Abstract
Purpose
To search for maternal, labor-related and fetal variables associated with low Apgar in the fifth minute in term pregnancy.
Methods
A retrospective case–control study with term births was conducted in a public teaching hospital from 2013 to 2020. Cases were term births with Apgar score less than 7 in the fifth minute, and controls, the next one or two births following a case, with Apgar of 7 or more. Non-cephalic presentations, multiples and malformations were excluded. We accessed 100 cases and 190 controls. We considered significant values of p < 0.05.
Results
Were accessed 27 variables which could be risk factors, from which 12 were associated with the outcome. We found a protective effect of prelabor cesarean for the outcome, odds ratio (OR) 0.38, p = 0.013. Consequently, we conducted two sets of analyses: in the whole group and in the group of laboring women. The values of OR were in general greater in the group of laboring women, compared with the whole group. We conducted multivariate analysis within the group of women in labor. The variables which fitted best in the model were nulliparity, male sex of the fetus, less than six prenatal visits and abnormal cardiotocography; all remained significant. An association of rupture of membranes more than 360 min with the outcome, even after controlling fpr duration of labor, was found; adjusted OR 2.45, p = 0.023.
Conclusion
Twelve variables were associated with the outcome. Prelabor cesarean had a protective effect. The time of ruptured membranes was associated with low Apgar.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Prelabor cesarean is protective against low Apgar in term. Most of cases of low Apgar in term come from pregnancies without maternal diseases. Time of ruptured membranes is associated with low Apgar in term. |
Introduction
The APGAR score was created by the anesthesiologist Virginia Apgar in 1953 to evaluate the need for neonatal resuscitation [1]. Since then, its utilization has been widened and, currently, the APGAR score is the standard method for evaluating the condition of a neonate in most settings that offer care for birth worldwide [2, 3]. The score has a scale of ten points, with lower scores (scores ≤ 6) representing worse prognoses for the neonate, and is usually applied in the first and fifth minute after birth [3]. The score measured in the fifth minute is more strongly associated with short- and long-term morbidity compared to that of the score in the first minute [4]. Most cases of low APGAR scores are secondary to neonatal hypoxia/asphyxia, but a few can have other causes such as depression by drugs, infections, and extreme prematurity [5]. This lack of absolute correspondence between the APGAR score and hypoxia/asphyxia motivated some authorities to publish articles recommending not to consider the low APGAR score and hypoxia/asphyxia as the same condition [6, 7]. Within this context and with the purpose of having a precise diagnosis of hypoxia/asphyxia, many institutions adopted the practice of collecting a sample of blood from the umbilical cord soon after birth to measure pH, blood gases, and concentration of bases. The documentation of acidosis, especially metabolic acidosis, would then be a more objective diagnosis of hypoxia/asphyxia. Some institutions utilize this tool selectively in cases of low APGAR scores, or after interventions indicated for suspected fetal distress. The objective is to obtain a more precise evaluation to better program neonatal care, but one that can also be utilized for medico-legal purposes [8, 9]. However, some authors propose utilizing this tool in every birth [10].
When compared to the APGAR score, however, postpartum cord blood analysis is reportedly worse at predicting adverse neonatal outcomes. The APGAR score is a better predictor of death in both term and preterm neonates [11]. It is also a better predictor of neonatal morbidity [12]. With an APGAR score of 7 or more in the fifth minute and abnormal values of cord blood analysis, only a small residual proportion of neonates had an abnormal evolution [13, 14].
Regarding long-term morbidity, Leinonen et al. [15] carried out a population-based study and reported that APGAR scores < 4 are a better predictor of neurologic morbidity compared to those of the lower cutoff values of cord blood analysis. Kayani et al. [16] compared children aged 2 years who had umbilical vein pH below 7.00 and normal APGAR, with that of controls with both normal pH and APGAR. They did not find any differences in the fields of cognition and motricity. Many other studies reported an association of low APGAR score, especially the fifth minute score, with many short-term as well as long-term adverse neonatal outcomes [17,18,19,20,21,22,23,24,25,26,27,28]. Razzaz et al. [29] reported a significant difference in neonatal morbidity and mortality even within what is considered the normal range of APGAR scores, from 8 to 10. Chong et al. [30] argue that a cutoff value of 8 for APGAR in the fifth minute would be a better predictor of mortality in the first year of life than that of the traditional value of ≤ 6. We can probably state that, if we have to adopt a single tool to evaluate the condition and risk of adverse outcomes of the neonate, the APGAR score is the most comprehensive.
Most cases of low APGAR scores are secondary to intrapartum events [31]. Cardiotocography (CTG) is probably the most utilized method to detect fetal distress during labor; however, reportedly, it has a great proportion of false positive results [32] and a not negligible proportion [33,34,35] of false negative results. A recent study showed that the analysis of the blood of the fetal scalp during labor is not an adequate tool to distinguish false positive from false negative tracings of CTG, due to its low sensitivity [36]. We can probably assume that, despite being an uncommon condition, a low APGAR score (especially in the fifth minute) is a relevant adverse outcome, and with the resources available presently, we cannot completely avoid its occurrence—hence, our interest in finding variables associated with this outcome, with the aim of identifying potential preventive measures.
Several research studies assessed antenatal and intrapartum variables associated with low APGAR score, many of which studied a single specific variable [21, 37,38,39,40,41,42,43,44], while others studied more than one [28, 45,46,47,48,49,50]. Some of these latter studies, however, assessed a relatively small number of variables. We believe that this issue has received less attention than it deserves and that a study which assesses a greater number of variables is necessary.
Objectives
To identify antenatal and intrapartum variables associated with APGAR scores ≤ 6 in the 5th minute in term pregnancies.
Methods
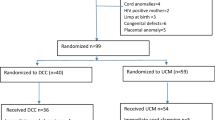
We conducted a retrospective case–control study. The cases were births of term singleton pregnancies assisted in a single institution and had APGAR score < 7 in the fifth minute. The controls were the two births following a case, which fulfilled the same criteria as that of the controls, but had APGAR scores ≥ 7 in the fifth minute. Intrapartum scalp blood analysis and postpartum cord blood analysis are not available in the institution. The following conditions were excluded: multiple gestation, non-cephalic presentations, stillbirths, malformations, or congenital diseases detected during prenatal care or in the first neonatal evaluation, and births with missing data about gestational age or APGAR score in the fifth minute.
The institution is Hospital Municipal Universitário de São Bernardo do Campo—a public teaching hospital dedicated to women’s health. It is located in São Bernardo do Campo, an industrial city with approximately 840,000 inhabitants, in the metropolitan area of São Paulo, Brazil. The hospital attends to an average of 4000 births per year and has 24-h dedicated teams of gynecologists/obstetricians, neonatologists, and anesthesiologists. It is also a field for the training of medical students and has medical residents in these three specialties. It is the only public institution that offers care for birth in this city and consequently attends to a mix of high- and low-risk pregnancies.
The search for the cases was performed through the book of births, filled by nurses in the obstetric ward, which records data about the APGAR score, gestational age, and presentation of the fetus (besides other details) of all assisted deliveries. We then manually accessed the paper-based medical records of all potential cases and controls for review. The data were extracted by the authors (LCMJ, CNP, CSG, ECGP) and entered into a pre-specified chart to obtain more homogeneity.
The variables assessed were those cited in previous works as being associated with low APGAR score [21, 28, 37,38,39,40,41,42,43,44,45, 47, 49, 50]. These were: maternal age, < 6 prenatal visits, smoking habit, regular use of illicit drugs, gestational age as continuous variable, by the best criterion, at the discretion of the researcher (most of the women had first trimester ultrasound), parity, one previous cesarean; maternal diseases: hypertensive diseases as a single variable including chronic hypertension, gestational hypertension and preeclampsia, diabetes (gestational and overt as a single variable), “other maternal diseases”, placenta previa; and birth conditions/risk factord related to birth: mode of delivery (prelabor cesarean section or women in labor), labor induction with oxytocin or prostaglandins (mostly misoprostol), labor augmentation with oxytocin, duration of labor from the beginning of the active phase of the first stage until delivery (as continuous variable), time from rupture of membranes until delivery both as continuous and dichotomous variables (excluding rupture at the moment of delivery, that is, 0 min), tachysystole (six or more contractions in 10 min) < 3 h before birth, intrapartum fever (37.8º C or more), meconium-stained amniotic fluid, malodorous amniotic fluid, abnormalities in clinical auscultation of fetal heart rate (sustained bradycardia or profound decelerations), abnormal CTG [51], wrong interpretation of CTG, fetal sex and birth weight, and labor accidents: placental abruption, cord prolapse, shoulder dystocia and uterine rupture. We created the composite outcome “obstetric catastrophes”, which included placental abruption, uterine rupture, cord prolapse, shoulder dystocia, and eclampsia, and conducted the analysis with these latter outcomes only as a composite outcome. Some known risk factors were not included, since we anticipated some difficulties in collecting data with accuracy: fetal growth restriction, because of a low antepartum diagnostic rate; oligohydramnios, because most of the women did not have an ultrasound examination shortly before birth; duration of the second stage, because the strategy of documentation (partograph) in the charts tended to underestimate the true duration of this period; and epidural analgesia, because most of the women did not receive conduction analgesia, and those who did, mostly received combined spinal–epidural analgesia. We considered the beginning of the active phase from the point at which cervical dilatation began progressing at an average rate of at least 1 cm/h, usually beginning with a 45 cm dilatation. We anticipated lack of accuracy in the information given by the woman about the exact time of rupture of the membranes because of frequent confusion with cervical secretions, urine, and aqueous fluid of candidiasis. We considered the time zero of rupture, for both cases and controls, as the moment at which a member of the attending team detected ruptured membranes. This strategy tends to underestimate the average time elapsed from rupture to delivery, especially for those women who had rupture before admission. Since the main purpose is to compare the duration in cases vs. that in controls, this underestimation is of less importance. Further, we believe there would be no reason to assume that the size or the direction of this underestimation would be different in cases vs. that in controls. In our hospital, most women are submitted to intrapartum CTG. However, it is very uncommon to perform strictly continuous CTG, in the sense that the tracing covers the entire process of labor, even in high-risk pregnancies. Usually, tracings are performed for 20 min in intervals of about 2 h, changing to smaller intervals in the second stage of labor or after abnormal tracings (usually class II) are found. This routine is utilized indistinctly in high- and low-risk pregnancies. In the intervals between CTG tracings, the fetal heart rate is measured clinically by auscultation at 30-min intervals, before, during, and after a contraction, and at 15-min intervals in the second stage. Most of the times, abnormalities in auscultation are confirmed through CTG. In our study, all CTG tracings were reviewed by the leading author (LCMJ). For the study, the definition of wrong interpretation of CTG was made by LCMJ with at least one more author, and disagreement was resolved through consensus. An interpretation was considered wrong, only if it was for “less”, that is, abnormal tracings wrongly considered as normal, which could lead to postponing or not deciding on a necessary intervention. We did not consider a wrong interpretation when it was for “more”, that is, a normal tracing which was considered as abnormal. This is because such a mistake could lead to an unnecessary intervention, but not a low APGAR score. Since abnormal CTG before prelabor cesareans was an uncommon occurrence, we decided to perform analyses with CTG only in the group of laboring women (see below).
Taking into account the previous reports of a protective effect of prelabor cesarean for severe neonatal complications in term pregnancy [52,53,54], we also anticipated that most of the risk factors for low APGAR would express their effect more strongly in laboring women than in those women subjected to prelabor cesareans. Therefore, we planned to conduct analyses (a) with the whole sample and (b) with laboring women (regardless of the final mode of delivery) excluding placental abruption. Placental abruption was excluded because it is a serious condition and it is usually difficult to decide if a case of abruption can be defined as labor.
We deliberately decided to assess the maximum number of variables. As stated in the introduction, the authors believe that low APGAR and other adverse neonatal outcomes in term pregnancy have received less attention in the literature than it deserves. Consequently, we gave an “exploratory” character to our study by not restricting it to a few specific potential associated factors.
The calculation of the size of the sample was not feasible because of the great number of variables assessed which could be associated with the outcome. Besides this, we were limited by the fact that the book of births covered the period from April 2013 to the present day. Thus, we decided to study the births from this date until July 31, 2020 when we began to conduct the analyses. We did not study the neonatal outcomes consequent to low APGAR score, such as neonatal death, admission to neonatal intensive care unit, and others, but rather, as stated above, focused on antenatal and intrapartum conditions that could be determinants of low APGAR. We excluded preterm births because prematurity, especially extreme prematurity, by itself can be the cause of a low APGAR score. Consequently, studying a sample with preterm births would make prematurity an important confounding factor for the other variables being assessed.
Since we collected data of births assisted until the end of July 2020, maternal infection with COVID-19 virus could be a new risk factor for a low APGAR score. However, none of the pregnant women included in the study had a clinical picture or laboratory findings suggestive of this condition.
We did not make imputations for missing data. The association between potential risk factors and the outcome was assessed through the Chi square test or Fischer’s exact test for categorical variables and Student’s t test or Mann–Whitney U test for continuous variables. Values of odds ratios (OR) and 95% confidence intervals (CI 95%) were calculated for each analysis. Mult-variate analyses were conducted through logistic regression. A significant association was considered for values of p < 0.05. Data were first entered into an EpiData (EpiData Association, Odense, Denmark) database. Stata (StataCorp. 2013. Stata Statistical Software: Release 13. College Station, TX: StataCorp LP) was used for the analyses.
All procedures performed were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was approved by the Ethics Committee of the Faculty of Medicine of ABC under number 1.9893.818 (Ethical process n. CAEE 3,481,414.0.0000.0082). The requirement of informed consent was waived by the committee as medical records and the data were anonymously analyzed and presented.
Results
From April 1, 2013 to July 31 2020, 100 cases of low APGAR score which fulfilled the inclusion criteria were found. For these cases, we included 190 controls, with a total of 290 observations. During this period, 28,453 term births were assisted, of which 132 term births (exclusions not made) had fifth minute APGAR score < 7 (a rate of 0.46%, or 4.6 cases per 1000 births). In this period, the monthly rate of cesarean sections for all births varied from 34 to 40%, with no clear tendency of growth or decline, with an average of about 36% for the entire period.
Table 1 shows the associations of variables with the outcome in the whole sample studied, and Table 2 shows the associations in the group of women in labor. Some variables in Table 2, like tachysystole and abnormal CTG, are not presented in Table 1, because they apply only to women in labor. Of the 27 variables that were studied, 12 were significantly associated with the outcome. Only the significantly associated variables were included in the tables, except for sex of the fetus and hypertensive disease. This was done to show that the sex of the fetus became significant in the context of laboring women, and that hypertensive disease, considered an important risk factor for the outcome, did not reach statistical significance. As expected, there was a strong association with the composite outcome “obstetric catastrophes”: OR 11.00; CI 95% 3.10–39.00; p < 0.001. However, these catastrophes accounted for only 15% of the cases of low APGAR score. As we anticipated, a protective effect of prelabor cesarean was found for the outcome, OR 0.37; CI 95% 0.17–0.81; p = 0.011. It is interesting to compare Tables 1 and 2. For all variables that are presented in both the tables, that is, variables that apply to both groups of women (women in labor and whole group), the values of OR are greater in the group of laboring women, despite a smaller sample, with the exception of malodorous amniotic fluid (same number of events) and ruptured membranes of more than 360 min (see commentary in “Discussion”). These findings reinforce the adequacy of our strategy to conduct a separate analysis for laboring women.
We then conducted a multivariate analysis. To be coherent with our arguments, we included in the latter only the analyses conducted on laboring women. Since we had many variables associated with the outcome, we had to make a choice about which variables to include in the multivariate model. We gave priority to variables that were more frequently found in clinical practice and also had greater values of OR. The model which fitted best included nulliparity, fetal sex, < 6 prenatal visits, and abnormal CTG (Table 3).
An interesting finding was the association of low APGAR score with the time of ruptured membranes. This association was found in the analysis by taking the time of ruptured membranes as a dichotomous variable (Tables 1, 2, 4), as well as a continuous variable (mean of 458 min in cases vs. 235 min in controls, p = 0.002; not shown in tables). From the clinical point of view, the duration of labor from the beginning of the active phase is a very likely confounding variable for the time of ruptured membranes in association with low APGAR. This is because long labors tend to have a longer time of ruptured membranes. Although the duration of labor was not associated with the outcome in the univariate analysis (median of 256 min in cases vs. 227 min in controls, p = 0.207; not shown in tables), we decided to conduct a distinct multivariate analysis including only the duration of the active phase of labor as a continuous variable and time of ruptured membranes as a dichotomic variable, to be sure that the duration of the active phase did not confound the association. There was an association between the time of ruptured membranes and low APGAR with two distinct cut-off values, 260 and 360 min, and no association with the duration of labor (Table 4).
Discussion
The present study found many variables associated with an APGAR score < 7 in the fifth minute of term pregnancy. A protective effect of prelabor cesarean was also found for this outcome; consequently, we conducted separate analyses with laboring women, which showed a greater influence of the risk factors over low APGAR in this group. An unexpected strong positive association was noted with the time of ruptured membranes, even after controlling for the duration of labor. The frequency of APGAR < 7 in the fifth minute of 0.46%, in term birth for the whole population of the study period in the institution (without the exclusions made for the study), compares well with recent reports of large series from developed countries, such as 0.9% of Lai et al. [47] (LAI 2017) and 1,4% of Thavarajah et al. [21], both from Australia, which are results after the exclusion of multiple gestation, malformed and non-cephalic; it is also lower than 1.3%, reported by Leinonen et al. [20], from the general population of term birth in Finland. However, it is slightly higher than 0.3% reported by Gutbir et al. [17] from Israel. We do not have a clear explanation for these differences. Similarly, in a period very similar to that of the study (from January 2014 to March 2021, with 28,080 term births), the neonatal death rate during the admission for birth in term pregnancy was 0.81/1000. This decreased to 0.28/1000 after exclusion of serious malformations. This can be considered a low rate.
The cesarean section rate in our hospital is higher than that in most institutions. There are several reasons for this. First, in the private institutions in Brazil, the cesarean rates are very high, reaching more than 80% in the Southeastern region [55], where our institution is located. It is common for women to move from the private to the public sector and vice versa in different pregnancies. Consequently, we have a larger number of women with two or more cesarean sections, compared to that of other hospitals worldwide. Since we do not perform trials of labor in such women, it lowers the number of women that can be subjected to a trial of labor. Besides, we also have many women with one previous cesarean section. Though we do accept a trial of labor in such women, their condition limits the likelihood of a vaginal birth. Second, there is perhaps a lower threshold for the decision of cesarean section in our institution, as well as in other public hospitals in the country, compared to institutions in the developed countries, although this difference is difficult to be measured or proven. Our protocols/guidelines do not include a cesarean section on maternal request. However, in some special and very uncommon cases, it may be allowed, but this has a very low impact on the global cesarean rate.
Some variables which were associated with low APGAR in other studies were not associated in the present one. However, some of these variables were more frequent in the cases than in controls, although not significantly, like “other maternal diseases”, 12.5 vs. 8.1% p = 0.271; “one previous cesarean, 15.9 vs. 12.2% p = 0.416; and intrapartum fever, 2.3% vs. 0, p = 0.065. A significant association with these variables would likely be found with a larger sample.
The question of neonatal outcomes associated with the mode of delivery in term pregnancy has been an issue of great debate. The first and very important aspect is that the vaginal and abdominal routes are not two independent routes. Quite often, cesarean sections are performed to solve the problems of the trial of vaginal birth, such as fetal distress in labor or cephalopelvic disproportion. Many studies reported a higher frequency of respiratory morbidity in prelabor cesareans when compared to vaginal births or with a trial of labor in term pregnancy [56,57,58,59]. However, it is necessary to consider the whole picture of neonatal outcomes. Respiratory morbidity in term pregnancy, although not uncommon, seldom causes death or long-term morbidity. This is quite different from severe hypoxic/ischemic encephalopathy, meconium aspiration syndrome, serious trauma, and other conditions, which are more frequent in a trial of labor or laboring women, when compared to prelabor cesareans. The same can be said about low APGAR scores. The present study showed a significantly lower frequency of this outcome in prelabor cesareans. A recommendation by the National Institutes of Health United States (NIH) in 2006 stated that comparisons of outcomes related to the mode of delivery should be made between prelabor cesareans and other forms of birth [60]. When conducted in this manner, some studies reported a protective effect of prelabor cesarean for serious neonatal morbidities [52,53,54] or neonatal death [53] in term pregnancy, as stated in the Methods section. Some studies showed an association of emergency cesarean delivery with low APGAR [21, 45, 47, 48], but did not discuss the above aspects in detail. In the works of Thavarajah et al. [21] and Lai et al. [47], both conducted in the same institution and similar periods, the authors report a significantly lower frequency of low APGAR score in prelabor cesareans and a significantly higher frequency in emergency cesarean and operative vaginal delivery. In the discussion of both the studies, the authors state that emergency cesarean and operative vaginal delivery are important risk factors for low APGAR. This gives the wrong idea of a cause–effect relationship. We had similar results in the present study: a protective effect of prelabor/elective cesarean (Table 1) and a greater significant frequency of emergency cesarean in the cases, 24.4% vs 12.2% in the controls, p = 0.019 (not shown in tables). Most likely, the correct interpretation is that the causes of low APGAR score in emergency cesarean and operative vaginal deliveries are mostly the conditions that indicated the procedure (reverse causality), and not the procedure itself. The finding of a protective effect of prelabor cesarean against low APGAR does not justify, of course, a police of cesarean section for every woman. Cesarean sections are associated with higher maternal morbidity and mortality [61,62,63,64]. Besides this, as stated above, vaginal births without fetal distress have less risks of respiratory morbidity. However, this finding goes against the simplistic statement that “the lower the cesarean rates, the better for the mother and her baby”.
Another relevant finding, as stated in “Results”, is that we did not find associations between the outcome with maternal diseases. In the analyses conducted within the group of laboring women, hypertensive disease almost reached significance, with an OR of 1.81 and a p value of 0.089 (Table 2). As stated above, it is likely that with a larger sample, a significant result would be obtained. Moreover, the variable “other maternal diseases”, although more common in the cases group, did not reach a significant association, as stated above. This weak expression of maternal diseases may be secondary to its low frequency in the population compared with other risk factors such as nulliparity or male sex. Besides this, many pregnant women with serious diseases, like severe preeclampsia, have medically indicated deliveries in the preterm period for maternal or fetal indications. But it is probably true that other conditions, such as nulliparity, fetal sex, maternal age, and conditions related to the course of labor, have a greater influence on the outcome than is usually considered. This raises the question: is the concept of high-risk pregnancy interchangeable with the concept of high-risk birth? The belief that the conditions which are good predictors of adverse neonatal outcomes at term are mainly those who classify the pregnancy as high risk has been challenged by some authors [50, 53, 65, 66]. Similar comments could be made about the outcome of “obstetric catastrophes”. In this composite outcome, we included acute conditions, usually associated with serious morbidity, and are almost unpredictable. Despite being strongly associated with the outcome, only 15% of the cases of low APGAR in our study were related to these conditions; the remaining 85% of cases were in “normal” birth contexts.
Some studies have reported the utility of Doppler assessment of umbilical and fetal vessels at the end of pregnancy in the prediction of low APGAR score and other adverse perinatal outcomes [67, 68]. Till date we have not been able to subject all women admitted for spontaneous labor or labor induction to Doppler assessment, though it is available in our institution.
Much controversy exists about the benefit of intrapartum CTG in preventing adverse outcomes for neonates [33, 69, 70]. In the present study, more than the association of abnormal CTG with low APGAR score, the strong association of wrong interpretation of CTG with the outcome (OR 2.99; p = 0.006; Table 2) is an argument to affirm that, at least in our context, intrapartum CTG very likely has some benefit in preventing low APGAR.
A third interesting finding was the association of the outcome with the time of ruptured membranes. It is probably the most unique finding of our work. We did not expect to find this association. We included this variable because of the report of Martinez-Biarge et al. [71], who found an association between time of ruptured membranes and hypoxic–ischemic encephalopathy, which could be a proxy for low APGAR score. However, they did not conduct analyses with low APGAR as an outcome. To our knowledge, only few studies reported this association. Omokhodion et al. [72], in Nigeria, found an association of APGAR score less than 7 at 1 min, but not at 5 min, with rupture of membranes for more than 24 h before labor. Kaye [73] reported an association of five-minute APGAR scores ≤ 4 with rupture of membranes more than 24 h before labor in term pregnancies in Uganda. However, both the studies included only cases that had been referred to a tertiary center; the authors inform that in the regions where the studies were conducted, most uncomplicated labors occurred at the primary level (not in hospitals). Hence, in both works, the frequency of low APGAR in the fifth minute was very high: 4% in the work of Omokhodion et al. [72] and 13.1% in the one of Kaye [73]. Also, they did not control the duration of labor. Salustiano et al. [49], in Brazil, found a greater duration of ruptured membranes in term pregnancy in cases (APGAR < 7 in five minutes), 10 h, than in controls, 8 h, but the difference was not significant (p = 0.11).
Although we did not include this variable in the multivariate analysis, we controlled for the duration of labor, which could be an important confounding variable, and the association persisted. The data of the present work does not allow us to find a clear motif for this association, but we attempt to put forward some theoretical explanations. Most would evoke fetal infection as the probable cause. Since we had only two cases of intrapartum fever, it is unlikely that intrapartum infection would be the only explanation for this association. Besides this, low APGAR score in the fifth minute is not so strongly related to infection. Jessop et al. [39] studied the association of histological findings related to inflammation in placental tissues in term singletons with adverse outcomes, including APGAR score < 7 in the first minute. They found an association of these findings, especially funisitis, with low APGAR score and the other outcomes. They suggest that fetal inflammatory response syndrome, and not necessarily clinically evident infection, could lead to adverse outcomes. Mynarek et al. [74] studied the association between ruptured membranes and the risk of cerebral palsy in term pregnancy. They found an association between rupture of more than 24 h before labor and the outcome. They also found that the pattern of cerebral MRI suggestive of hypoxic–ischemic injuries increased with increased intervals from rupture to delivery. They speculate that rupture of membranes could somehow increase the vulnerability of the fetal brain to hypoxic-ischemic injury. Another possible explanation could be more frequent cord compression secondary to oligohydramnios after rupture. Although a more frequent cord compression could contribute to low APGAR, some reports challenge the concept of this mechanism as the only explanation for this outcome. Ghosh et al. [75] studied two groups of low-risk term pregnancies, one with intact and the other with ruptured membranes. Both groups were submitted to the evaluation of amniotic fluid index (AFI) at admission for labor. They then compared within each group the frequency of adverse outcomes between oligohydramnios (AFI ≤ 50 mm) and normal amniotic fluid. They found a greater significant difference in operative delivery for fetal distress in the group with oligohydramnios with ruptured membranes, but not in the group with oligohydramnios and intact membranes. Interestingly, in principle, we would expect to have a greater frequency of interventions in the group with intact membranes, since, within this group, at least some cases of oligohydramnios would be secondary to placental malfunction. In addition, Omokhodion et al. [72], Kaye [73], and Mynarek et al. [74] reported an association between low APGAR score or markers of hypoxia/asphyxia with the time of ruptured membranes before established labor, when the frequency and intensity of contractions are low. Also in the present study, for the variable rupture of membranes for > 360 min, we found a higher value of OR in the general group, which also included non-laboring women (OR, 2.30; p = 0.003; Table 1), than in the group of laboring women (OR, 2.03; p = 0.031; Tables 1, 2). If more frequent cord compression was the only mechanism, the contrary would be expected. However, if we consider the condition of prelabor rupture of membranes in the context of the preterm population, the findings above are not in accordance with the ones of the PPROMEXIL trial [76], which found no significant difference in neonatal outcomes with labor induction at 34 weeks compared with expectant management until 37 weeks, and also with PPROMT trial [77], which reported better neonatal outcomes with expectant management in the same comparison. Other mechanisms may play a role in this specific population. Our findings are not enough to change management protocols about ruptured membranes, but they raise the question of the likely association between the time of rupture of membranes and low APGAR score in term pregnancy, and the need to study this issue more comprehensively.
The limitations of our study are similar to those inherent to retrospective studies, with the risk of information bias. Besides this, we could not assess some important variables, such as the duration of the second stage of labor and fetal growth restriction, due to unreliable information. The strong aspects of this study are the case–control design, which enabled us to identify many associations with a relatively small sample; the analyses for mode of delivery were conducted correctly; the confirmation of the association between variables not related to maternal diseases with the outcome as reported in recent studies and finding a unique association with time of ruptured membranes, even after controlling for duration of labor.
The authors of the present study intend to utilize the same sample to build a risk score for predicting low APGAR score in term pregnancy with variables that are accessible from the moment of admission for labor. This will be reported in another article.
References
Apgar V (1953) A proposal for a new method of evaluation of the newborn infant. Curr Res Anesth Analg 32(4):260–267
Skolnick AA (1996) Apgar quartet plays perinatologist’s instruments. JAMA 276:1939–1940. https://doi.org/10.1001/jama.1996.03540240017009
Cunningham FG, Leveno KJ, Bloom SL et al. (2018). Transtornos hipertensivos. Cap 40, Sec 11 In Cunningham FG, Leveno KJ, Bloom SL et al. (2018) Williams Obstetrics, 25. ed., São Paulo: Ed. McGraw Hill. pp. 710–55 (710–11).
Watterberg KL, Aucott S, Benitz WD et al (2015) The Apgar Score. Pediatrics 136(4):819–822. https://doi.org/10.1542/peds.2015-2651
Hogan L, Ingemarsson I, Thorngren-Jerneck K, Herbst A (2007) How often is a low 5-min Apgar score in term newborns due to asphyxia? Eur J Obstet Gynecol Reprod Biol 130(2):169–175. https://doi.org/10.1016/j.ejogrb.2006.03.002
American Academy of Pediatrics (1996) Use and abuse of the Apgar score. Pediatrics 98:141–142. https://doi.org/10.1542/peds.98.1.141
ACOG Committee on Obstetric Practice, American Academy of Pediatrics Committee on Fetus and Newborn (1996) Use and abuse of the Apgar score. Number 174 — July 1996 (Replaces No. 49, November 1986) Committee on Obstetric Practice and American Academy of Pediatrics: Committee on Fetus and Newborn. Int J Gyncol Obstet 54(3):303-5. https://doi.org/10.1016/S0020-7292(96)90089-2
Harris M, Beckley SL, Garibaldi JM, Keith RD, Greene KR (1996) Umbilical cord blood gas analysis at the time of delivery. Midwifery 12(3):146–150. https://doi.org/10.1016/S0266-6138(96)90059-5
Thorp JA, Rushing RS (1999) Umbilical cord blood gas analysis. Obstet Gynecol Clin North Am 26(4):695–709. https://doi.org/10.1016/s0889-8545(05)70107-8
White CRH, Doherty DA, Cannon JW, Kohan R, Newnham JP, Pennell CE (2016) Cost effectiveness of universal umbilical cord blood gas and lactate analysis in a tertiary level maternity unit. J Perinat Med 44(5):573–584
Casey BM, McIntire DD, Leveno KJ (2001) The continuing value of the Apgar score for the assessment of newborn infants. N Engl J Med 344(7):467–471. https://doi.org/10.1056/NEJM200102153440701
Yeh P, Emary K, Impey L (2012) The relationship between umbilical cord arterial pH and serious adverse neonatal outcome: analysis of 51,519 consecutive validated samples. BJOG 119(7):824–831. https://doi.org/10.1111/j.1471-0528.2012.03335.x
Anayegbunam A, Fleischer A, Whitty J, Brustman G, Langer O (1991) Association between umbilical artery cord pH, five-minute Apgar scores and neonatal outcome. Gynecol Obstet Invest 32(4):220–223. https://doi.org/10.1159/000293036
Sabol AB, Caughei AB (2016) Acidemia in neonates with a 5-minute Apgar score of 7 or greater - What are the outcomes? Am J Obstet Gynecol 215(4):486.e1–6. https://doi.org/10.1016/j.ajog.2016.05.035
Leinonen E, Gissler M, Haataja L et al (2019) Umbilical artery pH and base excess at birth are poor predictors of neurodevelopmental morbidity in early childhood. Acta Paediatr 108(10):1801–1810. https://doi.org/10.1111/apa.14812
Kayani SI, Tan MJ, Shaw NJ et al (2014) Two-year outcomes for infants with low cord pH at birth. J Matern Fetal Neonatal Med 27(10):1010–1014. https://doi.org/10.3109/14767058.2013.847915
Gutbir Y, Wainstock T, Sheiner E et al (2020) Low Apgar score in term newborns and long-term infectious morbidity: a population-based cohort study with up to 18 years of follow-up. Eur J Pediatr 179(6):959–971. https://doi.org/10.1007/s00431-020-03593-9
Leybovitz-Haleluya N, Wainstock T, Sheiner E, Segal I, Landau D, Walfisch A (2019) Low Apgar scores in term newborns and long-term gastro-intestinal morbidity: a population-based cohort study with up to 18 years of follow-up. J Matern Fetal Neonatal Med 32(10):1609–1614. https://doi.org/10.1080/14767058.2017.1411475
Ernest E, Wainstock T, Sheiner E, Segal I, Landau D, Walfisch A (2019) Apgar score and long-term respiratory morbidity of the offspring: a population-based cohort study with up to 18 years of follow-up. Eur J Pediatr 178(3):403–411. https://doi.org/10.1007/s00431-018-03311-6
Leinonen E, Gissler M, Haataja L et al (2018) Low Apgar scores at both one and five minutes are associated with long-term neurological morbidity. Acta Paediatr 107(6):942–951. https://doi.org/10.1111/apa.14234
Thavarajah H, Flatley C, Kumar S (2018) The relationship between the five minute Apgar score, mode of birth and neonatal outcomes. J Matern Fetal Neonatal Med 31(10):1335–1341. https://doi.org/10.1080/14767058.2017.1315666
Persson M, Razaz N, Tedroff K, Joseph KS, Cnattingius S (2018) Five and 10 minute Apgar Scores and risk of cerebral palsy and epilepsy: population based cohort study in Sweden. BMJ 360:k207. https://doi.org/10.1136/bmj.k207
Eun S, Lee JM, Yi DY et al (2016) Assessment of the association between Apgar scores and seizures in infants less than 1 year old. Seizure 37:48–54. https://doi.org/10.1016/j.seizure.2016.03.001
Tweed EJ, Mackay DF, Nelson SM, Cooper SA, Pell JP (2016) Five-minute Apgar score and educational outcomes: retrospective cohort study of 751,369 children. Arch Dis Child Fetal Neonatal Ed 101(2):F121–F126. https://doi.org/10.1136/archdischild-2015-308483
Iliodromiti S, Mackay DF, Smith GCS, Pell JP, Nelson SM (2014) Apgar score and the risk of cause-specific infant mortality: a population-based cohort study. Lancet 384(9956):1749–1755. https://doi.org/10.1016/s0140-6736(14)61135-1
Li F, Wu T, Lei X, Zhang H, Mao M, Zhang J (2013) The Apgar score and infant mortality. PLoS ONE 8(7):e69072. https://doi.org/10.1371/journal.pone.0069072
Sun Y, Vestergaard M, Pedersen CB, Christensen J, Olsen J (2006) Apgar scores and long-term risk of epilepsy. Epidemiology. https://doi.org/10.1097/01.ede.0000208478.47401.b6
Thorngren-Jerneck K, Herbst A (2001) Low 5-minute Apgar score: a population-based register study of 1 million term births. Obstet Gynecol 98(1):65–70. https://doi.org/10.1016/S0029-7844(01)01370-9
Razzaz N, Cnattingius S, Joseph KS (2019) Association between Apgar scores of 7 to 9 and neonatal mortality and morbidity: population based cohort study of term infants in Sweden. BMJ 7(365):l1656. https://doi.org/10.1136/bmj.l1656
Chong DSY, Karlberg J (2004) Refining the Apgar score cut-off point for newborns at risk. Acta Paediatr 93(1):53–59 (PMID: 14989440)
Hayes B, McGarvey C, Mulvany S et al (2013) A case-control study of hypoxic-ichemic encephalopathy in newborn infants at > 36 weeks gestation. Am J Obstet Gynecol 209(1):29.e1-29.e19. https://doi.org/10.1016/j.ajog.2013.03.023
Pinas A, Chandraharan E (2016) Continuous cardiotocography during labour: Analysis, classification and management. Best Pract Res Clin Obstet Gynaecol 30:33–47. https://doi.org/10.1016/j.bpobgyn.2015.03.022
Farquhar CM, Armstrong S, Masson V, Thompson JMD, Sadler L (2020) Clinician Identification of Birth Asphyxia Using Intrapartum Cardiotocography Among Neonates With and Without Encephalopathy in New Zealand. JAMA Netw Open 3(2):e1921363. https://doi.org/10.1001/jamanetworkopen.2019.21363
Schifrin BS, Soliman M, Koos B (2016) Litigation related to intrapartum fetal surveillance. Best Pract Res Clin Obstet Gynaecol 30:87–97. https://doi.org/10.1016/j.bpobgyn.2015.06.007
Bogdanovic G, Babovic A, Rizvanovic M, Ljuca D, Grgic G, Djuranovic-Milcic J (2014) Cardiotocography in the prognosis of perinatal outcome. Med Arh 68(2):102–105. https://doi.org/10.5455/medarh.2014.68.102-105
Al Wattar BH, Lakhiani A, Sacco A et al (2019) Evaluating the value of intrapartum fetal scalp blood sampling to predict adverse neonatal outcomes: a UK multicentre observational study. Eur J Obstet Gynecol Reprod Biol 240:62–67. https://doi.org/10.1016/j.ejogrb.2019.06.012
Blankenship SA, Raghuraman N, Delhi A et al (2020) Association of abnormal first stage of labor duration and maternal and neonatal morbidity. Am J Obstet Gynecol 223:445.e1–15. https://doi.org/10.1016/j.ajog.2020.06.053
Dior UP, Kogan L, Eventof-Friedman S et al (2016) Very high intrapartum fever in term pregnancies and adverse obstetric and neonatal outcomes. Neonatology 109(1):62–68. https://doi.org/10.1159/000440938
Jessop FA, Lees CC, Pathak S, Hook CE, Sebire NJ (2016) Funisitis is associated with adverse neonatal outcome in low-risk unselected deliveries at or near term. Virchows Arch 468(4):503–507. https://doi.org/10.1007/s00428-015-1899-0
Almeida NK, Almeida RM, Pedreira CE (2015) Adverse perinatal outcomes for advanced maternal age: a cross-sectional study of Brazilian births. J Pediatr (Rio J) 91:493–498. https://doi.org/10.1016/j.jped.2014.12.002
Aviram A, Raban O, Melamed N, Hadar E, Wiznitzer A, Yogev Y (2013) The association between young maternal age and pregnancy outcome. J Matern Fetal Neonatal Med 26(15):1554–1558. https://doi.org/10.3109/14767058.2013.794212
Wallin MC, Ekström P, Marsál K, Källén K (2010) Apgar score and perinatal death after one previous caesarean delivery. BJOG 117(9):1088–1097. https://doi.org/10.1111/j.1471-0528.2010.02614.x
Chen XK, Wu SW, Fleming N et al (2007) Teenage pregnancy and adverse birth outcomes: a large population based retrospective cohort study. Int J Epidemiol 36(2):368–373. https://doi.org/10.1093/ije/dyl284
Källén K (2001) The impact of maternal smoking during pregnancy on delivery outcome. Eur J Public Health 11(3):329–333. https://doi.org/10.1093/eurpub/11.3.329
Wen PY, Broom E, Flatley C, Kumar S (2020) Maternal demographic and intrapartum antecedents of severe neonatal outcomes at term. J Matern Fetal Neonatal Med 33(12):2103–2108. https://doi.org/10.1080/14767058.2018.1540581
Crovetto F, Fumagalli M, De Carli A et al (2018) Obstetric risk factors for poor neonatal adaptation at birth. J Matern Fetal Neonatal Med 31(18):2429–2435. https://doi.org/10.1080/14767058.2017.1344635
Lai S, Flatley C, Kumar S (2017) Perinatal risk factors for low and moderate five-minute Apgar scores at term. Eur J Obstet Gynecol Reprod Biol 210:251–256. https://doi.org/10.1016/j.ejogrb.2017.01.008
De Zorzi PM, Madi JM, Rombaldi RL et al (2012) Perinatal factors associated with pH <7.1 in umbilical artery and Apgar 5 min <7.0 in term newborn. Rev Bras Ginecol Obstet. https://doi.org/10.1590/S0100-72032012000800007
Salustiano EMA, Campos JADB, Ibidi SM et al (2012) Low Apgar scores at 5 minutes in a low risk population: maternal and obstetrical factors and postnatal outcome. Rev Assoc Med Bras 58(5):587–593. https://doi.org/10.1590/S0104-42302012000500017
Palsodottir K, Dagbjartsson A, Thorkelsson T, Hardartottir H (2007) Birth asphyxia and hypoxic ischemic encephalopathy, incidence and obstetric risk factors. Laeknabladid 93(9):595–601 (PMID: 17823499)
Macones GA, Hankins GD, Spong CY et al (2008) The 2008 NICHHD workshop report on electronic fetal monitoring: update on definitions, interpretation, and research guidelines. J Obstet Gynecol Neonatal Nurs 37(5):510–515. https://doi.org/10.1111/j.1552-6909.2008.00284.x
Dahlgren LS, von Dadelszen P, Christilaw J et al (2009) Caesarean section on maternal request: risks and benefits in healthy nulliparous women and their infants. J Obstet Gynaecol Can 31(9):808–817. https://doi.org/10.1016/S1701-2163(16)34299-2
Machado Junior LC, Sevrin CE, Oliveira E et al (2014) Association between mode of delivery and neonatal deaths and complications in term pregnancy: a cohort study in Brazil. Minerva Pediatr 66(2):111–122
Liu X, Landon MB, Cheng W, Chen Y (2015) Cesarean delivery on maternal request in China: what are the risks and benefits? Am J Obstet Gynecol 212:817.e1–9. https://doi.org/10.1016/j.ajog.2015.01.043
Alonso BD, Silva FMBD, Latorre MDRDO et al (2017) Ceasarean birth rates in public and privately funded hospitals: a cross-sectional study. Rev Saude Publica. https://doi.org/10.11606/S1518-8787.2017051007054
Farchi S, Di Lallo D, Franco F et al (2009) Neonatal respiratory morbidity and mode of delivery in a population-based study of low-risk pregnancies. Acta Obstet Gynecol Scand 88(6):729–732. https://doi.org/10.1080/00016340902818154
Quiroz LH, Chang H, Blomquist J, Okoh YK, Handa VL (2009) Scheduled cesarean delivery: maternal and neonatal risks in primiparous women in a community hospital setting. Am J Perinatol 26(4):271–277. https://doi.org/10.1055/s-0028-1103155
Hansen AK, Wisborg K, Uldbjerg N, Henriksen TB (2008) Risk of respiratory morbidity in term infants delivered by elective caesarean section: cohort study. BMJ 336(7635):85–87. https://doi.org/10.1136/bmj.39405.539282.be
Richardson BS, Czikk MJ, da Silva O, Natale R (2005) The impact of labor at term on measures of neonatal outcome. Am J Obstet Gynecol 192(1):219–226. https://doi.org/10.1016/j.ajog.2004.06.034
National Institute of Health (2006) State-of-the-Science conference statement on Cesarean Delivery on Maternal Request. NIH consensus Science Statements 23(1):1–29 (PMID: 17308552)
Machado Junior LC, Sevrin CE, Oliveira E et al (2009) Association between mode of delivery and maternal complications in a public hospital in Greater Metropolitan São Paulo. Brazil Cad Saude Publica 25(1):124–132. https://doi.org/10.1590/s0102-311x2009000100013
Yang J, Armson BA, Attenborough R et al (2022) Survey of mode of delivery and maternal and perinatal outcomes in Canada. J Obstet Gynaecol Can. https://doi.org/10.1016/j.jogc.2022.04.017
Kilsztajn S, Carmo MS, Machado LC Jr et al (2007) Caesarean sections and maternal mortality in Sao Paulo. Eur J Obstet Gynecol Reprod Biol 132(1):64–69. https://doi.org/10.1016/j.ejogrb.2006.06.005
Esteves-Pereira AP, Deneux-Tharaux C, Nakamura-Pereira M et al (2016) Caesarean delivery and postpartum maternal mortality: a population-based case control study in Brazil. PLoS ONE 11(4):e0153396. https://doi.org/10.1371/journal.pone.0153396
Joyce NM, Tully E, Kirkham C, Dicker P, Breathnach FM (2018) Perinatal mortality or severe neonatal encephalopathy among normally formed singleton pregnancies according to obstetric risk status:" is low risk the new high risk?" A population-based cohort study. Eur J Obstet Gynecol Reprod Biol 228:71–75. https://doi.org/10.1016/j.ejogrb.2018.06.010
Westergate JA, Gunn AJ, Gunn JA (1999) Antecedents of neonatal encephalopathy with fetal acidaemia at term. Br J Obstet Gynaecol 1106(8):774–782. https://doi.org/10.1111/j.1471-0528.1999.tb08397.x
Bligh LN, Alsolai A, Greer RM, Kumar S (2018) Screening for adverse perinatal outcomes: uterine artery Doppler, cerebroplacental ratio and estimated fetal weight in low-risk women at term. J Matern Fetal Neonatal Med 31(24):3301–3307. https://doi.org/10.1080/14767058.2017.1369518
Dunn L, Sherrel H, Kumar S (2017) Review: Systematic review of the utility of the fetal cerebroplacental ratio measured at term for the prediction of adverse perinatal outcome. Placenta 54:68–75. https://doi.org/10.1016/j.placenta.2017.02.006
Alfirevic Z, Devane D, Gyte GM, Cuthbert A (2017) Continuous cardiotocography (CTG) as a form of electronic fetal monitoring (EFM) for fetal assessment during labour. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD006066.pub3
Philopoulos D (2015) Electronic fetal monitoring and its relationship to neonatal and infant mortality in a national database: a sensivity analysis. J Gynecol Obstet Biol Reprod (Paris) 44(5):451–462. https://doi.org/10.1016/j.jgyn.2014.07.003
Martinez-Biarge M, Diez-Sebastian J, Wusthoff CJ et al (2013) Antepartum and intrapartum factors preceding neonatal hypoxic-ischemic encephalopathy. Pediatrics 132(4):e952–e959. https://doi.org/10.1542/peds.2013-0511
Omokhodion FO, Roberts OA, Onadeko MO, Beach JR, Cherry N, Burstyn I (2018) Social, obstetric and environmental determinants of low Apgar score among infants born in four selected hospitals in Ibadan. Nigeria J Obstet Gynaecol 38(4):454–460. https://doi.org/10.1080/01443615.2017.1367764
Kaye D (2003) Antenatal and intrapartum risk factors for birth asphyxia among emergency obstetric referrals in Mulago Hospital, Kampala. Uganda East Afr Med J 80(3):140–143. https://doi.org/10.4314/eamj.v80i3.8683
Mynarek M, Bjellmo S, Lyndersen S et al (2020) Prelabor rupture of membranes and the association with cerebral palsy in term born children: a national registry-based cohort study. BMC Pregnancy Childbirth. https://doi.org/10.1186/s12884-020-2751-3
Ghosh G, Marsál K, Gudmundsson G (2002) Amniotic fluid index in low-risk pregnancy as an admission test to the labor ward. Acta Obstet Gynecol Scand 81(9):852–855. https://doi.org/10.1034/j.1600-0412.2002.810909.x
van der Ham DP, Vijgen SMC, Nijhuis JG et al (2012) Induction of labor versus expectant management in women with preterm prelabor rupture of membranes between 34 and 37 weeks: a randomized controlled trial. PLoS Med 9(4):e1001208. https://doi.org/10.1371/journal.pmed.1001208
Morris JM, Roberts CL, Bowen JR et al (2016) Immediate delivery compared with expectant management after preterm pre-labour rupture of the membranes close to term (PPROMT trial): a randomised controlled trial. Lancet 387(10017):444–452. https://doi.org/10.1016/s0140-6736(15)00724-2
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
Author contributions
LcMJ: Project development, Investigation, Resource, Support in data analysis, Manuscript writing, Manuscript reviewing/editing. CNP: Investigation, Data Collection, Support in manuscript writing. CSG: Investigation, Data Collection, Support in manuscript writing. ECGP: Investigation, Data Collection, Support in manuscript writing. HBC: Data curation, Formal analysis.
Corresponding author
Ethics declarations
Conflict of interest
The authors have not disclosed any competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Junior, L.C.M., Pinto, C.N., Gerencer, C.S. et al. Association of maternal, fetal and labor variables with a low Apgar score in the fifth minute in term pregnancy: a case–control study. Arch Gynecol Obstet 308, 1473–1483 (2023). https://doi.org/10.1007/s00404-022-06832-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-022-06832-6