR) (n = 11, 8th rank), even though none of these acoustic parameters achieved a percentage of significance above 50%. Their high frequency of use is likely due to them being automatically provided by most voice programs, but their clinical usefulness may be less defined. They are less intuitive in communications with patients, in our and other’s experience, and have been shown not to correspond to more clinically relevant parameters [2, 17, 22, 45].
Nevertheless, acoustic OMIs could potentially aid in detecting differences in the regularity of phonation that may be missed with more broad spanning parameters such as perceptual evaluation. The challenge would be to find the appropriate ones for this specific patient population from the large number of parameters available. Despite its low ranking and lack of significance in our review, one example could be the soft phonation index (SPI) (n = 4, 19th rank, 0% percentage of significance) which reflects the approximation of vocal folds [53]. It’s possible usefulness has been shown in unilateral nodules, but, to our knowledge, has not been clarified in atrophy and/or sulcus [54]. Inconsistency in normal values and increased SPI for pressed phonation have been seen [53, 54]. This may hamper the interpretation of SPI in atrophy and sulcus.
Aerodynamic OMIs
Of the top 19 OMIs used, three were aerodynamic; maximum phonation time (MPT) (n = 19, 3th rank), mean flow rate (MFR) (n = 10, 10th rank), and dynamic range (DR) (n = 5, 15th rank). MPT is a well-known voice parameter; it is simple, reliably obtainable, but with the disadvantage that normative data will differ in sub-populations depending on gender or age [55, 56]. MPT has been found to be the most used and most significant OMI for UFVP (90% percentage of significance) [5]. Our results indicated a less prominent role in our patient group (68% percentage of significance), possibly due to the difference in underlying pathology, including the degree of glottal gap that needs correction. Aerodynamic OMIs that require a pneumotachograph are less easy to obtain, f.e. MFR or phonation quotient ((PQ)(vital capacity/MPT)) as alternative. MFR may be of value for glottic insufficiency with mobile vocal folds, as it is for immobile vocal fold in UVFP, stated by Desuter et al., with relatively high ranking and percentage of significance (86% percentage of significance) [5].
Another measurement of interest is the phonation threshold pressure (Pth). It reflects the minimum subglottic pressure needed to reach phonation onset and sustain phonation [57]. It may be more appropriate to capture the subtle changes in subglottic pressure when comparing pre- and posttreatment effect. It has found only limited use up till now, although a preliminary study in 2021 showed that measuring Pth in UVFP is feasible [58]. Attributing factors for this may be variations in procedural methodology for task elicitation as well as environmental and participant inconsistencies that might affect phonation threshold pressure values [59].
Endoscopic OMIs
Mucosal wave was the most used OMI (n = 21, 1th rank) followed by glottic closure (n = 18, 7th rank), although both had a relative low percentage of significance (47% and 28% respectively). It is therefore debatable if endoscopic parameters are the most suitable OMIs for this patient population due to the inherent inter-observer bias associated with this form of assessment and the combined pathology of atrophy and sulcus leading to further difficulties in assessing exams [4, 60].
However, as endoscopy is broadly used in this patient group, more systematic and detailed videolaryngostroboscopic assessment protocols should be investigated, f.e. as described in VALI (Voice-Vibratory Assessment with Laryngeal Imaging)[61]. Frame-by-frame- analysis (FBFA) could also be useful [62]. Another possibility would be to use disease specific laryngoscopic assessments. For vocal fold atrophy, the reliability of laryngoscopic features have been investigated with satisfying results and recently a validated classification of presbylarynx based on laryngoscopic findings has been published [63, 64].
As stated in the introduction, to properly assess the usefulness of an OMI, before it can be included in a COS, quality assessment has to be performed. In doing so, not only frequency of use, but also clinical relevance, applicability and psychometric validity are important factors to consider [10].
To address the issue of the relevance we calculated the “percentage of significance” for the most frequently used OMIs, defined by Desuter et al. as the percentage of number of studies with a significant change in a specific OMI, divided by total number of studies using this OMI [5]. We found the VHI-30 to be the only OMI with a percentage of significance higher than 80% and the VHI-10, GRBAS, MPT and the APQ to be the only parameters of 50% or more. Interestingly, Desuter et al. found percentages of significance higher than 80% for MPT (90%), mean airflow (86%) and the G of the GRBAS (85%) in his review on unilateral vocal fold paralysis. We hypothesize that this difference may reflect the pathophysiological difference between glottic insufficiency with mobile vocal folds and UVFP, supporting the notion that the relevance of OMIs may differ from disease to disease.
Studies tend to report mainly on the statistical significance of a change in an OMI, which does not necessarily correspond to a difference that is clinically relevant. But for patients and health professionals clinically relevant changes in outcome are of great importance.
Until now, the clinical relevance of a certain outcome has often been consensus based [31]. However, values for clinically relevant changes have been suggested for some of these OMIs. Van Gogh et al. defined what constitutes a clinically relevant change for the VHI-30 based on a selected Dutch population with dysphonia after treatment for early glottic cancer or benign voice disorders and a normal population [65]. More recently Young et al. formulated the MCID (minimal clinically important difference) for VHI-10 in patients with vocal fold paralysis. The authors highlight that not only the numerical change within a parameter that represents a minimal clinically relevant change is important, but also that this value may be disease specific [66]. Therefore, some OMIs may not be as valuable for a specific disease as traditionally assumed.
Applicability, whether a test can be performed or not, depends on logistic, technical and financial possibilities and limitations. For acoustic, aerodynamic, but also endoscopic OMIs this can be a limiting factor. For acoustic measurements special voice program software is needed to record and store a phonetogram, and to extract, calculate and store various voice parameters. These programs are commercially available, f.e. MDVP (multidimensional voice program software, computerized speech laboratory (KayPENTAX, Montvale, NJ)) and have their own set of parameters. For aerodynamic parameters as MFR a pneumotachograph is needed (phonatory aerodynamic system (PAS), KayPENTAX, Montvale, NJ).
The last important factor is psychometric validity. Psychometric validity has been only investigated for subjective OMIs [48, 67]. In the study of Francis et al. 32 PROMs were reviewed on development and validation and showed gross psychometric weaknesses as lack of patient involvement, lack of robust construct validity and lack of interpretability and scaling [67]. Speyer et al. reported on psychometric properties of 15 PROMs and concluded that many psychometric data were missing or indeterminate, VHI seeming to be the most promising questionnaire [48].
This study has some several weaknesses. First of all, no formal Risk of Bias (RoB) was performed. We found this of limited added value, because most studies, 32 out of 34 were cohort studies of which 25 retrospective, with a comparable risk of bias. Of the 2 clinical trials, there was only one double blind RCT, which has a low RoB. Secondly, no formal meta-analysis was performed. As statically significance does not always correspondent with clinically relevancy we chose “percentage of significance” to capture relevancy, although this may not be the most thorough way of doing this. Lastly, we would like to emphasize that the most frequently used OMIs, collected in this review, do not defacto represent the most appropriate OMIs for this patient group, and that besides frequency of use, also clinical relevance, applicability, and psychometric validity are important factors to consider.
Conclusion
In this systematic review we identified the most used OMIs to evaluate treatment effect in patients with non-paralytic glottic insufficiency caused by vocal fold atrophy with and without sulcus as a second step towards developing a COS for this population. The need for a COS is further demonstrated by the fact that studies in this review rely heavily on parameters that have a low percentage of significance in this population, with the exception of VHI-30 with a high percentage of significance of 90%. Future steps in this process will include a quality analysis of the identified OMIs for this specific use and final inclusion through a Delphi process.
References
Dejonckere PH, Bradley P, Clemente P, Cornut G, Crevier-Buchman L, Friedrich G, van de Heyning P, Remacle M, Woisard V (2001) A basic protocol for functional assessment of voice pathology, especially for investigating the efficacy of (phonosurgical) treatments and evaluating new assessment techniques. Guideline elaborated by the Committee on Phoniatrics of the European Laryngolo. Eur Arch Otorhinolaryngol 258(2):77–82
Van den Broek EMJM, Heijnen BJ, Hendriksma M et al (2019) Bilateral trial vocal fold injection with hyaluronic acid in patients with vocal fold atrophy with or without sulcus. Eur Arch Otorhinolaryngol. https://doi.org/10.1007/s00405-019-05347-2
Medeiros N, Castro MEM, van Lith-Bijl JT, Desuter GRR (2022) A systematic review on surgical treatments for sulcus vocalis and vocal fold scar. Laryngoscope 132:822–830. https://doi.org/10.1002/lary.29665
Lechien JR, Geneid A, Bohlender JE et al (2023) Consensus for voice quality assessment in clinical practice: guidelines of the European Laryngological Society and Union of the European Phoniatricians. Eur Arch Otorhinolaryngol 280:5459–5473. https://doi.org/10.1007/s00405-023-08211-6
Desuter G, Dedry M, Schaar B et al (2018) Voice outcome indicators for unilateral vocal fold paralysis surgery: a review of the literature. Eur Arch Otorhinolaryngol 275:459–468. https://doi.org/10.1007/s00405-017-4844-9
Desuter G, Dedry M, Schaar B et al (2019) Voice outcome indicators for unilateral vocal fold paralysis surgery: a survey among surgeons. Eur Ann Otorhinolaryngol Head Neck Dis 136:343–347. https://doi.org/10.1016/j.anorl.2018.07.009
Prinsen CAC, Mokkink LB, Bouter LM et al (2018) COSMIN guideline for systematic reviews of patient-reported outcome measures. Qual Life Res 27:1147–1157. https://doi.org/10.1007/s11136-018-1798-3
Mokkink LB, de Vet HCW, Prinsen CAC et al (2018) COSMIN Risk of Bias checklist for systematic reviews of Patient-Reported Outcome Measures. Qual Life Res 27:1171–1179. https://doi.org/10.1007/s11136-017-1765-4
Terwee CB, Prinsen CAC, Chiarotto A et al (2018) COSMIN methodology for evaluating the content validity of patient-reported outcome measures: a Delphi study. Qual Life Res 27:1159–1170. https://doi.org/10.1007/s11136-018-1829-0
Prinsen CAC, Vohra S, Rose MR et al (2016) How to select outcome measurement instruments for outcomes included in a “Core Outcome Set”—a practical guideline. Trials 17:449. https://doi.org/10.1186/s13063-016-1555-2
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA Statement. Open Med 3:e123-130
Bick E, Dumberger LD, Farquhar DR et al (2020) Does voice therapy improve vocal outcomes in vocal fold atrophy? Ann Otol Rhinol Laryngol 130:602–608. https://doi.org/10.1177/0003489420952464
Desjardins M, Halstead L, Simpson A et al (2022) Respiratory muscle strength training to improve vocal function in patients with presbyphonia. J Voice 36:344–360. https://doi.org/10.1016/j.jvoice.2020.06.006
Andreadis K, Hoffman K, D’Angelo D, Sulica L (2020) Sulcus vocalis: results of excision without reconstruction. Laryngoscope 130:2208–2212. https://doi.org/10.1002/lary.28378
Okui A, Konomi U, Kanazawa T et al (2020) Therapeutic efficacy of basic fibroblast growth factor in patients with vocal fold atrophy. Laryngoscope 130:2847–2852. https://doi.org/10.1002/lary.28541
Ma Y, Long J, Amin MR et al (2020) Autologous fibroblasts for vocal scars and age-related atrophy: a randomized clinical trial. Laryngoscope 130:2650–2658. https://doi.org/10.1002/lary.28453
van den Broek EMJM, Heijnen BJ, Hendriksma M et al (2020) Bilateral medialization thyroplasty in patients with vocal fold atrophy with or without sulcus. Eur Arch Otorhinolaryngol 277:2023–2029. https://doi.org/10.1007/s00405-020-05933-9
Hu H-C, Hung Y-T, Lin S-Y et al (2019) Office-based autologous fat injection laryngoplasty for glottic insufficiency in patients under 50 years old. J Voice 33:747–750. https://doi.org/10.1016/j.jvoice.2018.03.012
Allensworth JJ, O’Dell K, Ziegler A et al (2019) Treatment outcomes of bilateral medialization thyroplasty for presbylaryngis. J Voice 33:40–44. https://doi.org/10.1016/j.jvoice.2017.10.014
González-Herranz R, Hernandez García E, Granda-Rosales M et al (2019) Improved mucosal wave in unilateral autologous temporal fascia graft in sulcus vocalis type 2 and vocal scars. J Voice 33:915–922. https://doi.org/10.1016/j.jvoice.2018.06.013
Park YM, Lim JY, Kang MS, Choi HS (2019) Treatment Outcomes of Angiolytic Laser-Assisted Glottoplasty in Patients With Sulcus Vocalis. Annals of Otology, Rhinology and Laryngology 128:377–383. https://doi.org/10.1177/0003489419826132
van den Broek EMJM, Heijnen BJ, Hendriksma M et al (2019) Bilateral vocal fold injection with autologous fat in patients with vocal fold atrophy with or without sulcus. Eur Arch Otorhinolaryngol. https://doi.org/10.1007/s00405-019-05479-5
Miaśkiewicz B, Szkiełkowska A, Gos E et al (2018) Pathological sulcus vocalis: treatment approaches and voice outcomes in 36 patients. Eur Arch Otorhinolaryngol 275:2763–2771. https://doi.org/10.1007/s00405-018-5040-2
Takeharu K, Kurakami K, Konomi U et al (2018) Safety and short-term outcomes of basic fibroblast growth factor injection for sulcus vocalis. Acta Otolaryngol 138:1014–1019. https://doi.org/10.1080/00016489.2018.1497808
Kaneko M, Hirano S, Tateya I et al (2015) Multidimensional analysis on the effect of vocal function exercises on aged vocal fold atrophy. J Voice 29:638–644. https://doi.org/10.1016/j.jvoice.2014.10.017
Miaśkiewicz B, Szkiełkowska A, Piłka A, Skarżyński H (2015) Results of surgical treatment in patients with sulcus vocalis. Otolaryngol Pol 69:7–14. https://doi.org/10.5604/00306657.1182613
Young VN, Gartner-Schmidt J, Rosen CA (2015) Comparison of voice outcomes after trial and long-term vocal fold augmentation in vocal fold atrophy. Laryngoscope 125:934–940. https://doi.org/10.1002/lary.25043
Hwang CS, Lee HJ, Ha JG et al (2013) Use of pulsed dye laser in the treatment of sulcus vocalis. Otolaryngol Head Neck Surg 148:804–809. https://doi.org/10.1177/0194599813479909
Yilmaz T (2012) Sulcus vocalis: Excision, primary suture and medialization laryngoplasty: Personal experience with 44 cases. European Archives of Oto-Rhino-Laryngology 269:2381–2389. https://doi.org/10.1007/s00405-012-2058-8 LK - https://utrechtuniversity.on.worldcat.org/atoztitles/link?sid=EMBASE&sid=EMBASE&issn=09374477&id=10.1007%2Fs00405-012-2058-8&atitle=Sulcus+vocalis%3A+Excision%2C+primary+suture+and+medialization+laryngoplasty%3A+Personal+experience+with+44+cases&stitle=Eur.+Arch.+Oto-Rhino-Laryngol.&title=European+Archives+of+Oto-Rhino-Laryngology&volume=269&issue=11&spage=2381&epage=2389&aulast=Yilmaz&aufirst=Taner&auinit=T.&aufull=Yilmaz+T.&coden=EAOTE&isbn=&pages=2381-2389&
Hirano S, Tateya I, Kishimoto Y et al (2012) Clinical trial of regeneration of aged vocal folds with growth factor therapy. Laryngoscope 122:327–331. https://doi.org/10.1002/lary.22393
Gartner-Schmidt J, Rosen C (2011) Treatment success for age-related vocal fold atrophy. Laryngoscope 121:585–589. https://doi.org/10.1002/lary.21122
Mau T, Jacobson BH, Garrett CG (2010) Factors associated with voice therapy outcomes in the treatment of presbyphonia. Laryngoscope 120:1181–1187. https://doi.org/10.1002/lary.20890
Zhang F, Sprecher AJ, Wei C, Jiang JJ (2010) Implantation of gelatin sponge combined with injection of autologous fat for sulcus vocalis. Otolaryngol Head Neck Surg 143:198–203. https://doi.org/10.1016/j.otohns.2010.03.002
Pinto JA, da Silva Freitas MLA, Carpes AF et al (2007) Autologous grafts for treatment of vocal sulcus and atrophy. Otolaryngol Head Neck Surg 137:785–791. https://doi.org/10.1016/j.otohns.2007.05.059
Hsiung M-W, Pai L (2006) Autogenous fat injection for glottic insufficiency: analysis of 101 cases and correlation with patients’ self-assessment. Acta Otolaryngol 126:191–196. https://doi.org/10.1080/00016480500339854
Tsunoda K, Kondou K, Kaga K et al (2005) Autologous transplantation of fascia into the vocal fold: long-term result of type-1 transplantation and the future. Laryngoscope 115:1–10. https://doi.org/10.1097/01.mlg.0000183966.72921.31
Hsiung M-W, Kang B-H, Pai L et al (2004) Combination of fascia transplantation and fat injection into the vocal fold for sulcus vocalis: long-term results. Ann Otol Rhinol Laryngol 113:359–366. https://doi.org/10.1177/000348940411300504
Su C-Y, Tsai S-S, Chiu J-F, Cheng C-A (2004) Medialization laryngoplasty with strap muscle transposition for vocal fold atrophy with or without sulcus vocalis. Laryngoscope 114:1106–1112. https://doi.org/10.1097/00005537-200406000-00028
Hsiung M-W, Lin Y-S, Su W-F, Wang H-W (2003) Autogenous fat injection for vocal fold atrophy. Eur Arch Otorhinolaryngol 260:469–474. https://doi.org/10.1007/s00405-003-0622-y
Chen Y-Y, Pai L, Lin Y-S et al (2003) Fat augmentation for nonparalytic glottic insufficiency. ORL 65:176–183. https://doi.org/10.1159/000072256
Hsiung M-W, Chen Y-Y, Pai L et al (2003) Results of magnetic resonance imaging assessment, acoustic analysis, phonatory function and perceptual rating of glottic insufficiency before and after fat augmentation: correlated with subjective rating. ORL J Otorhinolaryngol Relat Spec 65:76–83. https://doi.org/10.1159/000070770
Ramig LO, Gray S, Baker K et al (2001) The aging voice: a review, treatment data and familial and genetic perspectives. Folia Phoniatr Logop 53:252–265. https://doi.org/10.1159/000052680
Remacle M, Lawson G, Degols JC et al (2000) Microsurgery of sulcus vergeture with carbon dioxide laser and injectable collagen. Ann Otol Rhinol Laryngol 109:141–148. https://doi.org/10.1177/000348940010900206
Pontes P, Behlau M (1993) Treatment of sulcus vocalis: Auditory perceptual and acoustical analysis of the slicing mucosa surgical technique. J Voice 7:365–376. https://doi.org/10.1016/S0892-1997(05)80260-7
Carroll TL, Rosen CA (2010) Trial vocal fold injection. J Voice 24:494–498. https://doi.org/10.1016/j.jvoice.2008.11.001
Carroll TL, Faudoa E, Van Doren M (2019) Evaluation of a shorter follow-up time to capture benefit of a trial vocal fold augmentation. J Voice 33:169–175. https://doi.org/10.1016/j.jvoice.2017.10.017
Gilbert MR, Gartner-Schmidt JL, Rosen CA (2017) The VHI-10 and VHI item reduction translations—are we all speaking the same language? J Voice 31:250.e1-250.e7
Speyer R, Kim JH, Doma K et al (2019) Measurement properties of self-report questionnaires on health-related quality of life and functional health status in dysphonia: a systematic review using the COSMIN taxonomy. Qual Life Res 28:283–296
Bach KK, Belafsky PC, Wasylik K, Postma GN, Koufman JA (2005) Validity and reliability of the glottal function index. Arch Otolaryngol Head Neck Surg 131:961–964
Nanjundeswaran C, Jacobson BH, Gartner-Schmidt J, Verdolini Abbott K (2015) Vocal Fatigue Index (VFI): development and validation. J Voice 29:433–440. https://doi.org/10.1016/j.jvoice.2014.09.012
Paolillo NP, Pantaleo G (2015) Development and validation of the voice fatigue handicap questionnaire (VFHQ): clinical, psychometric, and psychosocial facets. J Voice 29:91–100. https://doi.org/10.1016/j.jvoice.2014.05.010
Rabin R, de Charro F (2001) EQ-5D: a measure of health status from the EuroQol Group. Ann Med 33:337–343. https://doi.org/10.3109/07853890109002087
Roussel NC, Lobdell M (2006) The clinical utility of the soft phonation index. Clin Linguist Phon 20:181–186. https://doi.org/10.1080/02699200400026942
Mathew MM, Bhat JS (2009) Soft phonation index - a sensitive parameter? Indian J Otolaryngol Head Neck Surg 61:127–130. https://doi.org/10.1007/s12070-009-0050-4
Speyer R, Bogaardt HCA, Passos VL et al (2010) Maximum phonation time: variability and reliability. J Voice 24:281–284. https://doi.org/10.1016/j.jvoice.2008.10.004
Baken RJ OR (2000) Clinical measurement of speech and voice. Wien
Zhuang P, Sprecher AJ, Hoffman MR et al (2009) Phonation threshold flow measurements in normal and pathological phonation. Laryngoscope 119:811–815. https://doi.org/10.1002/lary.20165
Jen JH, Chan RW, Wu CH, Te WC (2021) Phonation threshold pressure/flow for reflecting glottal closure in unilateral vocal fold paralysis. Laryngoscope 131:E1598–E1604. https://doi.org/10.1002/lary.29274
Plexico LW, Sandage MJ, Faver KY (2011) Assessment of phonation threshold pressure: a critical review and clinical implications. Am J Speech Lang Pathol 20:348–366
Brunings JW, Vanbelle S, Akkermans A et al (2018) Observer agreement for measurements in videolaryngostroboscopy. J Voice 32:756–762. https://doi.org/10.1016/j.jvoice.2017.09.005
Poburka BJ, Patel RR, Bless DM (2017) Voice-vibratory assessment with laryngeal imaging (VALI) form: reliability of rating stroboscopy and high-speed videoendoscopy. J Voice 31:513.e1-513.e14. https://doi.org/10.1016/j.jvoice.2016.12.003
Carroll TL, Wu YHE, McRay M, Gherson S (2012) Frame by frame analysis of glottic insufficiency using laryngovideostroboscopy. J Voice 26:220–225. https://doi.org/10.1016/j.jvoice.2011.01.008
Santos M, Sousa CA, Freitas SV et al (2022) Presbylarynx: how easy is it to recognize the aging signs? A prospective study with inter- and intra-rater reliability in laryngoscopic evaluation. J Voice. https://doi.org/10.1016/j.jvoice.2022.04.004
Santos M, Freitas SV, Almeida e Sousa C, da Silva ÁM, (2023) Presbylarynx: validation of a classification based on morphological characteristics. Eur Arch Otorhinolaryngol 280:781–788. https://doi.org/10.1007/s00405-022-07618-x
Van Gogh CDL, Mahieu HF, Kuik DJ et al (2007) Voice in early glottic cancer compared to benign voice pathology. Eur Arch Otorhinolaryngol 264:1033–1038. https://doi.org/10.1007/s00405-007-0313-1
Young VVN, Jeong K, Rothenberger SD et al (2018) Minimal clinically important difference of voice handicap index-10 in vocal fold paralysis. Laryngoscope 128:1419–1424. https://doi.org/10.1002/lary.27001
Francis DO, Daniero JJ, Hovis KL et al (2017) Voice-related patient-reported outcome measures: A systematic review of instrument development and validation. J Speech Lang Hear Res 60:62–88
Acknowledgements
We would like to thank Dr Stegeman, epidemiologist UMC Utrecht, for her value attributions in this review and its discussion.
Funding
No funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
No conflict of Interest.
Ethical approval
Not applicable.
Informed consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
van den Broek, E.M.J.M., Mes, S.D., Heijnen, B.J. et al. Glottic insufficiency caused by vocal fold atrophy with or without sulcus: systematic review of outcome measurements. Eur Arch Otorhinolaryngol (2024). https://doi.org/10.1007/s00405-024-08751-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00405-024-08751-5