Abstract
Importance
The formation of adhesions after gynecological surgery not only has detrimental impacts on those affected, including pain, obstruction, and infertility, but also imposes a high economic burden on healthcare systems worldwide.
Objective
The aim of this review was to evaluate the adhesion prevention potential of all currently available adhesion barriers for gynecological surgery.
Evidence acquisition
We systematically searched MEDLINE and CENTRAL databases for randomized controlled trials (RCTs) on the use of adhesion barriers as compared with peritoneal irrigation or no treatment in gynecological surgery. Only RCTs with second-look surgery to evaluate adhesions in the pelvic/abdominal (but not intrauterine) cavity were included.
Results
We included 45 RCTs with a total of 4,120 patients examining a total of 10 unique types of barriers in second-look gynecological surgery. While RCTs on oxidized regenerated cellulose (significant improvement in 6 of 14 trials), polyethylene glycol with/without other agents (4/10), hyaluronic acid and hyaluronate + carboxymethylcellulose (7/10), icodextrin (1/3), dextran (0/3), fibrin-containing agents (1/2), expanded polytetrafluoroethylene (1/1), N,O-carboxymethylchitosan (0/1), and modified starch (1/1) overall showed inconsistent findings, results for expanded polytetrafluoroethylene, hyaluronic acid, and modified starch yielded the greatest improvements regarding adhesion reduction at 75%, 0–67%, and 85%, respectively.
Conclusions and relevance
Best results for adhesion prevention were reported after applying Gore-Tex Surgical Membrane, hyaluronic acid, and 4DryField®. As Gore-Tex Surgical Membrane is nonabsorbable, it is associated with a greater risk of new adhesion formation due to second-look surgery to remove the product. 4DryField® yielded the greatest improvement in adhesion score compared to all other barrier agents (85%). For better comparability, future studies should use standardized scores and put more emphasis on patient-reported outcome measures, such as pain and infertility.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Peritoneal adhesion formation after gynecological surgery
Adhesions develop as a natural response of the peritoneum to surgical tissue trauma induced by injury (curettage), infection, radiation, ischemia, desiccation, or foreign-body reaction [1]. Indeed, intra-abdominal adhesions form after 50–100% of all abdominal surgeries [2] and after 60–90% of all gynecological procedures [3]. Tissue challenged by trauma is often characterized by decreased fibrinolytic activity, allowing fibrinous adhesions to develop into permanent, vascularized adhesions after fibroblast invasion [1]. Peritoneal adhesions frequently form after gynecological surgery, for example, ovarian cystectomy, resection of endometriosis, tubal surgery, myomectomy, and others [4]. While the formation of adhesions due to surgical intervention has been clearly established, the additional negative effect of dry CO2 and the intra-abdominal pressure are still being discussed controversially [5]. Peritoneal adhesions can cause great suffering in patients, mostly including pain, obstruction, and secondary infertility, as well as imposing a significant financial burden on healthcare systems [5, 6]. Surprisingly, adhesion prophylaxis has still not been adequately addressed, for example, during endometriosis surgery, and financing effective prophylaxis through healthcare systems is underrepresented although it would be crucial to achieving a satisfying outcome in the long term [7]. This might be due to scarce data on patient-reported outcomes and, therefore, a lack of data for analyzing the cost-effectiveness.
Strategies to prevent formation of peritoneal adhesions
The exact mechanism underlying the formation of adhesions is still not completely understood. Therefore, aside from avoiding surgery, causal treatment is not available. Factors that seem to be associated with adhesion formation are use of CO2, level of intra-abdominal pressure, genetic factors, and traumatizing surgical techniques [8]. In addition, it has been shown in animal models that humidified CO2 at body temperature may prevent adhesion formation [9]. Indeed, further studies are needed to fully understand all the factors relevant to adhesion formation.
When surgery cannot be avoided, strategies for adhesion prophylaxis should include both improved surgical techniques, such as minimally invasive incisions, as well as the use of adhesion prophylactic agents and devices [7]. Laparoscopic approaches, for instance, cause less injury to the peritoneum than laparotomic approaches and therefore are associated with decreased adhesion formation [10]. Pharmaceutical approaches to prevent adhesions from developing include steroids (anti-inflammatory effect), heparin (anti-coagulatory effect), tissue-plasminogen activator (fibrinolysis), and promethazine (anti-inflammatory effect). All these molecules interfere with crucial pathways and responses during adhesion formation. Unfortunately, though, they have not been as effective as expected [11]. In contrast, the use of adhesion barriers in gynecological surgery showed promising results in randomized studies [6]. Nevertheless, the comprehensive use is not common sense due to extra costs and possible side effects.
In this review, we evaluated the effect of different barrier approaches to prevent postoperative adhesions in women after gynecological surgery using a systematic literature search of randomized controlled trials (RCTs) including second-look operations, which provide the highest level of evidence.
Methods
We searched MEDLINE and CENTRAL (Cochrane Central Register of Controlled Trials) databases in June 2023 for RCTs on the use of agents to prevent adhesions compared with flushing or no such preventive treatment in women undergoing gynecological surgery. The body of literature addressing anti-adhesive agents is extensive and the number of commercially available barriers is increasing; however, the data often lack efficacy outcome evaluated by second-look surgery. In this review, we therefore only included RCTs with second-look surgery for adhesion evaluation. Studies on intrauterine adhesions were excluded. We employed the following search strategy for MEDLINE: (second-look OR 2nd look OR reintervention OR reoperation OR endometr* OR ovar* OR laparosc* OR laparotom* OR gynec* OR fallop* OR salping*) AND adhes* AND (randomized controlled trial[Publication Type] OR randomized[Title/Abstract] OR placebo[Title/Abstract]) and the following for CENTRAL: randomized controlled trial in Abstract AND second-look OR 2nd look OR reintervention OR reoperation OR endometr* OR ovar* OT laparosc* OR gynec* OR fallop* OR salping* in Title Abstract Keyword AND Journal article in Publication Type.
Two authors (SS and BK) independently assessed the identified literature, based on set criteria to minimize the risk of bias. Data were extracted independently by two authors (IA, IMH) according to the study protocol that was prospectively registered in PROSPERO (international prospective register of systematic reviews) with the ID CRD42023428551. Inconsistencies were identified and resolved under the direction of the lead author (SS). Results were reported according to the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) statement [12]. Similar review articles were screened to assure the completeness of the search results.
Whenever possible, American Fertility Society (AFS) adhesion scores were used as an effect measure to assess the potential of the barriers. Using AFS scores, the different barrier devices can be directly compared. When AFS scores were not available, other scores were considered. To further enhance comparability, absolute scores at second-look surgery were chosen whenever available as they are more commonly used than the change in scores between the interventions. When no score was available, other measures such as adhesion incidence were applied. The outcome measure “adhesion-free patients” was considered to have the lowest explanatory power.
Results
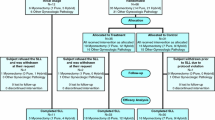
The systematic search of MEDLINE and CENTRAL identified 1237 unique articles. A total of 1192 articles were excluded after reviewing the titles and abstracts for the following reasons: no adhesion barrier or no evaluation of adhesion prevention, nongynecological study, nonhuman study, intrauterine adhesions, no RCT, no second-look surgery, study not in English language, or only interim results included (Fig. 1).
Barrier agents
The systematic search identified 10 unique types of barrier agents: oxidized regenerated cellulose (ORC) was used in 14 studies [10, 13,14,15,16,17,18,19,20,21,22,23,24,25], polyethylene glycol (PEG) with/without other agents in 10 studies [26,27,28,29,30,31,32,33,34,35], hyaluronic acid and hyaluronate + carboxymethylcellulose (HA/CMC) in 10 studies [36,37,38,39,40,41,42,43,44,45], icodextrin in 3 studies [46,47,48], 32% dextran 70 in 3 studies [49,50,51], fibrin-containing agents in 2 studies [52, 53], and expanded polytetrafluoroethylene (ePTFE) [54], N,O-carboxymethylchitosan (NOCC) [55] and modified starch [6] in 1 study each.
The basic function of all these products is to physically separate injured tissues during the initial healing process.
Oxidized regenerated cellulose (ORC)
Among the different types of adhesion prevention barriers, ORC was the one studied most widely, with 14 RCTs found (Tab. 1). Regenerated cellulose is manufactured from natural cellulose sources such as wood. Interceed® (Johnson & Johnson, New Brunswick, NJ, USA), the most commonly used ORC, forms a gelatinous barrier.
From the 14 studies reported, 6 showed a significant improvement in adhesion formation with ORC compared to controls, while 8 did not, with 2 even showing a deterioration [17, 22].
Polyethylene glycol (PEG) with/without other agents
PEG is a hydrophilic polymer used for adhesion prevention and is available in different forms: SprayShield™ (Covidien, Waltham, MA, USA, which replaced SprayGel™ (Confluent Surgical, Inc., Waltham,MA, USA)) and CoSeal® (Angiotech Pharmaceuticals, Inc, Vancouver, Canada) are two-component systems consisting of two different PEG solutions. SprayShield™ was developed on the basis of Spraygel™ and has a shorter absorption time that is supposed to reduce foreign-body reaction [56]. Oxiplex/AP Gel (FzioMed, Inc., San Luis Obispo, California, USA, alias Intercoat®) is a gel composed of PEG and CMC. PREVADH™ (Covidien, Trévoux, France) is a more recently studied barrier material consisting of porcine collagen, PEG, and glycerol that forms a hydrogel when hydrated [26]. The latest PEG-containing adhesion barrier is ACTAMAX™, a combination of two aqueous solutions of dextran aldehyde and PEG amine polymers, which form a hydrogel when mixed [34]. We found 10 RCTs using PEG-based barriers for adhesion prevention (Tab. 2).
SprayGel® significantly improved adhesion scores in 1 of 3 RCTs identified. The study evaluating CoSeal® reached significance but lacked investigator blinding and the evidence level is therefore considered lower than that from the other studies [31]. Results of the study using SprayShield™ did not reach statistical significance. However, they showed a positive trend. Oxiplex/AP gel was used in 3 RCTs: 2 of them found a significant improvement in AFS score compared to controls. The latest RCTs on the use of PEG with PREVADH™ and ACTAMAX™ did not show PEG to be superior to control treatment; however, ACTAMAX™ achieved a significantly greater change in score between both surgeries vs. in controls [34].
Hyaluronic acid (HA) / Hyaluronate with or without carboxymethyl cellulose (HA/CMC)
Hyaluronic acid (HA) is a natural component of the extracellular matrix that is also present in the peritoneal fluid. HA has been being used for adhesion prevention for over 10 years under the brand names HyaRegen® (BioRegen Biomedical, Changzhou, China), Hyalobarrier® (auto-crosslinked HA, Fidia Advanced Biopolymers, Abano Terme, Padova, Italy), Intergel® (ionically cross-linked HA, discontinued, Lifecore Biomedical, Inc., Chaska, MN, USA), and Sepracoat (unmodified HA, discontinued, Genzyme Corporation, Cambridge, MA, USA). The combination of hyaluronate and CMC for adhesion prevention has also been studied (Seprafilm® and the discontinued Sepraspray®, Genzyme Biosurgery, Cambridge, MA, USA). Seprafilm® (Baxter Healthcare, Deerfield, IL, USA) is modified chemically and transforms into a gel after being placed on the peritoneum. HA- and HA/CMC-based products were used in 10 RCTs for adhesion prevention (Tab. 3).
Of the 10 RCTs identified, 7 showed efficacy of HA or HA/CMC; 2 of the 3 studies on HA/CMC did not show any significant improvement. All but one of the studies assessed adhesion scores. Thornton et al. used adhesion scores to evaluate adhesion formation; however, they only displayed the results graphically and not numerically. Therefore, it was not possible to include score values in our analysis [45]. Separately published follow-up results of the myomectomy study by Pellicano et al. [44] further showed a significantly higher pregnancy rate after 6 and 12 (the latter after ovulation induction) months [57].
Icodextrin.
Adept® (Baxter Healthcare, Deerfield, IL, USA) is a nonviscous, iso-osmotic, clear 4% icodextrin solution that is approved for gynecological laparoscopic adhesiolysis surgery in the USA and Europe. Physical wound separation is achieved by hydroflotation. Adept® was used in 3 RCTs for adhesion prevention (Tab. 4).
Of the 3 RCTs identified, 1 showed a significant improvement, though not in the AFS scores—which were not provided—but rather in clinical success, defined as the percentage of patients in whom the number of sites with adhesions decreased by at least three or 30% of the number of sites analyzed [47]. Two studies reported adverse events, such as postoperative pain and headache [46, 48], nausea, vomiting, wound infection and vulval/genital edema [46], as well as a 45% increase in adhesion scores in the intervention group.
Expanded polytetrafluoroethylene (ePTFE)
ePTFE is a synthetic, nonresorbable barrier agent. We found 1 study using ePTFE (Gore-Tex Surgical Membrane, W.L. Gore and Associates, Inc., Flagstaff, AZ, USA) (Tab. 5).
The study identified showed a significant improvement of 75% in mean adhesion score.
Modified starch
Starch can be modified and crosslinked to improve its capabilities in medical applications. For adhesion prevention, modified starch powder is mixed with saline to form a gel. We identified one RCT using the modified starch 4DryField® for adhesion prevention (Tab. 6).
4DryField® reached a significant improvement of 85% in mean total adhesion score compared to peritoneal irrigation with saline. Total adhesion score was calculated by multiplication of the extent and severity score for the evaluated sites followed by summing up over all sites. Separately published follow-up results of this RCT further showed that fertility was significantly higher and pain scores reduced [58].
Other approaches
Three other types of adhesion barriers were examined in RCTs between 1983 and 2011, but none of them have been the subject of recently conducted trials: (1) dextran, a chain polysaccharide derivative of sugar beets, which is a very viscous agent that is very slowly absorbed from the peritoneal cavity; (2) gelatinous fibrin formed from fibrinogen and thrombin, which remains on the spot as a stable barrier for about 1 week. While impaired fibrinolysis has been identified as a factor increasing the probability of adhesion formation, the application of fibrin-containing gel or sheets—at least in theory—act as an adhesion preventive barrier agent as it directly closes wound surfaces, decreasing the physiological cascade of adhesion formation during wound-healing; (3) N,O–carboxymethylchitosan (NOCC), a purified derivative of chitin obtained from shrimp, which is similar in structure to HA and CMC. We identified three RCTs using 32% dextran 70 (Hyskon®, Hyskon, Pharmacia, Inc., Piscataway, NJ, USA) for adhesion prevention, two studies with fibrin barrier agents (Adhexil™ (Omrix Biopharmaceuticals, Inc., Ness Ziona, Israel) and fibrin gel/sheet), and one RCT using NOCC (Tab. 7).
Of the three RCTs studying 32% dextran 70, those of Larson et al. and Rosenberg and Board showed improvements in net changes of adhesion scores, which in the latter case was statistically significant [50, 51]. Nevertheless, Rosenberg and Board did not provide a statistical comparison of adhesion scores [51]. Of the two RCTs studying fibrin, treatment with Adhexil™ showed a trend toward improved adhesion prevention (mAFS); however, the difference was not statistically significant [52]. Takeuchi et al. compared fibrin sheet or fibrin gel with a control group and found that fibrin gel significantly reduced the incidence of adhesions; however, no score was assessed [53]. In their RCT on NOCC, Diamond et al. found an increase in adhesion scores in the intervention group at second look vs. controls [55]. When these were offset against the scores from the first surgery, both results were better for the intervention group, albeit not significantly.
Discussion
A strategy for preventing adhesion formation during gynecological surgery is crucial to avoiding complications and has the potential to spare resources and thus save costs in healthcare systems [8]. A promising and widely studied strategy is the use of barrier agents. Before adhesion barriers can be used routinely, however, their ability to prevent the formation of adhesions must be thoroughly tested in the respective context. Furthermore, potential side effects of resorbable and of nonresorbable agents need to be evaluated and compared to the appropriateness of the expected positive effects with the possible toxic risks. Various barriers have been used to prevent adhesions and numerous studies have been published in the field of gynecological surgery. The assessment of adhesions in second-look surgery varies between the individual studies. Therefore, a comparative efficacy evaluation of the barriers is challenging. To address this issue, we looked for adhesion scores as primary outcome measure. When scores were not available, we focused on the incidence of adhesions and, finally, adhesion-free patients, the latter being considered as the lowest explanatory quality. As most studies provided adhesion scores, informed judgments of the effectiveness of the respective barrier devices were feasible. Generally, an ideal barrier should exert its effect on the wound at least until the wound-healing processes are complete, which takes about 5–6 days postoperatively [59,60,61]. In a recent article on the topic, a time of about 7 days is suggested to be ideal [8]. Nevertheless, further research is needed to elucidate the exact timing of wound-healing processes and, consequentially, the optimal breakdown times for absorbable barrier agents. In addition, the ideal adhesion barrier should not have to be removed again, interfere with peritoneal healing, or be influenced by the presence of blood [36]. In summary, an ideal barrier agent should be biodegradable, laparoscopically applicable, clinically efficacious, and affordable for daily routine use [8].
The potential of adhesion prevention with icodextrin is not convincing as only 1 of 3 RCTs showed a significant improvement by 11% compared to controls [47]. The outcome measure here was “clinical success”, whereas it was mAFS in the other 2 studies. Adept® did not significantly reduce adhesions in several preclinical trials either [62,63,64,65,66]. Icodextrin solution is rapidly absorbed from the abdominal cavity and, thus, the retention time of about 7 days required for optimal adhesion prevention due to peritoneal healing is not achieved. This could potentially explain why Adept® delivered results inferior to those for (semi)solid barriers. Additional drawbacks of Adept® are the very narrow indication (it is only intended for gynecological laparoscopic adhesiolysis surgery) and the lacking possibility to use drains. Furthermore, Lee et al. [67] showed that using 4% icodextrin the incidence of small bowel obstruction was higher than when not using any anti-adhesion materials [27], and severe small bowel serosal fibrosis and dense adhesions were also reported in patients in whom 4% icodextrin was used in abdominal surgery [68].
The 3 RCTs using 32% dextran 70 (Hyskon®) for adhesion prevention did not yield significant improvements in adhesion scores; however, one study showed significant improvement in net adhesion scores [51] and one in adhesion scores of patients with a severe initial extent of adnexal adhesions [49]. The studies were conducted in 1983–1985, though, and no further RCTs have been published since then as safety concerns were reported regarding the proliferation of bacteria in Hyskon®-containing media and concerning the anaphylactoid potential reported [49].
Adhesion prevention with fibrin was assessed in 2 RCTs, with only one presenting mAFS scores. The latter study showed a trend toward successful adhesion prevention with Adhexil™ but only included a small population of 16 patients [52]. The other study found a significant reduction in the incidence of adhesions in the fibrin gel group, but no improvement with fibrin sheets [53]. As no score was assessed in the second RCT, no sound comparison of the effectiveness of fibrin in these 2 studies can be made. Although fibrin has important characteristics of an ideal barrier [69], such as safety and remaining on the surgical site for about 1 week, its efficacy is questionable based on the data presently available.
ePTFE-based Gore-Tex Surgical Membrane achieved a significant improvement in mean adhesion score in the RCT found [54]. In a comparative study without a control group not included in the present review, ePTFE was found to be superior to ORC (Interceed®) in preventing adhesions after adnexal surgery [70]. However, since the Gore-Tex Surgical Membrane has to be removed again as it is nonresorbable, ePTFE cannot be recommended for preventing adhesions due to the requirement of a subsequent surgical intervention, imposing once again the risk of adhesion formation, in addition to the other risks and complications that naturally occur during and after surgeries. ePTFE, therefore, plays no role in today’s surgical routine in gynecology.
NOCC was used in 1 RCT where severity and extent of adhesions at second look were even higher in the intervention group. Although a change in adhesion score and number of sites with recurrent adhesions were reduced, these results were not statistically significant [55]. NOCC does not mix easily with blood, a desired feature of an adhesion barrier. As this was the pilot clinical study using NOCC and no further RCTs were conducted, it is not possible to reach a valid conclusion regarding the effectiveness of NOCC in preventing adhesions in humans.
ORC (Interceed®) was the most widely studied barrier agent, with 14 RCTs identified; however, the adhesion prevention potential was unconvincing: 6 RCTs showed a significant improvement in adhesion prevention with ORC compared to controls [13, 14, 18, 20, 21, 24]. In only one of these trials was a more than 50% improvement achieved [20], indicating that a large proportion of adhesions remained. Furthermore, the explanatory power of most outcome measures is considered rather low (adhesion-free patients, adhesion incidence) and 2 studies even showed a trend toward elevated adhesion formation after applying Interceed®.[17, 22] The authors explained these results with the assumed differential potential of Interceed® in reducing de novo or reformed adhesions and because the whole ovary was not covered with the barrier. Similarly, unconvincing results have been reported in numerous preclinical studies in which Interceed® failed to prevent adhesion formation [65, 71,72,73,74,75,76]. Another drawback of this adhesion barrier is its reported incompatibility with the presence of blood [8] as this would lead to adhesion formation through the barrier [77, 78] and adhesions that formed despite the presence of Interceed® were histologically shown to include substantial amounts of product remnants at agglutination sites associated with a local inflammatory response [65]. Accordingly, it would be almost impossible to use this product after myomectomy considering that hemostasis at the myomectomy site is rarely complete [54]. Preclinical studies further showed an inflammatory response, as well as sloughing of intact peritoneum when ORC was applied, inducing de novo adhesion formation [65, 79].
PEG (SprayShield™, SprayGel™, Oxiplex/AP Gel alias Intercoat®, PREVADH™, and ACTAMAX™) was used in 10 RCTs. All 10 RCTS used adhesion scores and therefore exhibit high explanatory power. SprayGel® showed significant improvements in adhesion prevention in 1 of 3 RCTs identified, exhibiting a 58% improvement [80]. SprayShield™ was used in one study in 2014 and the results did not reach statistical significance. However, they showed a trend toward a positive effect of SprayShield™ [32]. In 2 of the 3 RCTs on Oxiplex/AP gel, a significant improvement in AFS score was shown compared to controls, while in the third, in which patients primarily received adhesiolysis, the score in the intervention group was identical for both the second and first surgeries. Furthermore, two RCTs examining Oxiplex/AP Gel for intrauterine adhesion prevention also found contradictory results [81, 82]. A general drawback of this product could be its long resorption time, which might induce foreign-body reactions. PREVADH™ did not significantly improve the mAFS. ACTAMAX™ did not lead to improved adhesion scores either but did achieve a significantly greater change in score than in controls. The authors noted that the ACTAMAX™ study was not powered to detect differences between randomized groups in efficacy outcomes. In summary, proof of efficacy of PEG-based barriers is inconclusive. The simple application and the possibility of covering large areas is an advantage of SprayShield™; however, its longer resorption time could possibly result in foreign-body reactions. The rather high costs of SprayGel™ are also a drawback of this specific PEG adhesion barrier [4].
Of the 10 RCTs identified using HA (HyaRegen®, Hyalobarrier®, Intergel® and Sepracoat) or HA/CMC (Seprafilm® and Sepraspray®), 7 showed superiority compared to controls, including 6 of 7 studies on HA and 1 of 3 studies on HA/CMC. All but one of the studies assessed adhesion scores. Liu et al. showed the greatest effect with HA with an improvement of 67% in mAFS score using the auto-crosslinked HyaRegen® gel [41]. However, the other auto-crosslinked HA barrier, Hyalobarrier®, showed no improvement at all [43] and the third trial using an auto-crosslinked HA barrier did not report adhesion scores. Accordingly, proof for efficacy of this material remains inconclusive. This is further reinforced by results of several trials examining intrauterine adhesion prevention with auto-crosslinked HA gels, which also achieved contradictory results [83,84,85,86], as well as preclinical studies which found that it was not effective [87,88,89]. HA-based barrier Intergel® was associated with safety issues in an abdominal surgery RCT concerning postoperative morbidities, particularly causing peritonitis, anastomotic dehiscence, and prolonged ileus [90]. This product has been discontinued. Furthermore, there are safety concerns based on in vitro data demonstrating that HA may promote tumor growth [91] although a clinical evaluation seems to dispel these doubts at least for the HA/CMC combination product Seprafilm®.[92] As Sepraspray® has been discontinued, the results for Seprafilm® are of greater interest concerning HA/CMC combinations. The most recent study on Seprafilm® did not show any difference in the mean adhesion score [40] and therefore could not confirm the significant improvements reported by Diamond in 1996 [36]. In several preclinical studies, it also failed to prevent adhesion formation [65, 75, 89]. Major drawbacks of Seprafilm® are that it cannot be applied easily laparoscopically yet and that it can be difficult to adequately cover uneven surfaces [36]. Furthermore, a large RCT in abdominal surgery found that Seprafilm® significantly increases the incidence of fistulas, peritonitis, and anastomotic leakage [93]. A significantly higher risk of anastomotic leak was confirmed based on data from eight studies and 3,037 patients [94]. Therefore, the routine use of HA or HA/CMC is not supported by the present data and further studies should be performed.
The modified starch-based device 4DryField® yielded a significant improvement, reaching a mean total adhesion score reduction of 85% in its first RCT [6]—the best result achieved with an absorbable barrier. In this RCT, patients with deep infiltrating endometriosis were included, who received excision for histological confirmation and symptom relief during the first and complete excision during the second intervention, where adhesion scoring was performed by an assessor blinded to the patient’s group assignment. The outcome is in line with results from non-RCTs: 85% after endometrioma resection [95], up to 75% after gynecological adhesiolysis [96], 87.5% after release of adhesive small bowel obstruction [97], and 100% in preclinical trials [98]. Furthermore, a preclinical trial comprising a direct comparison with Interceed® (ORC), Seprafilm® (HA/CMC), and Adept® (icodextrin) showed that 4DryField® is significantly more effective than any of these products [65]. Adhesion reduction was 93% with 4DryField®, 54% with Seprafilm®, 16% with Adept® and 4% with Interceed®. This trial also compared the two possible ways of application for 4DryField®. Applied as powder and subsequently transformed into a gel by dripping, adhesion reduction was 100%, while it was 85% when applied directly as an ex situ premixed gel (difference between these two ways of application not statistically significant). Additional preclinical and clinical trials showed that other currently available modified starch powder-based agents did not yield a statistically significant reduction in adhesion formation.[98,99,100]. It has been supposed that one factor contributing to these results is the fact that all of these exhibit retention times of only up to three days maximum and, therefore, are absorbed before mesothelial healing is completed [8, 100].
In general, patient-reported outcome measures are of high clinical relevance when considering the use of adhesion barriers. Despite the importance of such data, a 2020 Cochrane Review on barrier agents did not find any studies examining the influence on live birth rates or pelvic pain [101]. In addition, only seven of the 45 RCTs included in the present review article presented follow-up results for fertility and/or pelvic pain. Of these seven RCTs, all reported on pregnancy rates, but only one examined postoperative pain development. Three of the six RCTs examined pregnancy rates after application of Interceed®[17, 22, 23]. It is not possible to draw any conclusions on the influence of Interceed® for the studies by Greenblatt and Casper [17] and Saravelos and Li [22]. However, as in all patients, the product was applied to one ovary, while the other was left untreated and served as an internal control. In both studies, adhesion scores at the ovaries treated with Interceed® were higher than those of the control ovaries, showing statistically nonsignificant deteriorations [17, 22]. The third study on Interceed® did not reach a significant improvement in the incidence of adhesions but found an increased pregnancy rate after 2 years (78.3% vs. 46.7%, P < 0.049) [23]. Of the remaining four studies dealing with patient-reported outcome measures, one examined PREVADH™, one Hyskon®, one Hyalobarrier®, and one 4DryField®. The study on PREVADH™ did not yield a significant improvement in adhesion scores but did find an increased pregnancy rate after 3 years (64% vs. 24%, P = 0.02) [26]. The study on Hyskon® showed nonsignificant deteriorations in both adhesion scores (9.93 vs. 10.71) and pregnancy rate (23.5% vs. 33.3%) in the intervention group [50]. The studies on Hyalobarrier® and 4DryField® are the only RCTs achieving significant improvements in both adhesion scores and pregnancy rates in the intervention group [6, 57, 58]. After 12 months, pregnancy rate was 39% after myomectomy and 21% after endometriosis resection when no adhesion prevention was utilized. These rates could be doubled from 39 to 78% when Hyalobarrier® was applied and tripled from 21 to 64% when 4DryField® was used. In the study on Hyalobarrier®, patients who did not conceive after 6 months underwent ovulation induction from the 7th to the 12th follow-up month [57], while in the study on 4DryField® patients did not undergo ovulation induction. As several studies have described adhesions as the main cause of secondary infertility, it is conceivable that the higher pregnancy rates are related to reduced adhesion formation [58]. The 4DryField® study is the only RCT examining the influence of an adhesion barrier on pain relief. In the intervention group, all of the five pain scores examined [cycle-independent pelvic pain (CIPP), dysmenorrhea, dyspareunia, dyschezia, and dysuria] were lower than in the control group 12 months after the second intervention [58]. Furthermore, the scores for CIPP, dysmenorrhea, and dyspareunia were significantly lower than before the first surgery in the intervention group, while only dysmenorrhea was significantly reduced in the control group [58]. As a correlation between adhesion formation and pain has been shown in many studies, it is conceivable that the more favorable pain development in the intervention group is linked to an effective prevention of adhesion formation in this group as compared to the control group [58]. Furthermore, patients in the control group had increasing pain scores in the later course of the follow-up period, which might be explained by the general presence of adhesions and an increasing adhesion severity over time being causative for pain recurrence [58]. The intervention group, in contrast, did not show an increase in pain scores and generally exhibited more favorable pain score results, likely due to the reduction of adhesion formation by the adhesion barrier [58]. Considering the high relevance of follow-up results, particularly for patients but also for surgeons, hospitals, and the healthcare system, the very limited availability of such data from RCTs is hardly understandable and constitutes a severe gap in scientific knowledge. Therefore, it is urgently recommended to include follow-up data and patient-reported outcome measures in future RCTs.
Conclusion
It is crucial in gynecological surgery to prevent adhesions and protect women from pain, infertility, bowel obstructions, and need for repeated operations. The most promising adhesion score reductions, the outcome measure of the highest explanatory quality, were achieved using Gore-Tex Surgical Membrane with 75%, hyaluronic acid with 0–67%, and 4DryField® with 85% improvement. As Gore-Tex Surgical Membrane is nonabsorbable, it must be removed again surgically, imposing a greater risk of complications and, consequentially, adhesion formation in contrast to HA and 4DryField®. 4DryField®, which is associated with the most pronounced improvement in adhesion scores of all the barrier agents, combines a resorption time of about 7 days with ease of use and a favorable safety profile. Future studies should use standardized scores such as the mAFS to generally facilitate comparability of the results. Furthermore, patient-reported outcome measures, such as pain and infertility, should be emphasized more in future trials, and corresponding endpoints should be collected. If these aspects are considered, it could become easier to address the multiple and diffuse risks arising from postoperative adhesion formation. The authors’ vision that iatrogenically caused adhesions can be minimized due to a general use of agents for adhesion prevention that are not associated with any risk seems achievable.
Data availability
Data will be made available upon reasonable request by the authors.
References
Practice Committee of American Society for Reproductive Medicine in collaboration with Society of Reproductive Surgeons (2013) Pathogenesis, consequences, and control of peritoneal adhesions in gynecologic surgery: a committee opinion. Fertil Steril 99(6):1550–1555. https://doi.org/10.1016/j.fertnstert.2013.02.031
Brüggmann D, Tchartchian G, Wallwiener M, Münstedt K, Tinneberg HR, Hackethal A (2010) Intra-abdominal adhesions: definition, origin, significance in surgical practice, and treatment options. Deutsches Arzteblatt Int 107(44):769–775
Pados G, Venetis CA, Almaloglou K, Tarlatzis BC (2010) Prevention of intra-peritoneal adhesions in gynaecological surgery: theory and evidence. Reprod Biomed Online 21(3):290–303
Trew G (2004) Consensus in adhesion reduction management. Obstetrician Gynaecologist 6(2):1–16
Liakakos T, Thomakos N, Fine PM, Dervenis C, Young RL (2001) Peritoneal adhesions: etiology, pathophysiology, and clinical significance. Recent advances in prevention and management. Dig Surgery. 18(4):260–273
Krämer B, Andress J, Neis F, Hoffmann S, Brucker S, Kommoss S, Holler A (2021) Adhesion prevention after endometriosis surgery - results of a randomized, controlled clinical trial with second-look laparoscopy. Langenbecks Arch Surg 406(6):2133–2143
Somigliana E, Benaglia L, Vigano P, Candiani M, Vercellini P, Fedele L (2011) Surgical measures for endometriosis-related infertility: a plea for research. Placenta 32(Suppl 3):S238–S242
Krämer B, Neis F, Brucker SY, Kommoss S, Andress J, Hoffmann S (2021) Peritoneal adhesions and their prevention - current trends. Surg Technol Int 38:221–233
Binda MM (2015) Humidification during laparoscopic surgery: overview of the clinical benefits of using humidified gas during laparoscopic surgery. Arch Gynecol Obstet 292(5):955–971
Tinelli A, Malvasi A, Guido M, Tsin DA, Hudelist G, Hurst B et al (2011) Adhesion formation after intracapsular myomectomy with or without adhesion barrier. Fertil Steril 95(5):1780–1785
Brochhausen C, Schmitt VH, Planck CN, Rajab TK, Hollemann D, Tapprich C et al (2012) Current strategies and future perspectives for intraperitoneal adhesion prevention. J Gastrointest Surg 16(6):1256–1274
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. PLoS Med 18(3):e1003583
(1989) Prevention of postsurgical adhesions by INTERCEED(TC7), an absorbable adhesion barrier: a prospective randomized multicenter clinical study. INTERCEED(TC7) Adhesion Barrier Study Group. Fertility and sterility. 51(6):933–8
(1995) The efficacy of Interceed(TC7)* for prevention of reformation of postoperative adhesions on ovaries, fallopian tubes, and fimbriae in microsurgical operations for fertility: a multicenter study. Nordic Adhesion Prevention Study Group. Fertil Steril. 63(4):709–14
Azziz R (1993) Microsurgery alone or with INTERCEED absorbable adhesion barrier for pelvic sidewall adhesion re-formation. The INTERCEED (TC7) adhesion barrier study group II. Surgery, gynecology & obstetrics. 177(2):135–139
Franklin RR (1995) Reduction of ovarian adhesions by the use of Interceed. Ovarian Adhesion Study Group Obstet Gynecol 86(3):335–340
Greenblatt EM, Casper RF (1993) Adhesion formation after laparoscopic ovarian cautery for polycystic ovarian syndrome: lack of correlation with pregnancy rate. Fertil Steril 60(5):766–770
Keckstein J, Ulrich U, Sasse V, Roth A, Tuttlies F, Karageorgieva E (1996) Reduction of postoperative adhesion formation after laparoscopic ovarian cystectomy. Hum Reprod 11(3):579–582
Li TC, Cooke ID (1994) The value of an absorbable adhesion barrier, Interceed, in the prevention of adhesion reformation following microsurgical adhesiolysis. Br J Obstet Gynaecol 101(4):335–339
Mais V, Ajossa S, Marongiu D, Peiretti RF, Guerriero S, Melis GB (1995) Reduction of adhesion reformation after laparoscopic endometriosis surgery: a randomized trial with an oxidized regenerated cellulose absorbable barrier. Obstet Gynecol 86(4 Pt 1):512–515
Mais V, Ajossa S, Piras B, Guerriero S, Marongiu D, Melis GB (1995) Prevention of de-novo adhesion formation after laparoscopic myomectomy: a randomized trial to evaluate the effectiveness of an oxidized regenerated cellulose absorbable barrier. Hum Reprod 10(12):3133–3135
Saravelos H, Li TC (1996) Post-operative adhesions after laparoscopic electrosurgical treatment for polycystic ovarian syndrome with the application of Interceed to one ovary: a prospective randomized controlled study. Hum Reprod 11(5):992–997
Sawada T, Nishizawa H, Nishio E, Kadowaki M (2000) Postoperative adhesion prevention with an oxidized regenerated cellulose adhesion barrier in infertile women. J Reprod Med 45(5):387–389
Sekiba K (1992) Use of Interceed(TC7) absorbable adhesion barrier to reduce postoperative adhesion reformation in infertility and endometriosis surgery. The Obstetrics and Gynecology Adhesion Prevention Committee. Obstet Gynecol 79(4):518–522
Wallwiener D, Meyer A, Bastert G (1998) Adhesion formation of the parietal and visceral peritoneum: an explanation for the controversy on the use of autologous and alloplastic barriers? Fertil Steril 69(1):132–137
Canis MJ, Triopon G, Daraï E, Madelenat P, LeVêque J, Panel P, et al. (2014) Adhesion prevention after myomectomy by laparotomy: a prospective multicenter comparative randomized single-blind study with second-look laparoscopy to assess the effectiveness of PREVADH™. Eur J Obstetrics Gynecol Reproductive Biol 178:42–7.
diZerega GS, Coad J, Donnez J (2007) Clinical evaluation of endometriosis and differential response to surgical therapy with and without application of Oxiplex/AP* adhesion barrier gel. Fertil Steril 87(3):485–489
Johns DA, Ferland R, Dunn R (2003) Initial feasibility study of a sprayable hydrogel adhesion barrier system in patients undergoing laparoscopic ovarian surgery. J Am Assoc Gynecol Laparosc 10(3):334–338
Lundorff P, Donnez J, Korell M, Audebert AJ, Block K, diZerega GS (2005) Clinical evaluation of a viscoelastic gel for reduction of adhesions following gynaecological surgery by laparoscopy in Europe. Hum Reprod 20(2):514–520
Mettler L, Audebert A, Lehmann-Willenbrock E, Schive-Peterhansl K, Jacobs VR (2004) A randomized, prospective, controlled, multicenter clinical trial of a sprayable, site-specific adhesion barrier system in patients undergoing myomectomy. Fertil Steril 82(2):398–404
Mettler L, Hucke J, Bojahr B, Tinneberg H-R, Leyland N, Avelar R (2008) A safety and efficacy study of a resorbable hydrogel for reduction of post-operative adhesions following myomectomy†. Hum Reprod 23(5):1093–1100
Tchartchian G, Hackethal A, Herrmann A, Bojahr B, Wallwiener C, Ohlinger R et al (2014) Evaluation of SprayShield™ adhesion barrier in a single center: randomized controlled study in 15 women undergoing reconstructive surgery after laparoscopic myomectomy. Arch Gynecol Obstet 290(4):697–704
Ten Broek RP, Kok-Krant N, Verhoeve HR, van Goor H, Bakkum EA (2012) Efficacy of polyethylene glycol adhesion barrier after gynecological laparoscopic surgery: results of a randomized controlled pilot study. Gynecol Surg 9(1):29–35
Trew GH, Pistofidis GA, Brucker SY, Krämer B, Ziegler NM, Korell M et al (2017) A first-in-human, randomized, controlled, subject- and reviewer-blinded multicenter study of Actamax™ adhesion barrier. Arch Gynecol Obstet 295(2):383–395
Young P, Johns A, Templeman C, Witz C, Webster B, Ferland R et al (2005) Reduction of postoperative adhesions after laparoscopic gynecological surgery with Oxiplex/AP Gel: a pilot study. Fertil Steril 84(5):1450–1456
Diamond MP (1996) Reduction of adhesions after uterine myomectomy by Seprafilm membrane (HAL-F): a blinded, prospective, randomized, multicenter clinical study. Seprafilm Adhesion Study Group Fertil Steril 66(6):904–910
Diamond MP (1998) Reduction of de novo postsurgical adhesions by intraoperative precoating with Sepracoat (HAL-C) solution: a prospective, randomized, blinded, placebo-controlled multicenter study. The Sepracoat Adhesion Study Group. Fertil Steril 69(6):1067–1074
Fossum GT, Silverberg KM, Miller CE, Diamond MP, Holmdahl L (2011) Gynecologic use of sepraspray adhesion barrier for reduction of adhesion development after laparoscopic myomectomy: a pilot study. Fertil Steril 96(2):487–491
Johns DB, Keyport GM, Hoehler F, diZerega GS (2001) Reduction of postsurgical adhesions with Intergel adhesion prevention solution: a multicenter study of safety and efficacy after conservative gynecologic surgery. Fertil Steril 76(3):595–604
Kiefer DG, Muscat JC, Santorelli J, Chavez MR, Ananth CV, Smulian JC, Vintzileos AM (2016) Effectiveness and short-term safety of modified sodium hyaluronic acid-carboxymethylcellulose at cesarean delivery: a randomized trial. Am J Obstet Gynecol 214(3):373e1-e12
Liu C, Lu Q, Zhang Z, Xue M, Zhang Y, Zhang Y et al (2015) A randomized controlled trial on the efficacy and safety of a new crosslinked hyaluronan gel in reducing adhesions after gynecologic laparoscopic surgeries. J Minim Invasive Gynecol 22(5):853–863
Lundorff P, van Geldorp H, Tronstad SE, Lalos O, Larsson B, Johns DB, diZerega GS (2001) Reduction of post-surgical adhesions with ferric hyaluronate gel: a European study. Hum Reprod 16(9):1982–1988
Mais V, Bracco GL, Litta P, Gargiulo T, Melis GB (2006) Reduction of postoperative adhesions with an auto-crosslinked hyaluronan gel in gynaecological laparoscopic surgery: a blinded, controlled, randomized, multicentre study. Hum Reprod 21(5):1248–1254
Pellicano M, Bramante S, Cirillo D, Palomba S, Bifulco G, Zullo F, Nappi C (2003) Effectiveness of autocrosslinked hyaluronic acid gel after laparoscopic myomectomy in infertile patients: a prospective, randomized, controlled study. Fertil Steril 80(2):441–444
Thornton MH, Johns DB, Campeau JD, Hoehler F, DiZerega GS (1998) Clinical evaluation of 0.5% ferric hyaluronate adhesion prevention gel for the reduction of adhesions following peritoneal cavity surgery: open-label pilot study. Human Reprod (Oxford, England). 13(6):1480–5
Trew G, Pistofidis G, Pados G, Lower A, Mettler L, Wallwiener D et al (2011) Gynaecological endoscopic evaluation of 4% icodextrin solution: a European, multicentre, double-blind, randomized study of the efficacy and safety in the reduction of de novo adhesions after laparoscopic gynaecological surgery. Human Reprod (Oxford, England) 26(8):2015–2027
Brown CB, Luciano AA, Martin D, Peers E, Scrimgeour A, diZerega GS (2007) Adept (icodextrin 4% solution) reduces adhesions after laparoscopic surgery for adhesiolysis: a double-blind, randomized, controlled study. Fertil Steril 88(5):1413–1426
diZerega GS, Verco SJ, Young P, Kettel M, Kobak W, Martin D et al (2002) A randomized, controlled pilot study of the safety and efficacy of 4% icodextrin solution in the reduction of adhesions following laparoscopic gynaecological surgery. Hum Reprod 17(4):1031–1038
(1983) Reduction of postoperative pelvic adhesions with intraperitoneal 32% dextran 70: a prospective, randomized clinical trial. Fertility Steril 40(5):612–9
Larsson B, Lalos O, Marsk L, Tronstad SE, Bygdeman M, Pehrson S, Joelsson I (1985) Effect of intraperitoneal instillation of 32% dextran 70 on postoperative adhesion formation after tubal surgery. Acta Obstet Gynecol Scand 64(5):437–441
Rosenberg SM, Board JA (1984) High-molecular weight dextran in human infertility surgery. Am J Obstet Gynecol 148(4):380–385
Diamond MP, Korell M, Martinez S, Kurman E, Kamar M (2011) A prospective, controlled, randomized, multicenter, exploratory pilot study evaluating the safety and potential trends in efficacy of Adhexil. Fertil Steril 95(3):1086–1090
Takeuchi H, Kitade M, Kikuchi I, Shimanuki H, Kumakiri J, Kinoshita K (2005) Adhesion-prevention effects of fibrin sealants after laparoscopic myomectomy as determined by second-look laparoscopy: a prospective, randomized, controlled study. J Reprod Med 50(8):571–577
Group TMAMS (1995) An expanded polytetrafluoroethylene barrier (Gore-Tex surgical membrane) reduces post-myomectomy adhesion formation. The myomectomy adhesion multicenter study group. Fertil Steril 63(3):491–3
Diamond MP, Luciano A, Johns DA, Dunn R, Young P, Bieber E (2003) Reduction of postoperative adhesions by N, O-carboxymethylchitosan: a pilot study. Fertil Steril 80(3):631–636
Rajab TK, Kimonis KO, Ali E, Offodile AC, Brady M, Bleday R (2013) Practical implications of postoperative adhesions for preoperative consent and operative technique. Int J Surg 11(9):753–756
Pellicano M, Guida M, Bramante S, Acunzo G, Di Spiezio SA, Tommaselli GA, Nappi C (2005) Reproductive outcome after autocrosslinked hyaluronic acid gel application in infertile patients who underwent laparoscopic myomectomy. Fertil Steril 83(2):498–500
Krämer B, Andress J, Neis F, Hoffmann S, Brucker S, Kommoss S, Höller A (2023) Improvement in fertility and pain after endometriosis resection and adhesion prevention with 4DryField® PH: follow-up of a randomized controlled clinical trial. J Clin Med 12(10):3597
Ellis H, Harrison W, Hugh TB (1965) The healing of peritoneum under normal and pathological conditions. Br J Surg 52:471–476
Hubbard TB Jr, Khan MZ, Carag VR Jr, Albites VE, Hricko GM (1967) The pathology of peritoneal repair: its relation to the formation of adhesions. Ann Surg 165(6):908–916
Shapiro L, Holste JL, Muench T, diZerega G (2015) Rapid reperitonealization and wound healing in a preclinical model of abdominal trauma repair with a composite mesh. Int J Surg 22:86–91
Ditzel M, Deerenberg EB, Komen N, Mulder IM, Jeekel H, Lange JF (2012) Postoperative adhesion prevention with a new barrier: an experimental study. Eur Surg Res 48(4):187–193
Hwang HJ, An MS, Ha TK, Kim KH, Kim TH, Choi CS et al (2013) All the commercially available adhesion barriers have the same effect on adhesion prophylaxis?; A comparison of barrier agents using a newly developed, severe intra-abdominal adhesion model. Int J Colorectal Dis 28(8):1117–1125
Klink CD, Schickhaus P, Binnebösel M, Jockenhoevel S, Rosch R, Tolba R, Neumann UP, Klinge U (2013) Influence of 4% icodextrin solution on peritoneal tissue response and adhesion formation. BMC Surg. https://doi.org/10.1186/1471-2482-13-34
Poehnert D, Grethe L, Maegel L, Jonigk D, Lippmann T, Kaltenborn A et al (2016) Evaluation of the effectiveness of peritoneal adhesion prevention devices in a rat model. Int J Med Sci 13(7):524–532
Müller SA, Treutner KH, Haase G, Kinzel S, Tietze L, Schumpelick V (2003) Effect of intraperitoneal antiadhesive fluids in a rat peritonitis model. Arch Surg 138(3):286–290
Lee WS, Baek JH, Lee WK (2013) Direct comparison of Seprafilm (R) versus Adept (R) versus no additive for reducing the risk of small-bowel obstruction in colorectal cancer surgery. Surg Today 43(9):995–1002
Liu T-M, Kiu K-T, Yen M-H, Tam K-W, Chang T-C (2023) Efficacy and safety of purified starch for adhesion prevention in colorectal surgery. Heliyon. 9(11):e21657
Korell M, Studien W (2010) Methoden der Adhäsionsprophylaxe - Pro und Kontra. J für Gynäkologische Endokrinologie 20:6–13
Haney AF, Hesla J, Hurst BS, Kettel LM, Murphy AA, Rock JA et al (1995) Expanded polytetrafluoroethylene (Gore-Tex Surgical Membrane) is superior to oxidized regenerated cellulose (Interceed TC7+) in preventing adhesions. Fertil Steril 63(5):1021–1026
Best CL, Rittenhouse D, Vasquez C, Norng T, Subias E, Sueldo CE (1992) Evaluation of Interceed (TC7) for reduction of postoperative adhesions in rabbits. Fertil Steril 58(4):817–820
Pagidas K, Tulandi T (1992) Effects of Ringer’s lactate, lnterceed(TC7) and Gore-Tex† surgical membrane on postsurgical adhesion formation. Fertil Steril 57(1):199–201
Maxson WS, Herbert CM, Oldfield EL, Hill GA (1988) Efficacy of a modified oxidized cellulose fabric in the prevention of adhesion formation. Gynecol Obstet Invest 26(2):160–165
Cho WJ, Oh SH, Lee JH (2010) Alginate film as a novel post-surgical tissue adhesion barrier. J Biomater Sci Polym Ed 21(6):701–713
Takagi K, Tsuchiya T, Araki M, Yamasaki N, Nagayasu T, Hyon SH, Nakajima N (2013) Novel biodegradable powder for preventing postoperative pleural adhesion. J Surg Res 179(1):e13–e19
Peyton CC, Keys T, Tomblyn S, Burmeister D, Beumer JH, Holleran JL et al (2012) Halofuginone infused keratin hydrogel attenuates adhesions in a rodent cecal abrasion model. J Surg Res 178(2):545–552
DeCherney AH, diZerega GS (1997) Clinical problem of intraperitoneal postsurgical adhesion formation following general surgery and the use of adhesion prevention barriers. Surg Clin N Am 77(3):671–688
Wiseman D (1994) Polymers for the prevention of surgical adhesions. In: Domb AJ (ed) Polymeric site-specific pharmacotherapy. Wiley, New York, pp 370–421
Haney AF, Doty E (1992) Murine peritoneal injury and de novo adhesion formation caused by oxidized-regenerated cellulose (Interceed [TC7]) but not expanded polytetrafluoroethylene (Gore-Tex Surgical Membrane). Fertil Steril 57(1):202–208
Mettler L, Audebert A, Lehmann-Willenbrock E, Schive K, Jacobs VR (2003) Prospective clinical trial of SprayGel as a barrier to adhesion formation: an interim analysis. J Am Assoc Gynecol Laparosc 10(3):339–344
Di Spiezio SA, Spinelli M, Bramante S, Scognamiglio M, Greco E, Guida M et al (2011) Efficacy of a polyethylene oxide-sodium carboxymethylcellulose gel in prevention of intrauterine adhesions after hysteroscopic surgery. J Minim Invasive Gynecol 18(4):462–469
Fuchs N, Smorgick N, Ben Ami I, Vaknin Z, Tovbin Y, Halperin R, Pansky M (2014) Intercoat (Oxiplex/AP gel) for preventing intrauterine adhesions after operative hysteroscopy for suspected retained products of conception: double-blind, prospective, randomized pilot study. J Minim Invasive Gynecol 21(1):126–130
Li X, Wu L, Zhou Y, Fan X, Huang J, Wu J et al (2019) New crosslinked hyaluronan gel for the prevention of intrauterine adhesions after dilation and curettage in patients with delayed miscarriage: a prospective, multicenter, randomized. Controlled Trial J Minim Invasive Gynecol 26(1):94–99
Acunzo G, Guida M, Pellicano M, Tommaselli GA, Di Spiezio SA, Bifulco G et al (2003) Effectiveness of auto-cross-linked hyaluronic acid gel in the prevention of intrauterine adhesions after hysteroscopic adhesiolysis: a prospective, randomized, controlled study. Hum Reprod 18(9):1918–1921
Ducarme G, Davitian C, Zarrouk S, Uzan M, Poncelet C (2006) Interest of auto-cross-linked hyaluronic acid gel in the prevention of intrauterine adhesions after hysteroscopic surgery: a case-control study. J Gynecol Obstet Biol Reprod (Paris) 35(7):691–695
Thubert T, Dussaux C, Demoulin G, Rivain AL, Trichot C, Deffieux X (2015) Influence of auto-cross-linked hyaluronic acid gel on pregnancy rate and hysteroscopic outcomes following surgical removal of intra-uterine adhesions. Eur J Obstet Gynecol Reprod Biol 193:65–69
Sikkink CJ, de Man B, Bleichrodt RP, van Goor H (2006) Auto-cross-linked hyaluronic acid gel does not reduce intra-abdominal adhesions or abscess formation in a rat model of peritonitis. J Surg Res 136(2):255–259
De Clercq K, Schelfhout C, Bracke M, De Wever O, Van Bockstal M, Ceelen W et al (2016) Genipin-crosslinked gelatin microspheres as a strategy to prevent postsurgical peritoneal adhesions: In vitro and in vivo characterization. Biomaterials 96:33–46
van Steensel S, Liu H, Vercoulen TF, Hadfoune M, Breukink SO, Stassen LP et al (2020) Prevention of intra-abdominal adhesions by a hyaluronic acid gel; an experimental study in rats. J Biomater Appl 35(7):887–897
Tang CL, Jayne DG, Seow-Choen F, Ng YY, Eu KW, Mustapha N (2006) A randomized controlled trial of 0.5% ferric hyaluronate gel (Intergel) in the prevention of adhesions following abdominal surgery. Ann Surg 243(4):449–55
Toole BP, Wight TN, Tammi MI (2002) Hyaluronan-cell interactions in cancer and vascular disease. J Biol Chem 277(7):4593–4596
Tan A, Argenta P, Ramirez R, Bliss R, Geller M (2009) The use of sodium hyaluronate-carboxymethylcellulose (HA-CMC) barrier in gynecologic malignancies: a retrospective review of outcomes. Ann Surg Oncol 16(2):499–505
Beck DE, Cohen Z, Fleshman JW, Kaufman HS, van Goor H, Wolff BG, Adhesion Study Group Steering C. (2003) A prospective, randomized, multicenter, controlled study of the safety of Seprafilm adhesion barrier in abdominopelvic surgery of the intestine. Dis Colon Rectum 46(10):1310–9
Hajibandeh S, Hajibandeh S, Saeed S, Bird J, Kannappa L, Ratnayake I (2022) Effect of hyaluronate-based bioresorbable membrane (Seprafilm) on outcomes of abdominal surgery: a meta-analysis and trial sequential analysis of randomised controlled trials. Updates Surg 74(3):865–881
Torres-de la Roche LA, Devassy R, de Wilde MS, Cezar C, Krentel H, Korell M, De Wilde RL (2020) A new approach to avoid ovarian failure as well function-impairing adhesion formation in endometrioma infertility surgery. Arch Gynecol Obstet 301(5):1113–1115
Ziegler N, De Wilde RL (2022) Reduction of adhesion formation after gynaecological adhesiolysis surgery with 4DryField PH - a retrospective, controlled study with second look laparoscopies. J Obstet Gynaecol 42(4):658–664
Ahmad M, Crescenti F (2019) Significant adhesion reduction with 4DryField PH after release of adhesive small bowel obstruction. Surg J (N Y) 5(1):e28–e34
Poehnert D, Neubert L, Klempnauer J, Borchert P, Jonigk D, Winny M (2019) Comparison of adhesion prevention capabilities of the modified starch powder-based medical devices 4DryField((R)) PH and arista AH in the optimized peritoneal adhesion model. Int J Med Sci 16(10):1350–1355
Li MY, Khoo CK, Hunan R (2015) Use of an alternative haemostatic agent, HaemoCer, in minimally invasive and open gynaecological surgeries: a single centre experience. BJOG 122(S1):120
Poehnert D, Neubert L, Winny M (2024) Comparison of adhesion prevention capabilities of the modified starch powder-based medical devices 4DryField((R)) PH, HaemoCer PLUS and StarSil((R)) in the optimized peritoneal adhesion model. Int J Med Sci 21(3):424–430
Ahmad G, Kim K, Thompson M, Agarwal P, O’Flynn H, Hindocha A, Watson A (2020) Barrier agents for adhesion prevention after gynaecological surgery. Cochrane Database Syst Rev 3(3):Cd000475
Funding
This work was funded by PlantTec Medical GmbH, unrestricted educational grant.
Author information
Authors and Affiliations
Contributions
SD Schaefer: project development, data collection, data analysis, manuscript writing. I Alkatout: data collection, data analysis, manuscript editing. N Dornhoefer: data analysis, manuscript editing. J Herrmann: data analysis, manuscript editing. R Klapdor: data analysis, manuscript editing. I Meinhold-Heerlein: data collection, data analysis, manuscript editing. J Meszaros: data analysis, manuscript editing. A Mustea: data analysis, manuscript editing. P Oppelt: data analysis, manuscript editing. M Wallwiener: data analysis, manuscript editing. B Kraemer: project development, data collection, data analysis, manuscript writing.
Corresponding author
Ethics declarations
Conflict of interest
This study was made possible by an unrestricted educational grant from PlantTec Medical GmbH (Lueneburg, Germany). The company had no influence on the authors or results; medical writing and editing were supported by co.medical (Berlin, Germany). The authors declare the following disclosures. Authors who are not listed separately below have no disclosures to declare. SS: royalties from PlantTec Medical GmbH, Baxter, and Eisei; member of advisory board of German working group on gynecological endoscopy (AGE); and vice president of German working group on endometriosis (AGEM). IMH: royalties from PlantTec, Tesaro/GSK, MSD, and Roche and unrestricted research grants from Gedeon Richter, Bayer, Jenapharm, Takeda, and Storz. AM: royalties from PlantTec Medical GmbH, GSK, MSD, Novartis, Olympus, and Stryker. BK: royalties from PlantTec, Baxter, and Medtronic and member of the advisory board of the German working group on gynecological endoscopy (AGE) and the European Society of Gynecological Endoscopy (ESGE).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schaefer, S.D., Alkatout, I., Dornhoefer, N. et al. Prevention of peritoneal adhesions after gynecological surgery: a systematic review. Arch Gynecol Obstet 310, 655–672 (2024). https://doi.org/10.1007/s00404-024-07584-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-024-07584-1