Abstract
Purpose
The purpose of this study was to identify if switching from intramuscular (IM) to vaginal progesterone compared to staying on IM progesterone after a positive pregnancy test following embryo transfer (ET) is associated with miscarriage risk.
Methods
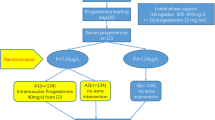
A retrospective cohort study was performed in a private university-affiliated fertility clinic and included women aged 18–50 years with a positive pregnancy test following ET. The two groups studied were: women who stayed on IM progesterone following a positive pregnancy test and those who switched to vaginal progesterone after a positive test. The main outcome measured was risk of miscarriage < 24 weeks gestation as a proportion of non-biochemical pregnancies.
Results
1988 women were included in the analysis. Among the baseline characteristics, the presence of prior miscarriages as well as prior failed ETs, and frozen cycles (vs fresh) as type of transfer were associated with IM progesterone use (p values ≤ 0.01). As per miscarriage risk < 24 weeks, 22.4% (274/1221) of patients in the IM progesterone group experienced a miscarriage compared with 20.7% (159/767) in the vaginal progesterone group (OR 0.90; 95% CI 0.73–1.13). A multivariable logistic regression model revealed an adjusted OR (aOR) of 0.97 (95% CI 0.77–1.22).
Conclusion
This study suggests that switching from IM to vaginal progesterone after a positive pregnancy test following an ET is not associated with miscarriage risk. Considering that IM progesterone imposes substantial discomfort, this study offers reassurance and some flexibility in treatment protocols. Further prospective studies are necessary to corroborate the results of this study.

Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study is available within the article. The final dataset used for the analysis in this study is available from the corresponding author upon reasonable request.
References
Fatemi HM, Popovic-Todorovic B, Papanikolaou E, Donoso P, Devroey P (2007) An update of luteal phase support in stimulated IVF cycles. Hum Reprod Update 13(6):581–590. https://doi.org/10.1093/humupd/dmm021
van der Linden M, Buckingham K, Farquhar C, Kremer JAM (2015) Metwally M (2015) Luteal phase support for assisted reproduction cycles. Cochrane Database Syst Rev 7:CD009154
Beltsos AN, Sanchez MD, Doody KJ, Bush MR, Domar AD, Collins MG (2014) Patients’ administration preferences: progesterone vaginal insert (Endometrin(R)) compared to intramuscular progesterone for Luteal phase support. Reprod Health 11:78. https://doi.org/10.1186/1742-4755-11-78
Propst AM, Hill JA, Ginsburg ES, Hurwitz S, Politch J, Yanushpolsky EH (2001) A randomized study comparing Crinone 8% and intramuscular progesterone supplementation in in vitro fertilization-embryo transfer cycles. Fertil Steril 76(6):1144–1149. https://doi.org/10.1016/s0015-0282(01)02872-2
Doody K, Bush M, Collins M (2012) Progesterone supplementation for luteal support: efficacy and patient experiences with vaginal inserts (Endometrin®) versus intramuscular injection. Fertil Steril 97(3):S18. https://doi.org/10.1016/j.fertnstert.2012.01.040
Yanushpolsky E, Hurwitz S, Greenberg L, Racowsky C, Hornstein M (2010) Crinone vaginal gel is equally effective and better tolerated than intramuscular progesterone for luteal phase support in in vitro fertilization-embryo transfer cycles: a prospective randomized study. Fertil Steril 94(7):2596–2599. https://doi.org/10.1016/j.fertnstert.2010.02.033
Cicinelli E, Schonauer LM, Galantino P, Matteo MG, Cassetta R, Pinto V (2000) Mechanisms of uterine specificity of vaginal progesterone. Hum Reprod 15(Suppl 1):159–165. https://doi.org/10.1093/humrep/15.suppl_1.159
Kahraman S, Karagozoglu SH, Karlikaya G (2010) The efficiency of progesterone vaginal gel versus intramuscular progesterone for luteal phase supplementation in gonadotropin-releasing hormone antagonist cycles: a prospective clinical trial. Fertil Steril 94(2):761–763. https://doi.org/10.1016/j.fertnstert.2009.10.004
Schoolcraft WB, Hesla JS, Gee MJ (2000) Experience with progesterone gel for luteal support in a highly successful IVF programme. Hum Reprod 15(6):1284–1288
Smitz J, Devroey P, Faguer B, Bourgain C, Camus M, Van Steirteghem AC (1992) A prospective randomized comparison of intramuscular or intravaginal natural progesterone as a luteal phase and early pregnancy supplement. Hum Reprod 7(2):168–175. https://doi.org/10.1093/oxfordjournals.humrep.a137611
Zarutskie PW, Phillips JA (2009) A meta-analysis of the route of administration of luteal phase support in assisted reproductive technology: vaginal versus intramuscular progesterone. Fertil Steril 92(1):163–169. https://doi.org/10.1016/j.fertnstert.2009.02.018
Abdelhakim AM, Abd-ElGawad M, Hussein RS, Abbas AM (2020) Vaginal versus intramuscular progesterone for luteal phase support in assisted reproductive techniques: a systematic review and meta-analysis of randomized controlled trials. Gynecol Endocrinol 36(5):389–397. https://doi.org/10.1080/09513590.2020.1727879
Silverberg KM, Vaughn TC, Hansard LJ, Burger NZ, Minter T (2012) Vaginal (Crinone 8%) gel vs. intramuscular progesterone in oil for luteal phase support in in vitro fertilization: a large prospective trial. Fertil Steril 97(2):344–348. https://doi.org/10.1016/j.fertnstert.2011.11.018
Unfer V, Casini ML, Costabile L, Gerli S, Baldini D, Di Renzo GC (2004) 17 alpha-hydroxyprogesterone caproate versus intravaginal progesterone in IVF-embryo transfer cycles: a prospective randomized study. Reprod Biomed Online 9(1):17–21
Dal Prato L, Bianchi L, Cattoli M, Tarozzi N, Flamigni C, Borini A (2008) Vaginal gel versus intramuscular progesterone for luteal phase supplementation: a prospective randomized trial. Reprod Biomed Online 16(3):361–367
Chi HB, Liu NN, Li R, Tao LY, Chen LX, Qiao J (2018) Comparison of vaginal gel and intramuscular progesterone for in vitro fertilization and embryo transfer with gonadotropin-releasing hormone antagonist protocol. Chin Med J (Engl) 131(13):1557–1561. https://doi.org/10.4103/0366-6999.235106
Damario MA, Goudas VT, Session DR, Hammitt DG, Dumesic DA (1999) Crinone 8% vaginal progesterone gel results in lower embryonic implantation efficiency after in vitro fertilization-embryo transfer. Fertil Steril 72(5):830–836. https://doi.org/10.1016/s0015-0282(99)00364-7
Ho CH, Chen SU, Peng FS, Chang CY, Yang YS (2008) Luteal support for IVF/ICSI cycles with Crinone 8% (90 mg) twice daily results in higher pregnancy rates than with intramuscular progesterone. J Chin Med Assoc 71(8):386–391. https://doi.org/10.1016/s1726-4901(08)70088-1
Jabara S, Barnhart K, Schertz JC, Patrizio P (2009) Luteal phase bleeding after IVF cycles: comparison between progesterone vaginal gel and intramuscular progesterone and correlation with pregnancy outcomes. J Exp Clin Assist Reprod 6:6
Khan N, Richter KS, Newsome TL, Blake EJ, Yankov VI (2009) Matched-samples comparison of intramuscular versus vaginal progesterone for luteal phase support after in vitro fertilization and embryo transfer. Fertil Steril 91(6):2445–2450. https://doi.org/10.1016/j.fertnstert.2008.03.072
Mitwally MF, Diamond MP, Abuzeid M (2010) Vaginal micronized progesterone versus intramuscular progesterone for luteal support in women undergoing in vitro fertilization-embryo transfer. Fertil Steril 93(2):554–569. https://doi.org/10.1016/j.fertnstert.2009.02.047
Papaleo E, Quaranta L, Molgora M (2010) Intramuscular vs intravaginal natural progesterone in patients undergoing in vitro fertilization-embryo transfer cycles. A retrospective observational, case-control study. Eur Rev Med Pharmacol Sci 14(2):103–106
Satir F, Toptas T, Inel M, Erman-Akar M, Taskin O (2013) Comparison of intravaginal progesterone gel and intramuscular 17-alpha-hydroxyprogesterone caproate in luteal phase support. Exp Ther Med 5(6):1740–1744. https://doi.org/10.3892/etm.2013.1049
Devine K, Richter KS, Jahandideh S, Widra EA, McKeeby JL (2021) Intramuscular progesterone optimizes live birth from programmed frozen embryo transfer: a randomized clinical trial. Fertil Steril 116(3):633–643. https://doi.org/10.1016/j.fertnstert.2021.04.013
Wang Y, He Y, Zhao X, Ji X, Hong Y, Wang Y, Zhu Q, Xu B, Sun Y (2015) Crinone gel for luteal phase support in frozen-thawed embryo transfer cycles: a prospective randomized clinical trial in the Chinese population. PLoS ONE 10(7):e0133027. https://doi.org/10.1371/journal.pone.0133027
Haddad G, Saguan DA, Maxwell R, Thomas MA (2007) Intramuscular route of progesterone administration increases pregnancy rates during non-downregulated frozen embryo transfer cycles. J Assist Reprod Genet 24(10):467–470. https://doi.org/10.1007/s10815-007-9168-z
Kaser DJ, Ginsburg ES, Missmer SA, Correia KF, Racowsky C (2012) Intramuscular progesterone versus 8% Crinone vaginal gel for luteal phase support for day 3 cryopreserved embryo transfer. Fertil Steril 98(6):1464–1469. https://doi.org/10.1016/j.fertnstert.2012.08.007
Mathur N, Uhler ML, Feinberg EC (2013) Comparison of luteal phase support regimens in frozen embryo transfer (FET) cycles from comprehensive chromosome screening. Fertil Steril 1:S298
Carr BR, Doody KJ (2009) Comparison of endometrin and intramuscular progesterone for frozen-thawed embryo transfer. Fertil Steril 1:S244
Shapiro DB, Pappadakis JA, Ellsworth NM, Hait HI, Nagy ZP (2014) Progesterone replacement with vaginal gel versus i.m. injection: cycle and pregnancy outcomes in IVF patients receiving vitrified blastocysts. Hum Reprod 29(8):1706–1711. https://doi.org/10.1093/humrep/deu121
Bakkensen JB, Racowsky C, Thomas AM, Lanes A, Hornstein MD (2020) Intramuscular progesterone versus 8% Crinone vaginal gel for luteal phase support following blastocyst cryopreserved single embryo transfer: a retrospective cohort study. Fertil Res Pract 6:10. https://doi.org/10.1186/s40738-020-00079-y
Casper RF (2014) Luteal phase support for frozen embryo transfer cycles: intramuscular or vaginal progesterone? Fertil Steril 101(3):627–628. https://doi.org/10.1016/j.fertnstert.2014.01.018
Bender Atik R, Christiansen OB, Elson J, Kolte AM, Lewis S, Middeldorp S, Nelen W, Peramo B, Quenby S, Vermeulen N, Goddijn M (2018) ESHRE guideline: recurrent pregnancy loss. Hum Reprod Open 2:004. https://doi.org/10.1093/hropen/hoy004
Devine K, Richter KS, Widra EA, McKeeby JL (2018) Vitrified blastocyst transfer cycles with the use of only vaginal progesterone replacement with Endometrin have inferior ongoing pregnancy rates: results from the planned interim analysis of a three-arm randomized controlled noninferiority trial. Fertil Steril 109(2):266–275. https://doi.org/10.1016/j.fertnstert.2017.11.004
Tal R, Seifer DB (2017) Ovarian reserve testing: a user’s guide. Am J Obstet Gynecol 217(2):129–140. https://doi.org/10.1016/j.ajog.2017.02.027
Wambach C, Ambartsumyan G, Surrey M, Hill D, Danzer H (2011) Mode of luteal phase progesterone support after frozen embryo transfer does not affect live birth rates. Fertil Steril 1:S26
Lightman A, Kol S, Itskovitz-Eldor J (1999) A prospective randomized study comparing intramuscular with intravaginal natural progesterone in programmed thaw cycles. Hum Reprod 14(10):2596–2599. https://doi.org/10.1093/humrep/14.10.2596
Schoolcraft WB, Katz-Jaffe MG (2013) Comprehensive chromosome screening of trophectoderm with vitrification facilitates elective single-embryo transfer for infertile women with advanced maternal age. Fertil Steril 100(3):615–619. https://doi.org/10.1016/j.fertnstert.2013.07.1972
Grifo JA, Hodes-Wertz B, Lee HL, Amperloquio E, Clarke-Williams M, Adler A (2013) Single thawed euploid embryo transfer improves IVF pregnancy, miscarriage, and multiple gestation outcomes and has similar implantation rates as egg donation. J Assist Reprod Genet 30(2):259–264. https://doi.org/10.1007/s10815-012-9929-1
Yang R, Guan Y, Perrot V, Ma J, Li R (2021) Comparison of the long-acting GnRH agonist follicular protocol with the GnRH antagonist protocol in women undergoing in vitro fertilization: a systematic review and meta-analysis. Adv Ther 38(5):2027–2037. https://doi.org/10.1007/s12325-020-01612-7
Yang AM, Xu X, Han Y, Wei JJ, Hao GM, Cui N, Zhao ZM, Wang W, Huang X (2021) Risk factors for different types of pregnancy losses: analysis of 15,210 pregnancies after embryo transfer. Front Endocrinol (Lausanne) 12:683236. https://doi.org/10.3389/fendo.2021.683236
Li R, Guan Y, Yang R, Perrot V, Juan MA (2020) Comparison of controlled ovary stimulation protocols for in vitro fertilisation: a meta-analysis. Fertil Steril 114(3):e442–e444. https://doi.org/10.1016/j.fertnstert.2020.08.1282
Kadoura S, Alhalabi M, Nattouf AH (2022) Conventional GnRH antagonist protocols versus long GnRH agonist protocol in IVF/ICSI cycles of polycystic ovary syndrome women: a systematic review and meta-analysis. Sci Rep 12(1):4456. https://doi.org/10.1038/s41598-022-08400-z
Hu L, Du J, Lv H, Zhao J, Chen M, Wang Y, Wu F, Liu F, Chen X, Zhang J, Ma H, Jin G, Shen H, Chen L, Ling X, Hu Z (2018) Influencing factors of pregnancy loss and survival probability of clinical pregnancies conceived through assisted reproductive technology. Reprod Biol Endocrinol 16(1):74. https://doi.org/10.1186/s12958-018-0390-6
Kyrou D, Fatemi HM, Zepiridis L, Riva A, Papanikolaou EG, Tarlatzis BC, Devroey P (2011) Does cessation of progesterone supplementation during early pregnancy in patients treated with recFSH/GnRH antagonist affect ongoing pregnancy rates? A randomized controlled trial. Hum Reprod 26(5):1020–1024. https://doi.org/10.1093/humrep/der012
Pan SP, Chao KH, Huang CC, Wu MY, Chen MJ, Chang CH, Yang JH, Yang YS, Chen SU (2018) Early stop of progesterone supplementation after confirmation of pregnancy in IVF/ICSI fresh embryo transfer cycles of poor responders does not affect pregnancy outcome. PLoS ONE 13(8):e0201824. https://doi.org/10.1371/journal.pone.0201824
Acknowledgements
We thank Dr. Laura Woods (University of London) for her support throughout the project.
Funding
The authors declare that no funds, grants or other support was received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation and data collection were performed by TS, NZ, LL and SP. Data analysis was performed by TS. The first draft of the manuscript was written by TS and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval and consent
Access to patients’ files was approved by the Quebec Commission of access to information (ref: 1014586-S). Ethics approval was obtained by the University of Montreal Hospital Center Research Ethics Committee (CR-CHUM; ref: 15.387).
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shaulov, T., Zanré, N., Phillips, S. et al. The association between the type of progesterone supplementation and miscarriage risk in women who have had a positive pregnancy test following embryo transfer: a retrospective cohort study. Arch Gynecol Obstet 308, 569–577 (2023). https://doi.org/10.1007/s00404-023-07047-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-023-07047-z