Abstract
Purpose
This study aimed to investigate the effects of activating transcription factor-5 (ATF5) on nasopharyngeal carcinoma (NPC) cell radioresistance.
Methods
HONE-1 nasopharyngeal carcinoma cells were irradiated by conventional fractionation to generate HONE-1R radiotherapy-resistant cells. Short hairpin RNA (shRNA) expression plasmids targeting the ATF5 gene were constructed and transfected into the HONE-1R cell line. The proliferation assay, colony formation analysis, Transwell Boyden chamber assay and other experimental methods were performed to verify changes in the radiosensitivity and other biological of NPC cells.
Results
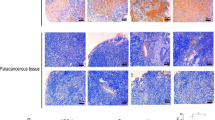
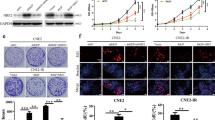
X-ray irradiation significantly promoted the upregulation of ATF5 protein levels in HONE-1 cells, and the protein expression of ATF5 increased with the dose of X-ray irradiation (p < 0.05). The colony formation rate, cell survival rate and migration ability of HONE-1R cells were significantly higher than those of HONE-1 cells (p < 0.05). Simultaneously, X-ray could also promote the morphology of epithelial-mesenchymal transition (EMT) in HONE-1 cells, inducing a lower expression level of E-cadherin and a higher expression level of N-cadherin in a dose-dependent manner (p < 0.05). Inhibiting ATF5 significantly reduced the colony formation rate, cell survival rate, migration and invasiveness of HONE-1R cells (p < 0.05). Moreover, sensitizing HONE-1R cells to X-ray irradiation significantly upregulated the expression of E-cadherin and downregulated the expression of N-cadherin in these cells (p < 0.05).
Conclusions
ATF5 may be a potential therapeutic target to improve radiosensitivity in NPC.





Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Change history
19 May 2020
The article ATF5 involved in radioresistance in nasopharyngeal carcinoma by promoting epithelial-to-mesenchymal phenotype transition, written by Yu Shuai, Erxi Fan, Qiuyue Zhong, Guangyong Feng, Qiying Chen, Xiaoxia Gou, Guihai Zhang, was originally published.
Abbreviations
- NPC:
-
Nasopharyngeal carcinoma
- ATF5:
-
Activating transcription factor-5
- DAB:
-
Diaminobenzidine
- shRNA:
-
Short hairpin RNA
- DAPI:
-
4′-6-Diamidino-2-phenylindole
- FBS:
-
Foetal bovine serum
- IMRT:
-
Intensity-modulated radiation therapy
- EMT:
-
Epithelial-mesenchymal transition
- MRLC:
-
Myosin regulatory light chain
References
Chua MLK, Wee JTS, Hui EP, Chan ATC (2016) Nasopharyngeal carcinoma. Lancet (Lond, Engl) 387(10022):1012–1024. https://doi.org/10.1016/s0140-6736(15)00055-0
Cui Z, Zhao Y (2019) microRNA-342-3p targets FOXQ1 to suppress the aggressive phenotype of nasopharyngeal carcinoma cells. BMC Cancer 19(1):104. https://doi.org/10.1186/s12885-018-5225-5
Huang J, Kong FF, Oei RW, Zhai RP, Hu CS, Ying HM (2019) Dosimetric predictors of temporal lobe injury after intensity-modulated radiotherapy for T4 nasopharyngeal carcinoma: a competing risk study. Radiat Oncol 14(1):31. https://doi.org/10.1186/s13014-019-1229-9
Kong F, Ying H, Du C, Huang S, Zhou J, Chen J, Sun L, Chen X, Hu C (2014) Patterns of local-regional failure after primary intensity modulated radiotherapy for nasopharyngeal carcinoma. Radiat Oncol 9:60. https://doi.org/10.1186/1748-717x-9-60
Chen L, Zhang Y, Lai SZ, Li WF, Hu WH, Sun R, Liu LZ, Zhang F, Peng H, Du XJ, Lin AH, Sun Y, Ma J (2019) 10-year results of therapeutic ratio by intensity-modulated radiotherapy versus two-dimensional radiotherapy in patients with nasopharyngeal carcinoma. Oncologist 24(1):e38–e45. https://doi.org/10.1634/theoncologist.2017-0577
Wang L, Guo Y, Xu J, Chen Z, Jiang X, Zhang L, Huang S, He X, Zhang Y (2017) Clinical analysis of recurrence patterns in patients with nasopharyngeal carcinoma treated with intensity-modulated radiotherapy. Ann Otol Rhinol Laryngol 126(12):789–797. https://doi.org/10.1177/0003489417734229
Lee V, Kwong D, Leung TW, Lam KO, Tong CC, Lee A (2017) Palliative systemic therapy for recurrent or metastatic nasopharyngeal carcinoma - How far have we achieved? Crit Rev Oncol/Hematol 114:13–23. https://doi.org/10.1016/j.critrevonc.2017.03.030
Melber A, Haynes CM (2018) UPR(mt) regulation and output: a stress response mediated by mitochondrial-nuclear communication. Cell Res 28(3):281–295. https://doi.org/10.1038/cr.2018.16
Deng P, Haynes CM (2017) Mitochondrial dysfunction in cancer: Potential roles of ATF5 and the mitochondrial UPR. J Cell Biol 47:43–49. https://doi.org/10.1083/jcb.201702058
Juliana CA, Yang J, Rozo AV, Good A, Groff DN, Wang SZ, Green MR, Stoffers DA (2017) ATF5 regulates beta-cell survival during stress. Proc Natl Acad Sci USA 114(6):1341–1346. https://doi.org/10.1073/pnas.1620705114
Feldheim J, Kessler AF, Schmitt D, Wilczek L, Linsenmann T, Dahlmann M, Monoranu CM, Ernestus RI, Hagemann C, Lohr M (2018) Expression of activating transcription factor 5 (ATF5) is increased in astrocytomas of different WHO grades and correlates with survival of glioblastoma patients. OncoTargets Therapy 11:8673–8684. https://doi.org/10.2147/ott.s176549
Nishioka T, Miyai Y, Haga H, Kawabata K, Shirato H, Homma A, Shibata K, Yasuda M (2009) Novel function of transcription factor ATF5: blockade of p53-dependent apoptosis induced by ionizing irradiation. Cell Struct Funct 34(1):17–22
Ishihara S, Yasuda M, Ishizu A, Ishikawa M, Shirato H, Haga H (2015) Activating transcription factor 5 enhances radioresistance and malignancy in cancer cells. Oncotarget 6(7):4602–4614. https://doi.org/10.18632/oncotarget.2912
Zhang H, Luo H, Jiang Z, Yue J, Hou Q, Xie R, Wu S (2016) Fractionated irradiation-induced EMT-like phenotype conferred radioresistance in esophageal squamous cell carcinoma. J Radiat Res 57(4):370–380. https://doi.org/10.1093/jrr/rrw030
Qu S, Liang ZG, Zhu XD (2015) Advances and challenges in intensity-modulated radiotherapy for nasopharyngeal carcinoma. APJCP 16(5):1687–1692
Shan GP, Wang BB, Zheng P, Du FL, Yang YW (2017) Efficacy and safety of chemotherapy combined with stereotactic radiotherapy in the treatment of nasopharyngeal carcinoma. Med Sci Monit 23:5630–5636
Peng L, Liu JQ, Xu C, Huang XD, Tang LL, Chen YP, Sun Y, Ma J (2019) The prolonged interval between induction chemotherapy and radiotherapy is associated with poor prognosis in patients with nasopharyngeal carcinoma. Radiat Oncol 14(1):9. https://doi.org/10.1186/s13014-019-1213-4
Wang X, Hu M, Xing F, Wang M, Wang B, Qian D (2017) Human cytomegalovirus infection promotes the stemness of U251 glioma cells. J Med Virol 89(5):878–886. https://doi.org/10.1002/jmv.24708
Jin Y, Xu K, Chen Q, Wang B, Pan J, Huang S, Wei Y, Ma H (2018) Simvastatin inhibits the development of radioresistant esophageal cancer cells by increasing the radiosensitivity and reversing EMT process via the PTEN-PI3K/AKT pathway. Exp Cell Res 362(2):362–369. https://doi.org/10.1016/j.yexcr.2017.11.037
Chen Y, Liu W, Wang P, Hou H, Liu N, Gong L, Wang Y, Ji K, Zhao L, Wang P (2016) Halofuginone inhibits radiotherapy-induced epithelial-mesenchymal transition in lung cancer. Oncotarget 7(44):71341–71352. https://doi.org/10.18632/oncotarget.11217
Theys J, Jutten B, Habets R, Paesmans K, Groot AJ, Lambin P, Wouters BG, Lammering G, Vooijs M (2011) E-Cadherin loss associated with EMT promotes radioresistance in human tumor cells. Radiother Oncol 99(3):392–397. https://doi.org/10.1016/j.radonc.2011.05.044
Lamouille S, Xu J, Derynck R (2014) Molecular mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell Biol 15(3):178–196. https://doi.org/10.1038/nrm3758
Ben-Shmuel S, Rashed R, Rostoker R, Isakov E, Shen-Orr Z, LeRoith D (2017) Activating transcription factor-5 knockdown reduces aggressiveness of mammary tumor cells and attenuates mammary tumor growth. Front Endocrinol (Lausanne) 8:173
Nukuda A, Endoh H, Yasuda M, Mizutani T, Kawabata K, Haga H (2016) Role of ATF5 in the invasive potential of diverse human cancer cell lines. Biochem Biophys Res Commun 474(3):509–514. https://doi.org/10.1016/j.bbrc.2016.04.131
Hynes RO (2002) Integrins: bidirectional, allosteric signaling machines. Cell 110(6):673–687. https://doi.org/10.1016/s0092-8674(02)00971-6
Mizutani T, Haga H, Koyama Y, Takahashi M, Kawabata K (2006) Diphosphorylation of the myosin regulatory light chain enhances the tension acting on stress fibers in fibroblasts. J Cell Physiol 209(3):726–731. https://doi.org/10.1002/jcp.20773
Acknowledgements
This work was supported by the National Natural Science Foundation funding Project.
Funding
National Natural Science Foundation funding Project (Number: 81260370).
Author information
Authors and Affiliations
Contributions
Guihai developed and planned the study. Yu, Erxi and Qiuyue performed the irradiation and were responsible for management of the cells. Yu, Erxi and Guangyong performed the experiments and Analyzed the data. All authors participated drafted the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interest.
Ethical approval
Ethical approval this study was approved by the Affiliated Hospital of Zunyi Medical University institutional review board (IRB).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original version of this article was revised due to the retrospective open access cancellation request.
The original version of this article was revised due to retrospective open access cancellation order.
Rights and permissions
About this article
Cite this article
Shuai, Y., Fan, E., Zhong, Q. et al. ATF5 involved in radioresistance in nasopharyngeal carcinoma by promoting epithelial-to-mesenchymal phenotype transition. Eur Arch Otorhinolaryngol 277, 2869–2879 (2020). https://doi.org/10.1007/s00405-020-05996-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-020-05996-8