Abstract
Objective
Quantitative real-time PCR (qPCR) is used to detect the differential expression of circular RNAs in patients of premature ovarian insufficiency (POI), to explore the new biomarkers of POI that can be detected from blood as soon as possible.
Methods
The study collected plasma samples from 30 patients in POI group and 30 normal people group who meet the inclusion criteria, who visited the gynecology clinic of The First Affiliated Hospital of Guangzhou University of Chinese Medicine from July 2019 to December 2020. Then, circRNAs in plasma were extracted for qPCR validation.
Results
1. qPCR technology was performed on hsa_circRNA_008901 and hsa_circRNA_403959, and it was found that the levels of both were considerably downregulated in POI group. Clinical evaluation showed that both hsa_circRNA_008901 and hsa_circRNA_403959 have good diagnostic value for POI.
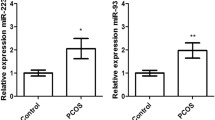
2. According to miRNA Regulatory Element (MRE) analysis, the predicted target miRNAs of hsa_circRNA_008901 are: hsa-miR-548c-3p, hsa-miR-924, hsa-miR-4677-5p, hsa-miR-6786-3p and hsa-miR-7974; the predicted target miRNAs of hsa_circRNA_403959 are: hsa-miR-1207-5p, hsa-miR-4691-5p, hsa-miR-4763-3p, hsa-miR-6807-5p and hsa-miR-7160-5p.
Conclusion
Compared with the normal group, the expression levels of hsa_circRNA_008901 and hsa_circRNA_403959 in the POI group were downregulated, suggesting that these two circRNAs may be potential biomarkers of POI. Bioinformatics analysis indicated that hsa_circRNA_008901 and hsa_circRNA_403959 may regulate their binding miRNA through the action form of “molecular sponge”, and then regulate the signaling pathway regulated by miRNA, and ultimately affect the disease progression of POI.








Similar content being viewed by others
Data availability
The detailed procedures of methods, 8 figures and 4 table are attached.
References
Nelson LM (2009) Clinical practice. Primary ovarian insufficiency. N Engl J Med 360(6):606–614
Shi H, Li X (2018) Effect of premature ovarian insufficiency on cardiovascular disease. Chin J Pract Gynecol Obstet 34(03):248–251
Lei X, Tan X (2018) Effect of premature ovarian insufficiency on bone metabolism. Chin J Pract Gynecol Obstet 34(03):245–248
Stevenson JC, Collins P, Hamoda H et al (2021) Cardiometabolic health in premature ovarian insufficiency. Climacteric 24(5):474–480
Coulam CB, Adamson SC, Annegers JF (1986) Incidence of premature ovarian failure. Obstet Gynecol 67(4):604–606
Golezar S, Ramezani Tehrani F, Khazaei S, Ebadi A, Keshavarz Z (2019) The global prevalence of primary ovarian insufficiency and early menopause: a meta-analysis. Climacteric 22(4):403–411
Jeck WR, Sorrentino JA, Wang K et al (2013) Circular RNAs are abundant, conserved, and associated with ALU repeats. RNA 19(2):141–157
Salzman J, Gawad C, Wang PL, Lacayo N, Brown PO (2012) Circular RNAs are the predominant transcript isoform from hundreds of human genes in diverse cell types. PLoS ONE 7(2):e30733
Salmena L, Poliseno L, Tay Y, Kats L, Pandolfi PP (2011) A ceRNA hypothesis: the Rosetta stone of a hidden RNA language? Cell 146(3):353–358
Hansen TB, Jensen TI, Clausen BH et al (2013) Natural RNA circles function as efficient microRNA sponges. Nature 495(7441):384–388
Zhang X, Ying X, Xu B (2020) Research progress of circular RNA in female reproductive system. J Int Reprod Health/Family Plan 39(04):308–313
Meng Y, Jiang H (2022) Research progress on circRNAs and their relationship with animal reproduction. Heilongjiang Anim Sci Vet Med 03:39–43
Cai Y, Lei X, Chen Z, Mo Z (2020) The roles of cirRNA in the development of germ cells. Acta Histochem 122(3):151506
Chen S (2019) CircRNA expression profile in plasma of patients with premature ovarian insufficiency and investigation of TCM constitution at different stages [master]. Guangzhou university of chinese medicine, Guangzhou
Chen Z, Tian Q, Qiao J et al (2017) Chinese expert consensus on clinical diagnosis and treatment of premature ovarian insufficiency. Chin J Obstet Gynecol 52(09):577–581
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116(2):281–297
Dandare A, Rabia G, Khan MJ (2021) In silico analysis of non-coding RNAs and putative target genes implicated in metabolic syndrome. Comput Biol Med 130:104229
Wang H, Chen X, Yang B, Xia Z, Chen Q (2020) MiR-924 as a tumor suppressor inhibits non-small cell lung cancer by inhibiting RHBDD1/Wnt/beta-catenin signaling pathway. Cancer Cell Int 20:491
Liu Y, Wang S, Pan S, Yan Q, Li Y, Zhao Y (2022) Circ_0000463 contributes to the progression and glutamine metabolism of non-small-cell lung cancer by targeting miR-924/SLC1A5 signaling. J Clin Lab Anal 36(1):e24116
Dong R, Liu J, Sun W, Ping W (2020) Comprehensive analysis of aberrantly expressed profiles of lncRNAs and miRNAs with associated ceRNA Network in lung adenocarcinoma and lung squamous cell carcinoma. Pathol Oncol Res 26(3):1935–1945
Dang W, Qin Z, Fan S et al (2016) miR-1207-5p suppresses lung cancer growth and metastasis by targeting CSF1. Oncotarget 7(22):32421–32432
Raitoharju E, Seppala I, Oksala N et al (2014) Blood microRNA profile associates with the levels of serum lipids and metabolites associated with glucose metabolism and insulin resistance and pinpoints pathways underlying metabolic syndrome: the cardiovascular risk in young finns study. Mol Cell Endocrinol 391(1–2):41–49
Choi JH, Kwon SM, Moon SU et al (2021) TPRG1-AS1 induces RBM24 expression and inhibits liver cancer progression by sponging miR-4691-5p and miR-3659. Liver Int 41(11):2788–2800
Nie X, He M, Wang J et al (2020) Circulating miR-4763-3p is a novel potential biomarker candidate for human adult fulminant myocarditis. Mol Ther Methods Clin Dev 17:1079–1087
Arslan S, Bayyurt B, Engin A, Bakir M (2021) MicroRNA analysis from acute to convalescence in Crimean Congo hemorrhagic fever. J Med Virol 93(8):4729–4737
Imai F, Kishi H, Nakao K, Nishimura T, Minegishi T (2014) IL-6 up-regulates the expression of rat LH receptors during granulosa cell differentiation. Endocrinology 155(4):1436–1444
Ishiguro K, Kojima T, Taguchi O, Saito H, Muramatsu T, Kadomatsu K (1999) Syndecan-4 expression is associated with follicular atresia in mouse ovary. Histochem Cell Biol 112(1):25–33
Franke TF, Hornik CP, Segev L, Shostak GA, Sugimoto C (2003) PI3K/Akt and apoptosis: size matters. Oncogene 22(56):8983–8998
Li D, Zhou L, Sun Z (2020) Research advances in premature ovarian insufficiency-related signaling pathways. Chin J Comp Med 30(02):121–127
Maidarti M, Anderson RA, Telfer EE (2020) Crosstalk between PTEN/PI3K/Akt signalling and DNA damage in the oocyte: implications for primordial follicle activation oocyte quality ageing. Cells 9(1):200
el Arafa SA, Zhu Q, Barakat BM et al (2009) Tangeretin sensitizes cisplatin-resistant human ovarian cancer cells through downregulation of phosphoinositide 3-kinase/Akt signaling pathway. Cancer Res 69(23):8910–8917
McGee EA, Hsueh AJ (2000) Initial and cyclic recruitment of ovarian follicles. Endocr Rev 21(2):200–214
Salvioli S, Capri M, Valensin S et al (2006) Inflamm-aging, cytokines and aging: state of the art, new hypotheses on the role of mitochondria and new perspectives from systems biology. Curr Pharm Des 12(24):3161–3171
Lee SH, Lee JH, Oh EY et al (2013) Ethanol extract of Cnidium officinale exhibits anti-inflammatory effects in BV2 microglial cells by suppressing NF-kappaB nuclear translocation and the activation of the PI3K/Akt signaling pathway. Int J Mol Med 32(4):876–882
Huang Y, Lv Y, Qi T et al (2021) Metabolic profile of women with premature ovarian insufficiency compared with that of age-matched healthy controls. Maturitas 148:33–39
Anagnostis P, Christou K, Artzouchaltzi AM et al (2019) Early menopause and premature ovarian insufficiency are associated with increased risk of type 2 diabetes: a systematic review and meta-analysis. Eur J Endocrinol 180(1):41–50
Schultze SM, Hemmings BA, Niessen M, Tschopp O (2012) PI3K/AKT, MAPK and AMPK signalling: protein kinases in glucose homeostasis. Expert Rev Mol Med 14:e1
Chi Y, Li J, Guan Y, Yang J (2010) Regulation of glucose metabolism by PI3K-Akt signaling pathway. Chin J Biochem Mol Biol 26(10):879–885
Funding
This research was supported by Science and Technology Program of Guangdong Province (2016A020215180) and Research Project of Traditional Chinese Medicine Bureau of Guangdong Province (20231170).
Author information
Authors and Affiliations
Contributions
Conception and design: YL, YZ. Acquisition of data: ZW, CZ, YO, YF. Analysis and interpretation of data: YZ; writing, review, and/or revision of the manuscript: YL, YZ.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Consent for publication
The authors confirmed that we have obtained written consent from the patient to publish the manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Z., Zheng, Y., Zhong, C. et al. Circular RNA as new serum metabolic biomarkers in patients with premature ovarian insufficiency. Arch Gynecol Obstet 308, 1871–1879 (2023). https://doi.org/10.1007/s00404-023-07219-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-023-07219-x