Abstract
Purpose
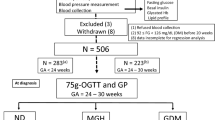
This study aimed to identify the optimal cutoff values of each component of metabolic syndrome (MetS) in the first trimester of pregnancy for predicting adverse pregnancy outcomes.
Methods
A total of 1076 pregnant women in the first trimester of gestation were recruited in this prospective longitudinal cohort study. Specifically, 993 pregnant women at 11–13 weeks of gestation who were followed up until the end of pregnancy were included in the final analysis. The cutoff values of each component of MetS in the occurrence of adverse pregnancy outcomes including gestational diabetes (GDM), gestational hypertensive disorders, and preterm birth were obtained via receiver operating characteristic (ROC) curve analysis using the Youden’s index.
Results
Among the 993 pregnant women studied, the significant associations between the first trimester MetS components and adverse pregnancy outcomes were as follows: triglyceride (TG) and body mass index (BMI) with preterm birth; mean arterial pressure (MAP), TG, and high-density lipoprotein cholesterol (HDL-C) with gestational hypertensive disorders; BMI, fasting plasma glucose (FPG), and TG with GDM (all p values < 0.05). The cutoff point values for the above-mentioned MetS components were: TG > 138 mg/dl and BMI < 21 kg/m2 for the occurrence of preterm birth; TG > 148 mg/dL, MAP > 84, and HDL-C < 84 mg/dl for gestational hypertensive disorders; BMI > 25 kg/m2, FPG > 84 mg/dl, and TG > 161 mg/dl for GDM.
Conclusion
The study findings imply the importance of early management of metabolic syndrome in pregnancy to improve maternal–fetal outcomes.

Similar content being viewed by others
Data availability
All relevant data in this study is available through an emailed request to the corresponding author, and her getting permission from Tarbiat Modares University, Deputy of Research.
Abbreviations
- MetS:
-
Metabolic syndrome
- TG:
-
Triglyceride
- BMI:
-
Body mass index
- MAP:
-
Mean arterial pressure
- HDL-C:
-
High-density lipoprotein cholesterol
- FPG:
-
Fasting plasma glucose
- GDM:
-
Gestational diabetes
References
Alberti KG, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA et al (2009) Harmonizing the metabolic syndrome: a joint interim statement of the international diabetes federation task force on epidemiology and prevention; national heart, lung, and blood institute; American heart association; world heart federation; international atherosclerosis society; and international association for the study of obesity. Circulation 120(16):1640–1645
Kaur J (2014) A comprehensive review on metabolic syndrome. Cardiol Res Pract 2014:943162
McCracken E, Monaghan M, Sreenivasan S (2018) Pathophysiology of the metabolic syndrome. Clin Dermatol 36(1):14–20
Saklayen MG (2018) The global epidemic of the metabolic syndrome. Curr Hypertens Rep 20(2):12
Maw SS (2021) Prevalence of metabolic syndrome, its risk factors and associated lifestyles in Myanmar adult people: a community based cross-sectional study. Metabolism Open 12:100135
Grieger JA, Bianco-Miotto T, Grzeskowiak LE, Leemaqz SY, Poston L, McCowan LM et al (2018) Metabolic syndrome in pregnancy and risk for adverse pregnancy outcomes: a prospective cohort of nulliparous women. PLoS Med 15(12):e1002710
Ferranti EP, Jones EJ, Hernandez TL (2016) Pregnancy reveals evolving risk for cardiometabolic disease in women. J Obstet Gynecol Neonatal Nurs 45(3):413–425
Barry DR, Utzschneider KM, Tong J, Gaba K, Leotta DF, Brunzell JD et al (2015) Intraabdominal fat, insulin sensitivity, and cardiovascular risk factors in postpartum women with a history of preeclampsia. Am J Obstetrics Gynecol 213(1):104
Moayeri M, Heida KY, Franx A, Spiering W, de Laat MWM, Oudijk MA (2017) Maternal lipid profile and the relation with spontaneous preterm delivery: a systematic review. Arch Gynecol Obstet 295(2):313–323
Varner MW, Rice MM, Landon MB, Casey BM, Reddy UM, Wapner RJ et al (2017) Pregnancies after the diagnosis of mild gestational diabetes mellitus and risk of cardiometabolic disorders. Obstet Gynecol 129(2):273–280
Grieger JA, Bianco-Miotto T, Grzeskowiak LE, Leemaqz SY, Poston L, McCowan LM et al (2018) Metabolic syndrome in pregnancy and risk for adverse pregnancy outcomes: a prospective cohort of nulliparous women. PLoS Med 15(12):e1002710
Stekkinger E, Scholten R, Van Der Vlugt M, Van Dijk A, Janssen M, Spaanderman M (2013) Metabolic syndrome and the risk for recurrent pre-eclampsia: a retrospective cohort study. BJOG 120(8):979–986
Chalas E (2020) The American College of Obstetricians and gynecologists in 2020: a clear vision for the future. Obstet Gynecol 135(6):1251–1254
Henry JB, Miller MC, Kelly KC, Champney D (2002) Mean arterial pressure (MAP): an alternative and preferable measurement to systolic blood pressure (SBP) in patients for hypotension detection during hemapheresis. J Clin Apheresis 17(2):55–64
Phipps E, Prasanna D, Brima W, Jim B (2016) Preeclampsia: updates in pathogenesis, definitions, and guidelines. Clin J Am Soc Nephrol 11(6):1102–1113
Association AD (2020) Standards of Medical Care in Diabetes—2020 abridged for primary care providers. Clin Diabetes 38(1):10
Jin W-Y, Lin S-L, Hou R-L, Chen X-Y, Han T, Jin Y et al (2016) Associations between maternal lipid profile and pregnancy complications and perinatal outcomes: a population-based study from China. BMC Pregnancy Childbirth 16(1):1–9
Hajar Sharami S, Abbasi Ranjbar Z, Alizadeh F, Kazemnejad E (2019) The relationship of hyperlipidemia with maternal and neonatal outcomes in pregnancy: a cross-sectional study. Int J Reprod Biomed 17(10):739–748
Wiznitzer A, Mayer A, Novack V, Sheiner E, Gilutz H, Malhotra A et al (2009) Association of lipid levels during gestation with preeclampsia and gestational diabetes mellitus: a population-based study. Am J Obstetrics Gynecol 201(5):482.e1-e4828
Lewandowska M, Sajdak S, Więckowska B, Manevska N, Lubiński J (2020) The influence of maternal BMI on adverse pregnancy outcomes in older women. Nutrients 12(9):2838
Wu DD, Gao L, Huang O, Ullah K, Guo MX, Liu Y et al (2020) Increased adverse pregnancy outcomes associated with stage 1 hypertension in a low-risk cohort: evidence from 47 874 cases. Hypertension 75(3):772–780
Alleman BW, Smith AR, Byers HM, Bedell B, Ryckman KK, Murray JC et al (2013) A proposed method to predict preterm birth using clinical data, standard maternal serum screening, and cholesterol. Am J Obstetrics Gynecol 208(6):472.e1-e11
Lei Q, Niu J, Lv L, Duan D, Wen J, Lin X et al (2016) Clustering of metabolic risk factors and adverse pregnancy outcomes: a prospective cohort study. Diabetes Metab Res Rev 32(8):835–842
Maymunah A-O, Kehinde O, Abidoye G, Oluwatosin A (2014) Hypercholesterolaemia in pregnancy as a predictor of adverse pregnancy outcome. Afr Health Sci 14(4):967–973
Catalano PM (2014) Trying to understand gestational diabetes. Diabet Med 31(3):273–281
Lain KY, Catalano PM (2007) Metabolic changes in pregnancy. Clin Obstet Gynecol 50(4):938–948
Basaran A (2009) Pregnancy-induced hyperlipoproteinemia: review of the literature. Reprod Sci 16(5):431–437
Spracklen CN, Smith CJ, Saftlas AF, Robinson JG, Ryckman KK (2014) Maternal hyperlipidemia and the risk of preeclampsia: a meta-analysis. Am J Epidemiol 180(4):346–358
Jiang S, Jiang J, Xu H, Wang S, Liu Z, Li M et al (2017) Maternal dyslipidemia during pregnancy may increase the risk of preterm birth: a meta-analysis. Taiwan J Obstet Gynecol 56(1):9–15
Ryckman K, Spracklen C, Smith C, Robinson J, Saftlas A (2015) Maternal lipid levels during pregnancy and gestational diabetes: a systematic review and meta-analysis. BJOG 122(5):643–651
Kitajima M, Oka S, Yasuhi I, Fukuda M, Rii Y, Ishimaru T (2001) Maternal serum triglyceride at 24–32 weeks’ gestation and newborn weight in nondiabetic women with positive diabetic screens. Obstet Gynecol 97(5 Pt 1):776–780
Son GH, Kwon JY, Kim YH, Park YW (2010) Maternal serum triglycerides as predictive factors for large-for-gestational age newborns in women with gestational diabetes mellitus. Acta Obstet Gynecol Scand 89(5):700–704
Hashemipour S, Haji Seidjavadi E, Maleki F, Esmailzadehha N, Movahed F, Yazdi Z (2018) Level of maternal triglycerides is a predictor of fetal macrosomia in non-obese pregnant women with gestational diabetes mellitus. Pediatr Neonatol 59(6):567–572
Vrijkotte TG, Krukziener N, Hutten BA, Vollebregt KC, van Eijsden M, Twickler MB (2012) Maternal lipid profile during early pregnancy and pregnancy complications and outcomes: the ABCD study. J Clin Endocrinol Metab 97(11):3917–3925
Kandimalla BH, Sirjusingh A, Nayak BS, Maiya SS (2011) Early antenatal serum lipid levels and the risk of pre-eclampsia in Trinidad and Tobago. Arch Physiol Biochem 117(4):215–221
Chatzi L, Plana E, Daraki V, Karakosta P, Alegkakis D, Tsatsanis C et al (2009) Metabolic syndrome in early pregnancy and risk of preterm birth. Am J Epidemiol 170(7):829–836
Xue R-h, Wu D-d, Zhou C-l, Chen L, Li J, Li Z-z et al (2021) Association of high maternal triglyceride levels early and late in pregnancy with adverse outcomes: a retrospective cohort study. J Clin Lipidol 15(1):162–172
Ren Z, Zhe D, Li Z, Sun X-P, Yang K, Lin L (2020) Study on the correlation and predictive value of serum pregnancy-associated plasma protein A, triglyceride and serum 25-hydroxyvitamin D levels with gestational diabetes mellitus. World J Clin Cases 8(5):864–873
Keshavarz P, Gh BFNM, Mirhafez SR, Nematy M, Azimi-Nezhad M, Afin SA et al (2017) Alterations in lipid profile, zinc and copper levels and superoxide dismutase activities in normal pregnancy and preeclampsia. Am J Med Sci 353(6):552–558
León-Reyes G, Espino YSS, Medina-Navarro R, Guzmán-Grenfell AM, Medina-Urrutia AX, Fuentes-García S et al (2018) Oxidative modifications of foetal LDL-c and HDL-c lipoproteins in preeclampsia. Lipids Health Dis 17(1):110
Li J, Lu J, Wang M, Hu W, Jin N, Li X et al (2021) Predictive value of second-trimester maternal lipid profiling in early-onset pre-eclampsia: a prospective cohort study and nomogram. Front Med (Lausanne) 8:688312
Metzger BE, Lowe LP, Dyer AR, Trimble ER, Chaovarindr U, Coustan DR et al (2008) Hyperglycemia and adverse pregnancy outcomes. N Engl J Med 358(19):1991–2002
Riskin-Mashiah S, Damti A, Younes G, Auslender R (2010) First trimester fasting hyperglycemia as a predictor for the development of gestational diabetes mellitus. Eur J Obstet Gynecol Reprod Biol 152(2):163–167
Tong JN, Wu LL, Chen YX, Guan XN, Tian FY, Zhang HF et al (2022) Fasting plasma glucose in the first trimester is related to gestational diabetes mellitus and adverse pregnancy outcomes. Endocrine 75(1):70–81
Sesmilo G, Prats P, Garcia S, Rodríguez I, Rodríguez-Melcón A, Berges I et al (2020) First-trimester fasting glycemia as a predictor of gestational diabetes (GDM) and adverse pregnancy outcomes. Acta Diabetol 57(6):697–703
Raets L, Beunen K, Benhalima K (2021) Screening for gestational diabetes mellitus in early pregnancy: what is the evidence? J Clin Med 10(6):1257
Agarwal MM, Dhatt GS, Shah SM (2010) Gestational diabetes mellitus: simplifying the international association of diabetes and pregnancy diagnostic algorithm using fasting plasma glucose. Diabetes Care 33(9):2018–2020
Zhu WW, Fan L, Yang HX, Kong LY, Su SP, Wang ZL et al (2013) Fasting plasma glucose at 24–28 weeks to screen for gestational diabetes mellitus: new evidence from China. Diabetes Care 36(7):2038–2040
Kouhkan A, Khamseh ME, Moini A, Pirjani R, Arabipoor A, Zolfaghari Z et al (2019) Diagnostic accuracy of body mass index and fasting glucose for the prediction of gestational diabetes mellitus after assisted reproductive technology. Int J Fertility Sterility 13(1):32
Liu L, Ma Y, Wang N, Lin W, Liu Y, Wen D (2019) Maternal body mass index and risk of neonatal adverse outcomes in China: a systematic review and meta-analysis. BMC Pregnancy Childbirth 19(1):105
Chu SY, Kim SY, Schmid CH, Dietz PM, Callaghan WM, Lau J et al (2007) Maternal obesity and risk of cesarean delivery: a meta-analysis. Obes Rev 8(5):385–394
Torloni MR, Betrán AP, Horta BL, Nakamura MU, Atallah AN, Moron AF et al (2009) Prepregnancy BMI and the risk of gestational diabetes: a systematic review of the literature with meta-analysis. Obes Rev 10(2):194–203
Chen YT, Zhang T, Chen C, Xia YY, Han TL, Chen XY et al (2021) Associations of early pregnancy BMI with adverse pregnancy outcomes and infant neurocognitive development. Sci Rep 11(1):3793
Bowers K, Tobias DK, Yeung E, Hu FB, Zhang C (2012) A prospective study of prepregnancy dietary fat intake and risk of gestational diabetes. Am J Clin Nutr 95(2):446–453
Han Z, Mulla S, Beyene J, Liao G, McDonald SD (2011) Maternal underweight and the risk of preterm birth and low birth weight: a systematic review and meta-analyses. Int J Epidemiol 40(1):65–101
Catalano PM, Shankar K (2017) Obesity and pregnancy: mechanisms of short term and long term adverse consequences for mother and child. BMJ 356:j1
Athukorala C, Rumbold AR, Willson KJ, Crowther CA (2010) The risk of adverse pregnancy outcomes in women who are overweight or obese. BMC Pregnancy Childbirth 10:56
Nishikawa E, Oakley L, Seed PT, Doyle P, Oteng-Ntim E (2017) Maternal BMI and diabetes in pregnancy: investigating variations between ethnic groups using routine maternity data from London, UK. PLoS ONE 12(6):e0179332
Jensen DM, Damm P, Sørensen B, Mølsted-Pedersen L, Westergaard JG, Ovesen P et al (2003) Pregnancy outcome and prepregnancy body mass index in 2459 glucose-tolerant Danish women. Am J Obstet Gynecol 189(1):239–244
Rosenberg TJ, Garbers S, Chavkin W, Chiasson MA (2003) Prepregnancy weight and adverse perinatal outcomes in an ethnically diverse population. Obstet Gynecol 102(5 Pt 1):1022–1027
Sebire NJ, Jolly M, Harris JP, Wadsworth J, Joffe M, Beard RW et al (2001) Maternal obesity and pregnancy outcome: a study of 287,213 pregnancies in London. Int J Obes Relat Metab Disord 25(8):1175–1182
Hendler I, Goldenberg RL, Mercer BM, Iams JD, Meis PJ, Moawad AH et al (2005) The Preterm Prediction Study: association between maternal body mass index and spontaneous and indicated preterm birth. Am J Obstet Gynecol 192(3):882–886
Sharifzadeh F, Kashanian M, Jouhari S, Sheikhansari N (2015) Relationship between pre-pregnancy maternal BMI with spontaneous preterm delivery and birth weight. J Obstet Gynaecol 35(4):354–357
Liu B, Xu G, Sun Y, Du Y, Gao R, Snetselaar LG et al (2019) Association between maternal pre-pregnancy obesity and preterm birth according to maternal age and race or ethnicity: a population-based study. Lancet Diabetes Endocrinol 7(9):707–714
McDonald SD, Han Z, Mulla S, Beyene J, Knowledge Synthesis G (2010) Overweight and obesity in mothers and risk of preterm birth and low birth weight infants: systematic review and meta-analyses. BMJ 341:3428
Ehrenberg HM, Dierker L, Milluzzi C, Mercer BM (2003) Low maternal weight, failure to thrive in pregnancy, and adverse pregnancy outcomes. Am J Obstet Gynecol 189(6):1726–1730
Mayrink J, Souza RT, Feitosa FE, Rocha Filho EA, Leite DF, Vettorazzi J et al (2019) Mean arterial blood pressure: potential predictive tool for preeclampsia in a cohort of healthy nulliparous pregnant women. BMC Pregnancy Childbirth 19(1):1–8
Gasse C, Boutin A, Cote M, Chaillet N, Bujold E, Demers S (2018) First-trimester mean arterial blood pressure and the risk of preeclampsia: the Great Obstetrical Syndromes (GOS) study. Pregnancy hypertension 12:178–182
Zhu J, Zhang J, Syaza Razali N, Chern B, Tan KH (2021) Mean arterial pressure for predicting preeclampsia in Asian women: a longitudinal cohort study. BMJ Open 11(8):e046161
Cnossen JS, Vollebregt KC, de Vrieze N, ter Riet G, Mol BW, Franx A et al (2008) Accuracy of mean arterial pressure and blood pressure measurements in predicting pre-eclampsia: systematic review and meta-analysis. BMJ 336(7653):1117–1120
Nevalainen J, Korpimaki T, Kouru H, Sairanen M, Ryynanen M (2017) Performance of first trimester biochemical markers and mean arterial pressure in prediction of early-onset pre-eclampsia. Metabolism 75:6–15
Poon LC, Zymeri NA, Zamprakou A, Syngelaki A, Nicolaides KH (2012) Protocol for measurement of mean arterial pressure at 11–13 weeks’ gestation. Fetal Diagn Ther 31(1):42–48
Acknowledgements
This study was performed as a thesis project for obtaining Ph.D. degree in reproductive health, and was funded by the Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran.
Funding
This work was funded by Tarbiat Modares University.
Author information
Authors and Affiliations
Contributions
M-B: conceptualization, methodology, supervision, writing—reviewing and editing. MA: data collection, writing—original draft, investigation, resources. B-G: methodology, writing original draft, editing. RT: visualization, project administration, reviewing and editing manuscript. RF: software, validation, formal analysis. All authors approved the final draft of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest to disclose by the authors.
Ethical approval
The study proposal was approved by the Medical ethics committee of Tarbiat Modares University, Tehran, Iran. (ETHICS ID: IR.MODARES-REC.1397.007).
Informed consent
A written, informed consent was obtained from all participants after explaining about the purpose of the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Asltoghiri, M., Moghaddam-Banaem, L., Behboudi-Gandevani, S. et al. Prediction of adverse pregnancy outcomes by first-trimester components of metabolic syndrome: a prospective longitudinal study. Arch Gynecol Obstet 307, 1613–1623 (2023). https://doi.org/10.1007/s00404-023-06967-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-023-06967-0