Abstract
Purpose
The ongoing coronavirus disease 2019 (COVID-19) pandemic, caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has imposed a heavy burden on medical systems. In addition to the respiratory system, the virus also causes injuries to other organs and systems such as the gastroenteric system, kidneys, and reproductive system. Female reproductive health requires more attention in this context.
Methods
We have performed a thorough review of the relevant literature that addresses the impacts of SARS-CoV-2 infection and COVID-19 vaccination on the female reproductive system.
Results
Most evidence shows that SARS-CoV-2 does not infect the female reproductive system. However, the virus may indirectly influence sex hormone concentrations through inflammation associated with cytokine storms and nervous system damage. Menstrual disorders in women infected with SARS-CoV-2 may be caused by down-regulation of angiotensin-converting enzyme 2, abnormal hormone levels, medications, and stress. There is no significant difference in ovarian follicle quality and in vitro fertilization parameters between the pre- and post-COVID-19 vaccination groups. In addition, most symptoms due to side effects of vaccination could recover within a short period of time.
Conclusion
SARS-CoV-2 infection affects female reproductive system function through multiple mechanisms. It is recommended that women of childbearing age be vaccinated with COVID-19 vaccine.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Some female patients infected with SARS-CoV-2 showed abnormal changes in sex hormone concentrations and menstruation. Additionally, the impact of COVID-19 vaccination on menstruation is no different from the short-term and reversible side effects of other vaccination. |
Introduction
Coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) broke out in December 2019. The disease has overwhelmed the healthcare systems around the globe at an unprecedented level. As of December 9, 2022, it has resulted in 643,875,406 confirmed cases and 6,630,082 deaths [1]. The main clinical manifestations of SARS-CoV-2 infection range from asymptomatic through mild respiratory infection and influenza-like illness (mainly fever, cough, and fatigue), to severe disease with accompanying lung damage, multiple organ failure, and death [2, 3]. New insights highlight that other organs and systems can also be infected by the virus. Indeed, diarrhea, nausea, and vomiting are reported in a minor fraction of the cases, suggesting the involvement of the gastroenteric system [4]. Furthermore, proteinuria and acute renal tubular damage in patients infected with SARS-CoV-2 indicated kidney impairment [5, 6]. In particular, the investigations whether the reproductive system may be affected by the coronavirus are ongoing. Some obvious clinical symptoms like scrotal discomfort caused by SARS-CoV-2 had been reported [7]. Leydig cell reduction, orchitis, and sex hormone abnormalities have also been found [8,9,10]. As well known, more than 70% of healthcare and social care workers worldwide are women, and these populations are more susceptible to exposure to the virus. Thus, female fertility and reproductive health also require more attention in the COVID-19 era. Considering these elements, this review starts with whether SARS-CoV-2 can directly infect the female reproductive system, and focuses on the impact of virus infection on female sex hormones, ovarian reserve, and menstruation. Additionally, the article also addresses whether COVID-19 vaccination has any severe side effects on the female reproductive system.
SARS-CoV-2 in female reproductive system
It is well known that SARS-CoV-2 utilizes angiotensin-converting enzyme 2 (ACE2) as the primary receptor-binding site to invade target host cells [11]. The ACE2 is expressed in many female reproductive organs, such as ovaries, uterus, and vagina [12,13,14]. In addition, basigin/CD147 has already been identified as a potential additional host receptor for viral entry [15]. This highly glycosylated transmembrane protein can also be found in the ovaries, uterus, and placenta [16,17,18]. Luongo et al. confirmed the viral infection of human granulosa cells and cumulus cells in vitro using immunofluorescence for spike and nucleocapsid virus proteins [16]. Thus, it has been hypothesized that the female reproductive system may be a potential target of SARS-CoV-2. However, several studies did not detect the virus in the oocytes and cervical exfoliated cells of female patients with COVID-19 [19, 20]. In addition, the presence of the virus in somatic ovarian cells has not been reported to date.
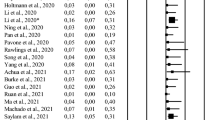
Many viruses have previously been isolated within vaginal fluid, such as hepatitis C virus and Zika virus, both of which are RNA viruses. Understanding whether the SARS-CoV-2 is present in vaginal fluid will provide useful information for determining the transmission route of SARS-CoV-2 and the ideal mode of delivery. Fortunately, several studies have failed to detect the presence of the virus in vaginal samples from pregnant and non-pregnant women of reproductive age with COVID-19 [21, 22]. Moreover, vaginal fluid samples from postmenopausal women with severe respiratory symptoms also tested negative for SARS-CoV-2 [23]. However, a study found a positive reverse transcriptase-polymerase chain reaction (RT-PCR) for the virus in the vaginal fluid of a postmenopausal woman among 51 pregnant and non-pregnant patients with mild-to-moderately severe COVID-19 [24]. Of note, Khoiwal et al. collected 15 vaginal and 12 cervical swabs from women with positive nasal PCR tests, and analyzed these samples for SARS-CoV-2 with two different techniques. In the first technique, they performed real-time RT-PCR treated as per the World Health Organization standard protocol. Subsequently, the second sample aliquot was processed for transcription-mediated amplification, and detection was performed by nucleic acid hybridization. All samples tested negative with RT-PCR. However, the virus was identified in the vaginal fluid of three participants with the transcription-mediated amplification system [25]. It is important to emphasize that the small sample size may be potential limitation of these studies. A larger cohort and longer duration of study is still needed to conclude whether SARS-CoV-2 infects the vagina. In addition, it is recommended to use different techniques to detect the virus in samples that were obtained from systems other than the respiratory system.
The effects of SARS-CoV-2 on hormones and ovarian reserve
Ovarian endocrine system seems not to be severely affected in most female patients with COVID-19. For example, Li et al. did not find statistically significant differences in the concentrations of all sex hormones between COVID-19 patients and the controls. Moreover, sub-group analysis of menstrual changes revealed no significant changes in sex hormone concentrations in either menstrual cycle changes, volume changes, or simultaneous cycle and volume changes [26]. However, it was undeniable that some patients had inappropriately high concentrations in their sex hormone, such as follicle-stimulating hormone (FSH), luteinizing hormone (LH), and prolactin (PRL), which may indicate ovarian suppression in these patients [26, 27]. It is well known that sex hormones have been shown to reduce inflammatory responses. Increased sex hormones in some COVID-19 patients may be a response to decrease inflammation associated with cytokine storms. Moreover, the nervous system could be destroyed by SARS-CoV-2 [28]. Thus, the significant increase in serum pituitary hormone LH and PRL level might partly result from the direct impact on brain tissue. In addition, women are prone to anxiety during the epidemic of COVID-19. 36 of the 75 (48%) patients reported a recent mental disorder, such as depression, anxiety, or insomnia, which could lead to PRL elevation and hypothalamic–pituitary–ovarian (HPO) axis dysfunction [27]. Given excessive immune or inflammatory responses and dysfunction of the HPO axis can contribute to abnormal ovarian function, some people speculated that female patients may have ovarian damage under COVID-19 [27].
Anti-Müllerian hormone (AMH) is one of the most accurate indicators for evaluating ovarian reserve and is not affected by exogenous sex hormones and menstruation [29, 30]. Most studies have demonstrated that SARS-CoV-2 infection did not seem to alter the serum AMH [26, 31, 32]. On the contrary, Ding et al. reported that the serum AMH concentrations of COVID-19 patients were considerably lower than serum AMH concentrations of healthy women [27]. Notably, this difference may be accounted for by the comparison of different groups, the small number of subjects, and the high incidence of severe COVID-19 disease in their study population (17/78). Moreover, low serum AMH concentrations were also linked to psychological stress and severity of anxiety [33]. Therefore, it can be hypothesized that SARS-CoV-2 infection may have little effect on ovarian reserve, and more clinical evidence and laboratory data are needed to support this suspicion.
The effects of SARS-CoV-2 on menstruation
The regular menstrual cycle ensures fertility and is an important marker of reproductive health. In turn, the irregularities in the menstrual cycle could pave the way for certain complications, such as low birth weight fetuses, preeclampsia, and an increased risk of metabolic disturbances [34, 35]. There have been many reports of menstrual disorders in women infected with SARS-CoV-2, including worsening premenstrual symptoms, prolonged menstrual cycle, and decreased volume [26, 36]. ACE2 not only plays a role in the pathogenesis of COVID-19 but also as an important component of the renin–angiotensin system. Angiotensin-2 (Ang II), the product of cleavage of angiotensin-1 by angiotensin-converting enzyme, acts as a vasoconstrictor and initiates menstruation through spiral artery vasoconstriction [37, 38]. Subsequently, it can be further cleaved by ACE2 to form peptide angiotensin-(1–7), which counteracts the activity of Ang II and has a vasodilatory effect [39]. However, SARS-CoV-2 invasion and cellular internalization lead to the down-regulation of membrane-bound ACE2 and increased serum ACE2, resulting in angiotensin-(1–7) depletion and unopposed Ang II activity [40]. This may be one of the reasons why SARS-CoV-2 infection leads to menstrual disorders. Moreover, as mentioned earlier, SARS-CoV-2 infection might lead to hormonal abnormalities in some patients. Abnormal hormonal patterns of FSH, LH, and estradiol can affect the length of the menstrual cycle. Specifically, short cycle is associated with earlier elevations in FSH and higher estradiol levels, while longer cycle is associated with higher LH and lower estradiol levels [41].
Compared with non-severe COVID-19 cases, patients with more severe cases had higher levels of irregular periods, higher levels of amenorrhea, and higher levels of menstrual pain [27, 42]. First, severely ill patients were more likely to be obese, or have metabolic syndrome, both of which are linked to menstrual irregularities [42, 43]. Second, as an important treatment for patients with severe illness, dexamethasone might also be a risk factor for menstrual changes [44]. It may affect menstrual cycle patterns and blood loss [45]. Likewise, some antipyretics and analgesics used to treat symptoms of COVID-19, such as aspirin, paracetamol, and other non-steroidal anti-inflammatories, can reduce menstrual blood loss through affecting prostaglandin synthesis and endometrial prostaglandin levels [46]. Moreover, hospitalization may be a stressful event for COVID-19 patients and thus affect the menstrual cycle. Compared to those with a moderate COVID-19 Perceived Stress Scale, respondents with a high Perceived Stress Scale had a prolongation of menstruation [47].
Fortunately, 84% returned to a normal menstrual volume, and 99% of patients returned to normal cycle within 1–2 months after discharge in a study [26]. Likewise, most respondents also returned to regular menstrual cycle within 6 months of recovering from COVID-19 in another study [48]. Thus, menstrual changes caused by COVID-19 were most likely transient and could recover within a short period of time.
COVID-19 vaccination and female reproductive health
Universal vaccination programs have been effective in managing several infectious diseases, such as mumps, rubella, polio, and measles [49, 50]. Thus, safe and effective vaccination remains one of the most important ways to fight COVID-19. To date, the development of a vaccine against COVID-19 has progressed globally. Multiple types of vaccines are being administered to recipients, including inactivated, viral vector-based, mRNA, and protein subunit vaccines [51].
Compared to non-vaccinated controls, women who had been vaccinated against COVID-19 did not exhibit differences in any of the surrogate parameters for ovarian follicle quality [52]. Moreover, in in vitro fertilization treatment, many parameters, such as the number of mature oocytes retrieved, fertilization rate, and the ratio of top-quality embryos per fertilized oocyte, also did not significantly differ between the pre- and post-mRNA SARS-CoV-2 vaccination groups [53]. However, vaccine hesitancy or refusal has been exacerbated by recent reports that COVID-19 vaccination can cause menstrual abnormalities. A study from the Middle East and North Africa region indicated that 66.3% of women experienced menstrual abnormalities after vaccination, including but not limited to longer menstruation duration and prolonged menstrual cycle [54]. In addition, the incidence of irregular menstruation after the second dose of COVID-19 vaccine appears to be slightly higher than the first dose, suggesting a potential additive effect [55]. As far as we known, vaccination triggers the necessary immune response. The immune activation may temporarily interfere with the HPO axis [56]. In addition, cyclic breakdown and restoration of the uterine endometrium are mediated by immune cells residing in the endometrium [57]. Activation of these immune cells could be responsible for heavier, irregular, and untimely bleeding [58]. Moreover, immune-mediated vaccine-induced thrombocytopenia is another plausible hypothesis for post-vaccination menstrual changes [59]. As mentioned above, elevated levels of stress and anxiety during the pandemic have also been linked to irregular menstrual cycle.
In fact, other vaccines also have some side effects. For example, Gong et al. have reported elevated FSH, irregular menstruation, and premature menopause associated with human papillomavirus vaccines [60]. Similarly, the symptom was also reported after hepatitis B vaccination and typhoid vaccination [61, 62]. Fortunately, menstrual irregularities that occurred after the first and second doses of the COVID-19 vaccine self-resolved in approximately half the cases within 2 months without associated clinical consequences [55]. Therefore, it can be assumed that menstrual changes are not different from other short-term and reversible side effects of vaccination.
Conclusion
Most studies have shown that SARS-CoV-2 did not infect the female reproductive system. However, the existing literature is divided on the presence of the virus in vaginal fluid. Some female patients infected with SARS-CoV-2 showed abnormal sex hormone concentrations, suggesting that ovarian suppression may have occurred. Increase in sex hormones may be a response to decrease inflammation associated with cytokine storms. In addition, nervous system damage might also be the cause of changes in sex hormone concentrations. Fortunately, the ovarian reserve does not seem to be affected by SARS-CoV-2 infection. Abnormal changes in menstruation can also be observed in some women with COVID-19, including worsening premenstrual symptoms, prolonged menstrual cycle, and decreased menstrual volume. First, this may be due to the down-regulation of ACE2 expression caused by the virus infection. Second, it appears to be caused by abnormal hormone levels. Moreover, medications and stress were also considered factors. Thankfully, most menstrual changes can be recovered in a short period of time.
COVID-19 vaccination remains an effective approach against SARS-CoV-2. Previous studies have demonstrated that vaccination has no effect on ovarian follicle and in vitro fertilization treatment. However, several recent reports of menstrual abnormalities caused by vaccinations have raised concerns about vaccinations. In fact, this is no different from the short-term and reversible side effects of other vaccination. Moreover, most symptoms disappear within 2 months. Therefore, we still recommend COVID-19 vaccination for women of reproductive age.
Data availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
References
World Health Organization (2022) WHO Coronavirus (COVID-19) Dashboard. https://covid19.who.int/. Accessed 11 Dec 2022
Xu T, Huang R, Zhu L, Wang J, Cheng J, Zhang B, Zhao H, Chen K, Shao H, Zhu C et al (2020) Epidemiological and clinical features of asymptomatic patients with SARS-CoV-2 infection. J Med Virol 92(10):1884–1889
Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, Liu L, Shan H, Lei CL, Hui DS et al (2020) Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 382(18):1708–1720
Ashktorab H, Pizuorno A, Oskroch G, Fierro NA, Sherif ZA, Brim H (2021) COVID-19 in Latin America: symptoms, morbidities, and gastrointestinal manifestations. Gastroenterology 160(3):938–940
Li Z, Wu M, Yao J, Guo J, Liao X, Song S, Li J, Duan G, Zhou Y, Wu X et al (2020) Caution on kidney dysfunctions of COVID-19 patients. MedRxiv https://doi.org/10.1101/2020.02.08.20021212
Diao B, Wang C, Wang R, Feng Z, Zhang J, Yang H, Tan Y, Wang H, Wang C, Liu L et al (2021) Human kidney is a target for novel severe acute respiratory syndrome coronavirus 2 infection. Nat Commun 12(1):1–9
Pan F, Xiao X, Guo J, Song Y, Li H, Patel DP, Spivak AM, Alukal JP, Zhang X, Xiong C et al (2020) No evidence of severe acute respiratory syndrome–coronavirus 2 in semen of males recovering from coronavirus disease 2019. Fertil Steril 113(6):1135–1139
Yang M, Chen S, Huang B, Zhong JM, Su H, Chen YJ, Cao Q, Ma L, He J, Li XF et al (2020) Pathological findings in the testes of COVID-19 patients: clinical implications. Eur Urol Focus 6(5):1124–1129
Bridwell RE, Merrill DR, Griffith SA, Wray J, Oliver JJ (2021) A coronavirus disease 2019 (COVID-19) patient with bilateral orchitis. Am J Emerg Med 42(260):e3-260.e5
Ma L, Xie W, Li D, Shi L, Mao Y, Xiong Y, Zhang Y, Zhang M (2020) Effect of SARS-CoV-2 infection upon male gonadal function: a single center-based study. MedRxiv
Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH, Nitsche A et al (2020) SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 181(2):271-280.e8
Reis FM, Bouissou DR, Pereira VM, Camargos AF, dos Reis AM, Santos RA (2011) Angiotensin-(1–7), its receptor Mas, and the angiotensin-converting enzyme type 2 are expressed in the human ovary. Fertil Steril 95(1):176–181
Vaz-Silva J, Carneiro M, Ferreira M, Pinheiro S, Silva D, Silva A, Witz C, Reis A, Santos R, Reis F (2009) The vasoactive peptide angiotensin-(1–7), its receptor Mas and the angiotensin-converting enzyme type 2 are expressed in the human endometrium. Reprod Sci 16(3):247–256
The Human Protein Atlas (2022) Tissue expression of ACE2-Staining in vagina. https://www.proteinatlas.org/ENSG00000130234-ACE2/tissue/vagina. Accessed 11 Dec 2022
Wang K, Chen W, Zhou YS, Lian JQ, Zhang Z, Du P, Gong L, Zhang Y, Cui HY, Geng JJ et al (2020) SARS-CoV-2 invades host cells via a novel route: CD147-spike protein. biorxiv
Luongo FP, Dragoni F, Boccuto A, Paccagnini E, Gentile M, Canosi T, Morgante G, Luddi A, Zazzi M, Vicenti I et al (2022) SARS-CoV-2 infection of human ovarian cells: a potential negative impact on female fertility. Cells 11(9):1431
Essahib W, Verheyen G, Tournaye H, Van de Velde H (2020) SARS-CoV-2 host receptors ACE2 and CD147 (BSG) are present on human oocytes and blastocysts. J Assist Reprod Genet 37(11):2657–2660
Pique-Regi R, Romero R, Tarca AL, Luca F, Xu Y, Alazizi A, Leng Y, Hsu CD, Gomez-Lopez N (2020) Does the human placenta express the canonical cell entry mediators for SARS-CoV-2? Elife 9:e58716
Barragan M, Guillén J, Martin-Palomino N, Rodriguez A, Vassena R (2021) Undetectable viral RNA in oocytes from SARS-CoV-2 positive women. Hum Reprod 36(2):390–394
Cui P, Chen Z, Wang T, Dai J, Zhang J, Ding T, Jiang J, Liu J, Zhang C, Shan W et al (2020) Severe acute respiratory syndrome coronavirus 2 detection in the female lower genital tract. Am J Obstet Gynecol 223(1):131–134
Aslan MM, Uslu Yuvacı H, Köse O, Toptan H, Akdemir N, Köroğlu M, Cevrioğlu AS, Özden S (2022) SARS-CoV-2 is not present in the vaginal fluid of pregnant women with COVID-19. J Matern Fetal Neonatal Med 35(15):2876–2878
Takmaz O, Kaya E, Erdi B, Unsal G, Sharifli P, Agaoglu NB, Ozbasli E, Gencer S, Gungor M (2021) Severe acute respiratory syndrome coronavirus (SARS-CoV-2) is not detected in the vagina: a prospective study. PLoS ONE 16(9):e0253072
Qiu L, Liu X, Xiao M, Xie J, Cao W, Liu Z, Morse A, Xie Y, Li T, Zhu L (2020) SARS-CoV-2 is not detectable in the vaginal fluid of women with severe COVID-19 infection. Clin Infect Dis 71(15):813–817
Barber E, Kovo M, Leytes S, Sagiv R, Weiner E, Schwartz O, Mashavi M, Holtzman K, Bar J, Engel A et al (2021) Evaluation of SARS-CoV-2 in the vaginal secretions of women with COVID-19: a prospective study. J Clin Med 10(12):2735
Khoiwal K, Kalita D, Shankar R, Kumari R, Dhundi D, Bahadur A, Panda PK, Chaturvedi J (2021) Identification of SARS-CoV-2 in the vaginal fluid and cervical exfoliated cells of women with active COVID-19 infection: a pilot study. Int J Gynecol Obstet 153(3):551–553
Li K, Chen G, Hou H, Liao Q, Chen J, Bai H, Lee S, Wang C, Li H, Cheng L et al (2021) Analysis of sex hormones and menstruation in COVID-19 women of child-bearing age. Reprod Biomed Online 42(1):260–267
Ding T, Wang T, Zhang J, Cui P, Chen Z, Zhou S, Yuan S, Ma W, Zhang M, Rong Y et al (2021) Analysis of ovarian injury associated with COVID-19 disease in reproductive-aged women in Wuhan, China: an observational study. Front Med. https://doi.org/10.3389/fmed.2021.635255
Mao L, Jin H, Wang M, Hu Y, Chen S, He Q, Chang J, Hong C, Zhou Y, Wang D et al (2020) Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol 77(6):683–690
Somunkiran A, Yavuz T, Yucel O, Ozdemir I (2007) Anti-Müllerian hormone levels during hormonal contraception in women with polycystic ovary syndrome. Eur J Obstet Gynecol Reprod Biol 134(2):196–201
Hehenkamp WJ, Looman CW, Themmen AP, de Jong FH, Te Velde E, Broekmans FJ (2006) Anti-Mullerian hormone levels in the spontaneous menstrual cycle do not show substantial fluctuation. J Clin Endocrinol Metab 91(10):4057–4063
Wang M, Yang Q, Ren X, Hu J, Li Z, Long R, Xi Q, Zhu L, Jin L (2021) Investigating the impact of asymptomatic or mild SARS-CoV-2 infection on female fertility and in vitro fertilization outcomes: a retrospective cohort study. EClinicalMedicine 38:101013
Kolanska K, Hours A, Jonquière L, d’Argent EM, Dabi Y, Dupont C, Touboul C, Antoine J-M, Chabbert-Buffet N, Daraï E (2021) Mild COVID-19 infection does not alter the ovarian reserve in women treated with ART. Reprod Biomed Online 43(6):1117–1121
Yeğin GF, Desdicioğlu R, Seçen Eİ, Aydın S, Bal C, Göka E, Keskin HL (2022) Low anti-Mullerian hormone levels are associated with the severity of anxiety experienced by healthcare professionals during the COVID-19 pandemic. Reprod Sci 29(2):627–632
Bonnesen B, Oddgeirsdóttir HL, Naver KV, Jørgensen FS, Nilas L (2016) Women with minor menstrual irregularities have increased risk of preeclampsia and low birthweight in spontaneous pregnancies. Acta Obstet Gynecol Scand 95(1):88–92
Rostami Dovom M, Ramezani Tehrani F, Djalalinia S, Cheraghi L, Behboudi Gandavani S, Azizi F (2016) Menstrual cycle irregularity and metabolic disorders: a population-based prospective study. PLoS ONE 11(12):e0168402
Khan SM, Shilen A, Heslin KM, Ishimwe P, Allen AM, Jacobs ET, Farland LV (2022) SARS-CoV-2 infection and subsequent changes in the menstrual cycle among participants in the Arizona CoVHORT study. Am J Obstet Gynecol 226(2):270–273
Donoghue M, Hsieh F, Baronas E, Godbout K, Gosselin M, Stagliano N, Donovan M, Woolf B, Robison K, Jeyaseelan R et al (2000) A novel angiotensin-converting enzyme–related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1–9. Circ Res 87(5):e1–e9
Ahmed A, Li X, Shams M, Gregory J, Rollason T, Barnes N, Newton J (1995) Localization of the angiotensin II and its receptor subtype expression in human endometrium and identification of a novel high-affinity angiotensin II binding site. J Clin Investig 96(2):848–857
Oliveira MA, Fortes ZB, Santos RA, Kosla MC, De Carvalho MHC (1999) Synergistic effect of angiotensin-(1–7) on bradykinin arteriolar dilation in vivo. Peptides 20(10):1195–1201
Abassi Z, Higazi AAR, Kinaneh S, Armaly Z, Skorecki K, Heyman SN (2020) ACE2, COVID-19 infection, inflammation, and coagulopathy: missing pieces in the puzzle. Front Physiol 11:574753
Mumford SL, Steiner AZ, Pollack AZ, Perkins NJ, Filiberto AC, Albert PS, Mattison DR, Wactawski-Wende J, Schisterman EF (2012) The utility of menstrual cycle length as an indicator of cumulative hormonal exposure. J Clin Endocrinol Metab 97(10):E1871–E1879
Lebar V, Lagana AS, Chiantera V, Kunic T, Lukanovic D (2022) The Effect of COVID-19 on the menstrual cycle: a systematic review. J Clin Med. https://doi.org/10.3390/jcm11133800
Memtsoudis SG, Ivascu NS, Pryor KO, Goldstein PA (2020) Obesity as a risk factor for poor outcome in COVID-19-induced lung injury: the potential role of undiagnosed obstructive sleep apnoea. Br J Anaesth 125(2):e262–e263
National Institutes of Health (2022) Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. https://www.covid19treatmentguidelines.nih.gov/. Accessed 11 Dec 2022
Warner P, Whitaker LHR, Parker RA, Weir CJ, Douglas A, Hansen CH, Madhra M, Hillier SG, Saunders PTK, Iredale JP et al (2021) Low dose dexamethasone as treatment for women with heavy menstrual bleeding: a response-adaptive randomised placebo-controlled dose-finding parallel group trial (DexFEM). EBioMedicine 69:103434
Rodriguez MB, Lethaby A, Farquhar C (2019) Non-steroidal anti-inflammatory drugs for heavy menstrual bleeding. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD000400.pub4
Ozimek N, Velez K, Anvari H, Butler L, Goldman KN, Woitowich NC (2022) Impact of stress on menstrual cyclicity during the coronavirus disease 2019 pandemic: a survey study. J Womens Health 31(1):84–90
Saxena RK, Basavaraju M, Meghana T (2022) Effect of COVID-19 infection on menstruation: a retrospective study. J South Asian Fed Obstet Gynaecol 14(2):161–165
Centers for Disease Control and Prevention (1998) Measles, mumps, and rubella -- vaccine use and strategies for elimination of measles, rubella, and congenital rubella syndrome and control of mumps: recommendations of the Advisory Committee on Immunization Practices (ACIP). https://wonder.cdc.gov/wonder/Prevguid/m0053391/m0053391.asp. Accessed 11 Dec 2022
Greenwood B (2014) The contribution of vaccination to global health: past, present and future. Philos Trans Royal Soc B: Biol Sci 369(1645):20130433
World Health Organization (2022) COVID-19 vaccine tracker and landscape. https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines. Accessed 11 Dec 2022
Bentov Y, Beharier O, Moav-Zafrir A, Kabessa M, Godin M, Greenfield CS, Ketzinel-Gilad M, Ash Broder E, Holzer HE, Wolf D et al (2021) Ovarian follicular function is not altered by SARS–CoV-2 infection or BNT162b2 mRNA COVID-19 vaccination. Hum Reprod 36(9):2506–2513
Orvieto R, Noach-Hirsh M, Segev-Zahav A, Haas J, Nahum R, Aizer A (2021) Does mRNA SARS-CoV-2 vaccine influence patients’ performance during IVF-ET cycle? Reprod Biol Endocrinol 19(1):1–4
Muhaidat N, Alshrouf MA, Azzam MI, Karam AM, Al-Nazer MW, Al-Ani A (2022) Menstrual symptoms after COVID-19 vaccine: a cross-sectional investigation in the MENA region. Int J Women’s Health 14:395
Laganà AS, Veronesi G, Ghezzi F, Ferrario MM, Cromi A, Bizzarri M, Garzon S, Cosentino M (2022) Evaluation of menstrual irregularities after COVID-19 vaccination: results of the MECOVAC survey. Open Med 17(1):475–484
Hassan YA, Ali MD, Al-Eid RR, Al-Ghuraya FA, Alqasimi ZE, Ahmad A, Eltrafi Z, Ghosn SA (2022) A retrospective evaluation of side-effects associated with the booster dose of Pfizer-BioNTech/BNT162b2 COVID-19 vaccine among females in Eastern Province, Saudi Arabia. Vaccine 40(49):7087–7096
Monin L, Whettlock EM, Male V (2020) Immune responses in the human female reproductive tract. Immunology 160(2):106–115
Nazir M, Asghar S, Rathore MA, Shahzad A, Shahid A, Khan AA, Malik A, Fakhar T, Kausar H, Malik J (2022) Menstrual abnormalities after COVID-19 vaccines: a systematic review. Vacunas 23:S77–S87
Perricone C, Ceccarelli F, Nesher G, Borella E, Odeh Q, Conti F, Shoenfeld Y, Valesini G (2014) Immune thrombocytopenic purpura (ITP) associated with vaccinations: a review of reported cases. Immunol Res 60(2):226–235
Gong L, Ji HH, Tang XW, Pan LY, Chen X, Jia YT (2020) Human papillomavirus vaccine-associated premature ovarian insufficiency and related adverse events: data mining of vaccine adverse event reporting system. Sci Rep 10(1):1–8
Shingu T, Uchida T, Nishi M, Hayashida K, Kashiwagi S, Hayashi J, Kaji M (1983) Menstrual abnormalities after hepatitis B vaccine. Kurume Med J 29(3):123–125
Lamb AR (1913) Experiences with prophylactic typhoid vaccination: its effect on menstruation. Arch Intern Med 12(5):565–577
Funding
This work was supported by the Natural Science Foundation of Shandong Province under Grant No. ZR2020QC100.
Author information
Authors and Affiliations
Contributions
FC, SL, and HL initiated the project. FC and SL wrote the initial draft of the manuscript with input from HL. DL searched the literature. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Not relevant for this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, S., Liu, H., Li, D. et al. Female reproductive health during the COVID-19 pandemic: latest evidence and understanding. Arch Gynecol Obstet 308, 1691–1696 (2023). https://doi.org/10.1007/s00404-023-06976-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-023-06976-z