Abstract
Purpose
There is no standard definition of disease-free interval before local recurrence after treatment in head and neck carcinoma (HNSCC). We evaluated an easy-to-use stratification and its association with survival in a large cohort of patients.
Methods
We performed a retrospective cohort analysis of prognostic variables in 325 HNSCC patients with a local recurrence after definitive radiotherapy or concurrent chemoradiotherapy. Endpoints were overall survival (OS) and post-recurrence survival (PRS).
Results
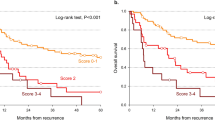
Variables associated with the survival were the patient age (OS p < 0.0001, PRS p < 0.0001), the initial disease stage (OS p = 0.24, PRS p = 0.0358), localization (OS p = 0.012, PRS p = 0.0002), a complete initial response to treatment (OS p < 0.0001, PRS p = 0.019), synchronous regional or distant metastatic disease (OS p = 0.0094, PRS p < 0.0001), a salvage surgery (OS p < 0.0001, PRS p < 0.0001) and time to recurrence (OS p = 0.0002, PRS p = 0.0029). Time to recurrence could be stratified between specific prognostic time categories that comprised disease persistence, early recurrence (< 12 months), standard recurrence (12 months–5 years) and late recurrence (> 5 years).
Conclusion
In HNSCC patients, time to local recurrence is a prognostic variable that can be defined using an easy-to-use stratification.



Similar content being viewed by others
References
NIH. Understanding recurrence: what is cancer recurrence? https://www.cancer.org/treatment/survivorship-during-and-after-treatment/understanding-recurrence/what-is-cancer-recurrence.html. Accessed 4 sept 2020
Cohen EE, LaMonte SJ, Erb NL, Beckman KL, Sadeghi N, Hutcheson KA et al (2016) American Cancer Society head and neck cancer survivorship care guideline. CA Cancer J Clin 66:203–239. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/clinical-trial-endpoints-approvalcancer-drugs-and-biologics. Accessed 4 sept 2020
FDA (2007) Guidance for industry clinical trial endpoints for the approval of cancer drugs and biologics
Ulm K (2011) Primary endpoints in cancer trials. J Thorac Dis 3:82–83
Fiteni F, Westeel V, Pivot X, Borg C, Vernerey D, Bonnetain F (2014) Endpoints in cancer clinical trials. J Visc Surg 151:17–22
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R et al (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45:228–247
Rohde M, Rosenberg T, Pareek M, Nankivell P, Sharma N, Mehanna H et al (2020) Definition of locally recurrent head and neck squamous cell carcinoma: a systematic review and proposal for the Odense-Birmingham definition. Eur Arch Otorhinolaryngol 277:1593–1599
Savina M, Gourgou S, Italiano A, Dinart D, Rondeau V, Penel N et al (2018) Meta-analyses evaluating surrogate endpoints for overall survival in cancer randomized trials: a critical review. Crit Rev Oncol Hematol 123:21–41
Hamoir M, Holvoet E, Ambroise J, Lengele B, Schmitz S (2017) Salvage surgery in recurrent head and neck squamous cell carcinoma: oncologic outcome and predictors of disease free survival. Oral Oncol 67:1–9
Chang JH, Wu CC, Yuan KS, Wu ATH, Wu SY (2017) Locoregionally recurrent head and neck squamous cell carcinoma: incidence, survival, prognostic factors, and treatment outcomes. Oncotarget 8:55600–55612
Tan HK, Giger R, Auperin A, Bourhis J, Janot F, Temam S (2010) Salvage surgery after concomitant chemoradiation in head and neck squamous cell carcinomas - stratification for postsalvage survival. Head Neck 32:139–147
Rasmussen JH, Gronhoj C, Hakansson K, Friborg J, Andersen E, Lelkaitis G et al (2019) Risk profiling based on p16 and HPV DNA more accurately predicts location of disease relapse in patients with oropharyngeal squamous cell carcinoma. Ann Oncol 30:629–636
Forastiere AA, Goepfert H, Maor M, Pajak TF, Weber R, Morrison W et al (2003) Concurrent chemotherapy and radiotherapy for organ preservation in advanced laryngeal cancer. N Engl J Med 349:2091–2098
Sandulache VC, Vandelaar LJ, Skinner HD, Cata J, Hutcheson K, Fuller CD et al (2016) Salvage total laryngectomy after external-beam radiotherapy: a 20-year experience. Head Neck 38(Suppl 1):E1962–E1968
Gorphe P, Tao Y, Blanchard P, Even C, Moya-Plana A, Mirghani H et al (2017) Relationship between the time to locoregional recurrence and survival in laryngeal squamous-cell carcinoma. Eur Arch Otorhinolaryngol 274:2267–2271
Caula A, Boukhris M, Guerlain J, Tao Y, Breuskin I, Mirghani H et al (2019) Correlation between the duration of locoregional control and survival in T1–T2 oropharyngeal cancer patients. Eur Arch Otorhinolaryngol 276:1161–1166
Zafereo ME, Hanasono MM, Rosenthal DI, Sturgis EM, Lewin JS, Roberts DB et al (2009) The role of salvage surgery in patients with recurrent squamous cell carcinoma of the oropharynx. Cancer 115:5723–5733
NIH. NCI dictionaries: relapse-free survival. Accessed 4 sept 2020
Kemp R, Prasad V (2017) Surrogate endpoints in oncology: when are they acceptable for regulatory and clinical decisions, and are they currently overused? BMC Med 15:134
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to disclose.
Ethical approval
This study received approval from the local Research Ethics Committee, in accordance with the World Medical Association—Declaration of Helsinki—ethical principles for medical research and with the national regulatory laws.
Research involving human participants and/or animals
No.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gorphe, P., Moya-Plana, A., Guerlain, J. et al. Disease-free time stratification in locally recurrent head and neck carcinoma after definitive radiotherapy or chemoradiotherapy. Eur Arch Otorhinolaryngol 279, 3063–3069 (2022). https://doi.org/10.1007/s00405-021-07116-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-021-07116-6