Abstract
Purpose
Pregnancies complicated by placenta accreta spectrum (PAS) are associated with severe maternal morbidities. The aim of this study is to describe the neonatal outcomes in pregnancies complicated with PAS compared with pregnancies not complicated by PAS.
Methods
A retrospective cohort study conducted at a single tertiary center between 03/2011 and 01/2022, comparing women with PAS who underwent cesarean delivery (CD) to a matched control group of women without PAS who underwent CD. We evaluated the following adverse neonatal outcomes: umbilical artery pH < 7.0, umbilical artery base excess ≤ − 12, APGAR score < 7 at 5 min, neonatal intensive care unit (NICU) admission, mechanical ventilation, hypoxic ischemic encephalopathy, seizures and neonatal death. We also evaluated a composite adverse neonatal outcome, defined as the occurrence of at least one of the adverse neonatal outcomes described above. Multivariable regression analysis was used to determine which adverse neonatal outcome were independently associated with the presence of PAS.
Results
265 women with PAS were included in the study group and were matched to 1382 controls. In the PAS group compared with controls, the rate of composite adverse neonatal outcomes was significantly higher (33.6% vs. 18.7%, respectively, p < 0.001). In a multivariable logistic regression analysis, Apgar score < 7 at 5 min, NICU admission and composite adverse neonatal outcome were independently associated with PAS.
Conclusion
Neonates in PAS pregnancies had higher rates of adverse outcomes. Apgar score < 7 at 5 min, NICU admission and composite adverse neonatal outcome were independently associated with PAS.
Similar content being viewed by others
Explore related subjects
Find the latest articles, discoveries, and news in related topics.Avoid common mistakes on your manuscript.
Neonates in placenta accreta spectrum pregnancies have higher rates of adverse neonatal outcomes. Low Apgar score, NICU admission and composite adverse neonatal outcome were independently associated with placenta accreta spectrum. |
Introduction
Placenta accreta spectrum (PAS) is the pathologic placental adherence to the myometrium, most commonly hypothesized as due to a defect in the uterine endometrial-myometrial interface leading to abnormal trophoblast invasion of the myometrium [1]. PAS includes placenta accreta, placenta increta and placenta percreta [2, 3]. The incidence of PAS is estimated to be as high as 1.1% of all births [4], and is rising globally due to an increase in the prevalence of the established risk factors, mainly cesarean deliveries (CD) and also other uterine surgeries such as surgical uterine evacuation, myomectomy and infertility treatments [5,6,7,8].
The most common surgical approach for management of PAS pregnancies is cesarean hysterectomy or peripartum hysterectomy [4]. In recent years, mostly for women interested in future fertility, uterine preserving PAS surgery has been increasingly performed [9, 10].
Pregnancies complicated by PAS are associated with severe maternal morbidities such as life threatening hemorrhage, and damage to adjacent organs, mainly urinary tract and gastrointestinal tract [11]. While maternal outcomes following pregnancies complicated by PAS are well reported and described, reports regarding neonatal outcomes following these pregnancies are scarce. CD is a well-established risk factor for adverse neonatal outcomes compared with vaginal delivery [12], thus it is reasonable that a complicated CD due to PAS may further impact neonatal outcomes. Previous retrospective studies most consistently reported high rates of neonatal intensive care unit (NICU) admissions and a relatively high need for mechanical ventilation in PAS pregnancies [13,14,15,16]. These studies reported only a limited number of neonatal outcomes and did not match them to non-PAS pregnancies.
The aim of this study is to evaluate the association between PAS and adverse neonatal outcomes.
Materials and methods
This was a retrospective matched cohort study conducted at a single tertiary center. Historical data via electronic medical records of pregnant women diagnosed with PAS who underwent CD between 03/2011 and 01/2022 were reviewed. Data were originally collected prospectively including umbilical artery pH and base excess for all women. Women diagnosed with PAS undergoing CD were matched to a control group of women not diagnosed with PAS undergoing a repeat CD with a 1:6 ratio. Vaginal deliveries were excluded. Matching was performed for gestational age at CD and the number of prior CDs.
We excluded pregnancies with intrauterine fetal death, multiple gestations and congenital anomalies.
We evaluated the following adverse neonatal outcomes: umbilical artery pH < 7.0, umbilical artery base excess ≤ − 12, APGAR score < 7 at 5 min, neonatal intensive care unit (NICU) admission, mechanical ventilation, hypoxic ischemic encephalopathy, seizures and neonatal death. We also evaluated a composite adverse neonatal outcome, defined as the occurrence of at least one of the adverse neonatal outcomes described above.
Maternal baseline characteristics included age, body mass index, smoking, diabetes mellitus, hypertensive disease, time interval since last CD, number of previous CDs, in vitro fertilization and emergent CD.
PAS was defined in accordance with the ‘International Federation of Gynecology and Obstetrics’ classification for the clinical diagnosis of placenta accreta spectrum disorders [17].
Hypertensive Disorders of Pregnancy (HDP) were defined as the presence gestational hypertension or pre-eclampsia according to the ACOG most recent practice bulletin [18]. Gestational diabetes mellitus (GDM) was defined according to the values proposed by Carpenter and Coustan [19]. Smoking was defined as smoking at least one cigarette (or equivalent) per day. Intrauterine growth restriction was defined as birthweight percentile of ≤ 10% according to local charts [20]. Hypoxic ischemic encephalopathy was defined in accordance with international guidelines and current literature [21, 22].
PAS management protocol
Sonographic findings suggestive of PAS include irregular placental lacunae, loss of the hypoechoic retroplacental-myometrial clear zone, presence of bridging vessels between the placenta and the bladder wall and interruption of the bladder–uterine interface [23]. All women referred to our tertiary center with sonographic or clinical suspicion for PAS are routinely evaluated by an expert sonographer. Once sonographic PAS diagnosis is established, women are further evaluated by a multidisciplinary team that includes an obstetrical PAS specializing surgeon, a gynecologist, an expert sonographer and an obstetric anesthesiologist.
Uterine preserving procedure
The uterine-preserving procedure is a surgical option for women interested in fertility preservation or who may be at risk of significant bleeding or damage to other organs during a hysterectomy. Following general anesthesia, a preoperative ureteric stent is placed. During the procedure, the surgeons carefully assess the external surface of the uterus for signs of invasion and placental infiltration. The surgeons dissect the bladder from the uterus and ligate the vessels between the uterus to surrounding tissue. Next, an incision is made in the upper segment of the uterus, avoiding the superior edge of the placenta, and the fetus is delivered. The attached placenta is carefully removed, while any large, inseparable sections of retained placenta are removed in en-bloc. The uterine wall is then sutured and reconstructed. To control bleeding, the surgeons may use uterotonic drugs, surgical vessel ligation, an intrauterine balloon tamponade or uterine compression sutures. In cases where there is a high risk of major bleeding, an interventional radiologist may perform a temporary bilateral occlusion of the uterine arteries via internal iliac artery catheterization to reduce bleeding if needed [24].
In some cases, severe intraoperative bleeding precludes uterine preservation. In these cases, after delivery of the fetus, the placenta is left in place, the uterus is immediately sutured, and a hysterectomy is completed.
Statistical analysis
Normality of the data was assessed using the Shapiro–Wilk or Kolmogorov–Smirnov tests. We compared study groups using Mann–Whitney U test. The chi-square and Fisher’s exact tests were used for comparison of categorical variables. A comparison of PAS versus non-PAS was performed. Multivariable regression analysis was used to determine which adverse neonatal outcomes were independently associated with the presence of PAS. Variables reaching a p < 0.05 in the univariate analysis were included in the multivariable regression analysis. Statistical analyses were conducted using the IBM Statistical Package for the Social Sciences (IBM SPSS v.29; IBM Corporation Inc, Armonk, NY, USA).
Results
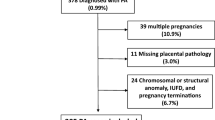
In the study period, there were 99,476 deliveries, 25,931)26.1%) of which were CDs. Out of these, 265 women PAS met inclusion criteria and were matched to a control group of 1,382 women (Fig. 1).
Maternal baseline characteristics are presented in Table 1. Women with PAS pregnancies, compared to women with non-PAS pregnancies, had lower rates of hypertensive disease (3.4% vs 6.9%, respectively, p = 0.031), underwent more CDs (2.0 vs. 2.0, respectively, p = 0.013) and underwent less emergent CDs (17.8% vs. 42.3%, respectively, p < 0.001) and had higher rates of general anesthesia in the current CD (73.2% vs. 16.3%, respectively, p < 0.001). No other characteristics differed significantly between the two groups. Indications for CD are presented in Table S1.
Neonatal outcomes are presented in Table 2. For women with PAS pregnancies, compared to women with non-PAS pregnancies, gestational age at delivery (361 vs. 360 weeks, respectively, p = 0.001) was higher, birthweight (2660.0 g vs. 2625.0, respectively, p = 0.006) was lower. Umbilical artery pH (7.26 vs. 7.28, respectively, p < 0.001) was lower, while the incidence of APGAR score < 7 at 5 min (8.3% vs. 1.0%, respectively, p < 0.001), length of hospital stay (7.0 days vs. 6.0, respectively, p < 0.001), NICU admission (29.8% vs. 18.2%, respectively, p < 0.001), mechanical ventilation (9.8% vs. 3.8%, respectively, p < 0.001) and the neonatal composite score (33.6% vs. 18.7%, respectively, p < 0.001) were significantly higher. No other parameters differed significantly between the two groups.
Adjusted odds ratio for the risk of adverse neonatal outcomes among patients with PAS compared to controls is presented in Table 3. The following variables were independently associated with PAS: Apgar < 7 at 5 min [adjusted odds ratio (aOR) 95% confidence interval (CI) 2.21 (1.07–4.56), p = 0.031], NICU admission (aOR 95% CI 1.84 (1.05–3.24), p = 0.033) and composite adverse neonatal outcome (aOR 95% CI 2.38 (1.40–4.05), p = 0.001).
Discussion
The aim of this study is to describe the neonatal outcomes in pregnancies complicated with PAS compared with pregnancies not complicated by PAS.
We found that Apgar scores below 7, NICU admissions and composite adverse neonatal outcome were independent parameters positively associated with adverse neonatal outcomes.
The occurrence of adverse neonatal outcomes was significantly higher in the PAS group compared to the non-PAS group. Several previous studies described neonatal outcomes among women undergoing surgery due to PAS. These include gestational age at delivery, birthweight, Apgar scores, rates of NICU admissions and need for neonatal mechanical ventilation [13, 15]. Kasraeian et al. [13] described neonatal outcomes in 198 cesarean hysterectomy cases in PAS. Mean gestational age at delivery was at 34 weeks, mean birthweight was 2213 g, Apgar scores in the first and fifth minute were above 6 for most of the neonates. Fifty-seven percent of the neonates were admitted to the NICU. Palacios–Jaraquemada et al. [15] described neonatal outcomes in 315 PAS pregnancies undergoing uterine preserving CD. 5-min Apgar scores were high, admission to the NICU in 81–100% and mechanical ventilation in 2–25% in all cases, rates were higher with higher degree of placental invasion. Eshkoli et al. [16] reviewed 139 PAS-pregnancies and did not find increased risk for adverse perinatal outcomes such as low Apgar scores at 1 and 5 min and perinatal mortality. Fishel Bartal et al. [25] reviewed 109 PAS pregnancies and reported composite neonatal morbidity (Apgar’s score < 5 at 5 min, mechanical ventilation, or respiratory distress syndrome) in 30% of the patients. Mean birth weight was 1728–2446 g and was lower in emergent CDs compared with planned CDs. Of note, compared to our research all these studies did not compare PAS pregnancies to a non-PAS group, had a relatively smaller amount of patients and described a relatively smaller amount of adverse outcomes. Fetal pH levels at birth were not described in any of these previous studies. These studies mainly showed high rates of NICU admissions and mechanical ventilation in PAS pregnancies. Apgar scores were inconsistent, while in our study Apgar score < 7 was significantly lower in PAS pregnancies. Gestational age at birth and birthweight were earlier and lower compared to our study’s results.
We found higher birthweights in the PAS group compared with the non-PAS group. Similarly, Jauniaux et. al found that fetal growth was not impaired when comparing pregnancies complicated by placenta previa with PAS (82 pregnancies) and with pregnancies without PAS (209 pregnancies) [26]. Nevertheless, other studies did demonstrate that preterm birth and small for gestational age infants appear to be more common in pregnancies complicated by PAS [27,28,29]. The abnormal fetal growth in pregnancies complicated by PAS may result from pathological implantation of the placenta that interferes with normal placental function. Gielchinsky et al. [27] compared 310 PAS pregnancies to non-PAS control group and found significantly more preterm deliveries and small for gestational age neonates in the PAS group, concluding that these findings may arise from pathological implantation of the placenta, resulting in interference with normal fetal growth.
Adverse neonatal outcome in PAS pregnancies, after adjustment for possible confounders, were independently positively associated with Apgar scores below 7 at 5 min, NICU admissions and composite adverse neonatal outcome. Possible reasons for the worse neonatal outcome in PAS pregnancies compared to non-PAS pregnancies may be attributed to the complexity of CD in a PAS pregnancy, as reported in previous studies, including higher rates of maternal bleeding and maternal blood transfusion [16]. The uterine incision in CD in a PAS pregnancy sometimes includes entry through the placenta before extracting the fetus. The abnormal placentation associated with PAS, as previously described [27], may allow less blood flow and thus lower reserves for the fetus in the minutes from beginning of surgery to the evacuation, thus rendering the fetus to worse outcomes. However, the exact reasons for this outcomes are rather intriguing and prompt further studies and research to understand them and hopefully manage to advise how to avoid them.
This study is not without limitations. Its retrospective design potentially introduces biases inherent to this type of study such as information bias due to different potential confounders. It is a single-center study, thus limited by the risk of bias due to individual clinician decisions regarding patient treatment. Since our tertiary medical center is a regional referral center for PAS pregnancies, the cohort presented in this study may be exposed to referral bias and might not be applicable and generalizable to other medical centers. Furthermore, the possibility of bias due to lack of adjustment for all possible confounders cannot be ruled out.
The main strength of this study is the inclusion of a relatively high number of PAS pregnancies matched to a larger cohort of non-PAS pregnancies describing the outcome of many neonatal characteristics never described before. Matching was performed for gestational age at CD and the number of prior CDs, thus limiting the possibility of selection bias. All PAS pregnancies included in our study were prenatally diagnosed and confirmed clinically or histologically.
Further prospective research including larger populations is required to further ratify these results and better inform and consult women in risk for PAS regarding their infants possible adverse outcomes.
Conclusion
Pregnancies complicated by PAS are associated with adverse neonatal outcomes. This study demonstrated that neonates in PAS pregnancies had a higher proportion of Apgar score < 7 at 5 min, NICU admission and composite adverse neonatal outcomes compared with controls. Further studies are needed to underline the association of PAS and these adverse neonatal outcomes.
Data availability
Not applicable.
References
American College of Obstetricians and Gynecologists; Society for Maternal-Fetal Medicine (2018) Obstetric care consensus no. 7: placenta accreta spectrum, (in eng). Obstet Gynecol 132(6):e259–e275. https://doi.org/10.1097/AOG.0000000000002983
Belfort MA (2010) Placenta accreta, (in eng). Am J Obstet Gynecol 203(5):430–439. https://doi.org/10.1016/j.ajog.2010.09.013
Bartels HC et al (2021) Maternal morbidity in placenta accreta spectrum following introduction of a multi-disciplinary service compared to standard care: an Irish perspective, (in eng). Ir J Med Sci 190(4):1451–1457. https://doi.org/10.1007/s11845-020-02473-3
Jauniaux E, Bunce C, Grønbeck L, Langhoff-Roos J (2019) Prevalence and main outcomes of placenta accreta spectrum: a systematic review and meta-analysis, (in eng). Am J Obstet Gynecol 221(3):208–218. https://doi.org/10.1016/j.ajog.2019.01.233
Hull AD, Moore TR (2011) Multiple repeat cesareans and the threat of placenta accreta: incidence, diagnosis, management, (in eng). Clin Perinatol 38(2):285–296. https://doi.org/10.1016/j.clp.2011.03.010
Baldwin HJ et al (2018) Antecedents of abnormally invasive placenta in primiparous women: risk associated with gynecologic procedures, (in eng). Obstet Gynecol 131(2):227–233. https://doi.org/10.1097/AOG.0000000000002434
Baldwin HJ, Nippita TA, Torvaldsen S, Ibiebele I, Ford JB, Patterson JA (2020) Outcomes of subsequent births after placenta accreta spectrum, (in eng). Obstet Gynecol 136(4):745–755. https://doi.org/10.1097/AOG.0000000000004051
Hung TH, Shau WY, Hsieh CC, Chiu TH, Hsu JJ, Hsieh TT (1999) Risk factors for placenta accreta, (in eng). Obstet Gynecol 93(4):545–550. https://doi.org/10.1016/s0029-7844(98)00460-8
Fox KA et al (2015) Conservative management of morbidly adherent placenta: expert review, (in eng). Am J Obstet Gynecol 213(6):755–760. https://doi.org/10.1016/j.ajog.2015.04.034
Sentilhes L et al (2010) Maternal outcome after conservative treatment of placenta accreta, (in eng). Obstet Gynecol 115(3):526–534. https://doi.org/10.1097/AOG.0b013e3181d066d4
Cahill AG et al (2018) Placenta accreta spectrum, (in eng). Am J Obstet Gynecol 219(6):B2–B16. https://doi.org/10.1016/j.ajog.2018.09.042
Mariam BG, Tilahun T, Merdassa E, Tesema D (2021) Indications, Outcome and risk factors of cesarean delivery among pregnant women utilizing delivery services at selected Public Health Institutions, Oromia Region, South West Ethiopia, (in eng). Patient Relat Outcome Meas 12:227–236. https://doi.org/10.2147/PROM.S304672
Kasraeian M et al (2021) A 5-year experience on perinatal outcome of placenta accreta spectrum disorder managed by cesarean hysterectomy in southern Iranian women, (in eng). BMC Womens Health 21(1):243. https://doi.org/10.1186/s12905-021-01389-z
Morlando M et al (2021) Maternal and neonatal outcomes in planned versus emergency cesarean delivery for placenta accreta spectrum: a multinational database study, (in eng). Acta Obstet Gynecol Scand 100(Suppl 1):41–49. https://doi.org/10.1111/aogs.14120
Palacios-Jaraquemada JM, Basanta N, Fiorillo A, Labrousse C, Martínez M (2021) Neonatal outcome after conservative-reconstructive surgery for placenta accreta spectrum disorders, (in eng). J Matern Fetal Neonatal Med. https://doi.org/10.1080/14767058.2021.1873944
Eshkoli T, Weintraub AY, Sergienko R, Sheiner E (2013) Placenta accreta: risk factors, perinatal outcomes, and consequences for subsequent births, (in eng). Am J Obstet Gynecol 208(3):219.e1–7. https://doi.org/10.1016/j.ajog.2012.12.037
Jauniaux E, Ayres-de-Campos D, Langhoff-Roos J, Fox KA, Collins S (2019) FIGO Placenta Accreta Diagnosis and Management Expert Consensus Panel, FIGO classification for the clinical diagnosis of placenta accreta spectrum disorders, (in eng). Int J Gynaecol Obstet 146(1):20–24. https://doi.org/10.1002/ijgo.12761
American College of Obstetrics and Gynecology (2020) Gestational hypertension and preeclampsia: ACOG practice bulletin summary, number 222, (in eng). Obstet Gynecol 135(6):1492–1495 https://doi.org/10.1097/AOG.0000000000003892
Carpenter MW, Coustan DR (1982) Criteria for screening tests for gestational diabetes, (in eng). Am J Obstet Gynecol 144(7):768–773. https://doi.org/10.1016/0002-9378(82)90349-0
Dollberg S, Haklai Z, Mimouni FB, Gorfein I, Gordon ES (2005) Birth weight standards in the live-born population in Israel, (in eng). Isr Med Assoc J 7(5):311–314
American College of Obstetricians and Gynecologists Committee on Obstetric Practice (2005) ACOG Committee Opinion. Number 326, December 2005. Inappropriate use of the terms fetal distress and birth asphyxia, (in eng). Obstet Gynecol 106(6):1469–1470. https://doi.org/10.1097/00006250-200512000-00056
Shah PS, Beyene J, To T, Ohlsson A, Perlman M (2006) Postasphyxial hypoxic-ischemic encephalopathy in neonates: outcome prediction rule within 4 hours of birth, (in eng). Arch Pediatr Adolesc Med 160(7):729–736. https://doi.org/10.1001/archpedi.160.7.729
Jauniaux E, Collins S, Burton GJ (2018) Placenta accreta spectrum: pathophysiology and evidence-based anatomy for prenatal ultrasound imaging, (in eng). Am J Obstet Gynecol 218(1):75–87. https://doi.org/10.1016/j.ajog.2017.05.067
Sivan E et al (2010) Prophylactic pelvic artery catheterization and embolization in women with placenta accreta: can it prevent cesarean hysterectomy? (in eng). Am J Perinatol 27(6):455–461. https://doi.org/10.1055/s-0030-1247599
Fishel Bartal M et al (2022) Planned versus unplanned delivery for placenta accreta spectrum, (in eng). Am J Perinatol 39(3):252–258. https://doi.org/10.1055/s-0040-1714676
Jauniaux E et al (2019) Impact of placenta previa with placenta accreta spectrum disorder on fetal growth, (in eng). Ultrasound Obstet Gynecol 54(5):643–649. https://doi.org/10.1002/uog.20244
Gielchinsky Y, Mankuta D, Rojansky N, Laufer N, Gielchinsky I, Ezra Y (2004) Perinatal outcome of pregnancies complicated by placenta accreta, (in eng). Obstet Gynecol 104(3):527–530. https://doi.org/10.1097/01.AOG.0000136084.92846.95
Seet EL, Kay HH, Wu S, Terplan M (2012) Placenta accreta: depth of invasion and neonatal outcomes, (in eng). J Matern Fetal Neonatal Med 25(10):2042–2045. https://doi.org/10.3109/14767058.2012.678429
Liang Y et al (2022) Risk factors and pregnancy outcome in women with a history of cesarean section complicated by placenta accreta. Matern-Fetal Med 4(3):179–185. https://doi.org/10.1097/fm9.0000000000000142
Funding
Open access funding provided by Tel Aviv University. The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. The first draft of the manuscript was written by ST-C and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. ST-C Protocol/project development; Data collection and management; Data analysis; Manuscript writing/editing. EC Protocol/project development; Data collection and management. LF Data collection and management; Data analysis. NM Data collection and management; Data analysis. AO Data collection and management; Data analysis. GL Data analysis; Manuscript writing/editing. RM Protocol/project development; Data collection or management; Data analysis; Manuscript writing/editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Institutional Review Board approval (9805-22-SMC) was obtained for the study waiving informed consent. The Institutional Review Board approved the use of patient database retrospectively.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Toussia-Cohen, S., Castel, E., Friedrich, L. et al. Neonatal outcomes in pregnancies complicated by placenta accreta- a matched cohort study. Arch Gynecol Obstet 310, 269–275 (2024). https://doi.org/10.1007/s00404-023-07353-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-023-07353-6