Abstract
Objective
This study aims to assess the factors associated with the success and failure rate of the external cephalic version (ECV) in breech fetuses. Secondary outcomes were fetal presentation in labor and mode of delivery.
Methods
This cross-sectional study examined the live birth certificates from 2003 through 2020 from US states and territories that implemented the 2003 revision. A total of 149,671 singleton pregnancies with information about ECV success or failure were included. The outcome was ECV success/failure, while the exposures were possible factors associated with the outcome.
Results
The successful ECV procedures were 96,137 (64.23%). Among the successful ECV procedures, the prevalence of spontaneous vaginal delivery was 71.63%. Among the failed ECV procedures, 24.74% had a cephalic presentation at delivery, but 63.11% of these pregnancies were delivered by cesarean section. Nulliparity, female sex, low fetal weight centile, high pre-pregnancy BMI, high BMI at delivery, and high maternal weight gain during pregnancy were associated with an increased ECV failure (p < 0.001). African American, American Indian and Alaska Native race categories were significant protective factors against ECV failure (p < 0.001). Maternal age had a U-shape risk profile, whereas younger maternal age (< 25 years) and old maternal age (> 40 years) were significant protective factors against ECV failure (p < 0.001).
Conclusions
A high prevalence of successful ECV procedures and subsequent spontaneous vaginal delivery were found. The present results found nulliparity, maternal race, maternal age, female fetal sex, low fetal weight, and maternal anthropometric features correlated to ECV results. These findings can potentially improve the knowledge about the factors involved in ECV, allowing more informed counseling to the women undergoing this procedure.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
This study sought to evaluate possible factors associated with the success/failure of the external cephalic version (ECV) in breech fetuses. Nulliparity, female fetal sex, low fetal weight, high pre-pregnancy BMI, high BMI at delivery, and increased weight gain during pregnancy, were negatively correlated to ECV success. Black, American Indian and Alaska Native race categories were associated with ECV success. A U-shape relation to risk was found for maternal age, showing an increase in ECV success at the extremes of the curve. These results can improve the knowledge about the ECV-associated factors, allowing better-informed counseling for women undergoing this procedure. Highlights: Nulliparity, female fetal sex, low fetal weight, high pre-pregnancy BMI, high BMI at delivery, and increased maternal weight gain during pregnancy were associated with an increased risk of external cephalic version failure; Black, American Indian and Alaska Native race categories were significantly associated with external cephalic version success; A U-shape relation to risk was found for maternal age, showing an increase in external cephalic version success at the extremes of the curve in young and old maternal ages. |
Introduction
The reduction of cesarean section (CS) deliveries is within the top priorities in the actual agenda of modern obstetrics. Since the increased number of primary CS leads to an increase in maternal morbidity and mortality [1, 2]. Due to the evidence that emerged during the end of the past century and the beginning of the present century, the preferred method of delivery for term breech presentation has been via CS because of the increased fetal morbidity associated with vaginal delivery [3,4,5]. With the intent to reduce the primary CS rate, the guidelines of the major obstetrics societies recommend the external cephalic version (ECV) of the fetus presenting in breech presentation [6, 7].
The increased burden of fetal complications associated with breech presentation and the women’s decision-making process for the delivery can raise psychological stress and anxiety [8]. For women with a substantial willingness for vaginal delivery, it could be challenging to choose between the diverse options for dealing with breech presentation. Vaginal breech delivery is associated with increased perinatal risks [9]. CS reduces the fetal risk but raises the threat to the mother and future pregnancies [1, 2]. The external cephalic version, if successful, allows a vaginal cephalic delivery but can be associated with complications and failure. A better understanding of the factors that lead to the success or failure of the ECV procedure can lead to improved management and an improved women’s decision-making process. Furthermore, a better understanding of the delivery mode outcome can add valuable information for enhancing these pathways.
The present study aims to assess the factors associated with the success rate of the external cephalic version in breech fetuses. The secondary outcomes were to assess subsequent fetal presentation in labor and the mode of delivery.
Methods
Design, setting, and sample
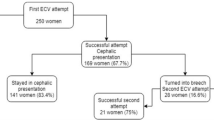
This cross-sectional retrospective study employed birth certificate data from the US National Center for Health Statistics as part of the National Vital Statistics System [10]. The period assessed in this study is from 2003 to 2020. Data about ECV success or failure were collected by introducing the 2003 revision of the US Standard Certificate of Live Birth. The new version was phased in and had not full coverage to all States until 2016 [11]. This study used data from 149,671 singleton births undergone a successful or failed external cephalic version in the states that implemented 2003 birth certificate revision and that have recorded the specific data about the considered maneuver. The following inclusion and exclusion criteria were applied during the sampling procedure. All consecutive records of singleton pregnancies reporting information about a successful or failed external cephalic version were included. The following exclusion criteria were applied in succession: records with imputed values for sex or multiple pregnancies, multiple pregnancies, unknown or not performed ECV maneuver, and chromosomal anomalies. Figure 1 shows the flowchart for the population selection. In reporting this study, we have followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines (http://www.strobe-statement.org/) to ensure that it is documented as thoroughly and accurately as possible. The local Ethics Committee Approval for this study was not required because the data used are de-identified and publicly available. The study was carried out according to the Helsinki declaration.
Measurement
From the original data sets, the following variables were extracted: maternal age, parity, race, pre-pregnancy body mass index (BMI), BMI at delivery, weight gain during pregnancy, data about successful or failed ECV, fetal presentation at delivery, mode of delivery, multiple pregnancies, multiple pregnancies imputation label, neonatal sex, neonatal sex imputation label, gestational age at delivery, neonatal weight, chromosomal anomalies. A detailed explanation of the variables is available at the following link: https://www.cdc.gov/nchs/data_access/Vitalstatsonline.htm#Tools. Maternal age was stratified into five age classes. Parity was coded as nulliparous vs. parous women. The race was coded according to six categories: single Caucasian race, single African American race, American Indian and Alaska Native (AIAN) single race, Asian single race, Native Hawaiian and Other Pacific Islanders (NHOPI) single race, and more than one race. Neonatal weight centile was calculated using the Hadlock prenatal formula and employing neonatal weight and gestational age at delivery [12]. Then, the Neonatal weight centile was categorized into the following strata: < 10th centile, 10–49th centile, 50–90th centile, and > 90 centiles. Fetal presentation at delivery was classified as Cephalic, Breech, other presentation, and Unknown. Mode of delivery was classified as Spontaneous, Forceps, Vacuum, Cesarean, and Unknown. For all the other variables, unknown values were considered as NA. The previous variables were selected according to the known risk factors for ECV failure or to the favoring factors for ECV success [13,14,15,16,17,18,19,20,21,22,23].
Data analysis
All analyses were performed using R software (version 4.2.0) [24]. The two-sided probability value p < 0.05 was considered statistically significant. The normal distribution of continuous variables was evaluated with the Kolmogorov–Smirnov test. Data are presented as the median and interquartile range (IQR) for continuous non-parametric variables; mean ± standard deviation for continuous parametric variables. Categorical variables (dichotomous or polychotomous) were coded as the percentage and the absolute values, except for NA’s missing values (unless otherwise specified). The results of logistic regression models were presented as odds ratio (OR) and 95% confidence interval (CI.95). The following statistical tests were applied for categorical variables: chi-square test or Fisher exact test. For continuous non-parametric variables, we applied the Wilcoxon test, and for parametric ones, the t-test.
Logistic regression analysis was also performed, considering failed ECV as the dependent variable and possible predictors as an independent variable. We took all possible predictive factors with p < 0.05 from univariate analysis in the multivariate model. The multivariate models with and without interactions were analyzed. When considering interactions, the initial multivariate model incorporated all variables and their interactions. When interactions turned out to be non-significant, the analysis without interaction model was employed. In addition, we considered logistic regression models without imputation and with the random imputation of missing values. A sensitivity analysis of the multivariate models was also performed. Pre-pregnancy BMI and BMI at the time of delivery were considered in two separate models because of the strong interaction between the two variables and the higher number of missing values in the BMI at the time of delivery than in pre-pregnancy BMI. The missing values of pre-pregnancy BMI were 26.63%, of BMI at delivery were 61.99%, and of weight gain during pregnancy were 62.44%. The weight gain during pregnancy was not considered in the multivariate analysis because of the high rate of missing values and the strong interaction with BMI. p values in multivariate models were adjusted using the false discovery rate test.
Results
The final study cohort comprises 96,137 successful ECV (64.23%) procedures and 53,534 unsuccessful procedures. The prevalence of female fetuses was 51.34% (76843/149671). Table 1 shows other characteristics of the population studied. Figure 2 shows the pregnancy outcome at delivery considering the successful or unsuccessful ECV. Most of the successful ECVs had a spontaneous birth of a cephalic fetus (Fig. 2A and B). Among the failed ECV procedures, 24.74% had a cephalic presentation at delivery (Fig. 2A and B). 63.11% of these pregnancies will deliver by CS, 34.28% spontaneously, and 2.59% by operative vaginal delivery (the remaining were unknown). Among the failed procedures, 1.06% delivered in breech presentation (spontaneous or operative) (Fig. 2A and B). Among the successful ECV procedures, 2.94% had a breech presentation at delivery, 85.51% of these delivered by CS, 14.43% by vaginal delivery, and the others are unknown (Fig. 2A and B).
Table 1 shows the differences between singleton pregnancies with a successful ECV vs. failed ECV. Table 2 and Fig. 3 show the univariate and multivariate logistic regression without interactions. Figure 3 shows the model without imputation of the missing values and considering the pre-pregnancy BMI. In these models, women younger than 29 and older than 34, the African American race, and high fetal weight centile were protective against ECV failure. Meanwhile, nulliparity, female fetal sex, and low fetal weight centile were risk factors for failed ECV. The same pattern was observed in the model with the imputed values (Supplemental Table 1). In these models (with imputed random values instead of the missings), we observed the AIAN and more than one race categories as risk factors for ECV failure. In Supplemental Table 1, we show also the multivariate models with significant interactions. According to this model, women younger than 25, older than 40, and African American women were protective factors. Additionally, fetal female sex, pre-pregnancy BMI, nulliparity, and low fetal weight centile were risk factors for failed ECV. According to this model, which considers interactions, the AIAN race was protective (OR 0.63 CI.95 0.37–1.07). In particular, considering in a non-imputed model only pre-pregnancy BMI and ethnicity, we found that in univariate analysis, AIAN was a significant risk factor for ECV failure (OR 1.15, CI 95 1.05–1.27, p = 0.002) but in multivariate analysis resulted in being an important protective factor (OR 0.49, CI 95 0.32–0.75, p = 0.001). The increased risk was significantly mediated by an interaction with pre-pregnancy BMI (interaction term pre-pregnancy BMI (km/m2):race AIAN OR 1.02, CI 95 1.01–1.04, p < 0.001). In Supplemental Table 1D, the same pattern of significant differences was observed. Supplemental Tables 1E–H show the models considering the BMI at the delivery time. The same differences were also observed in these models, and BMI at delivery was also a significant risk factor for ECV failure. Supplemental Tables 1G and H show the models with the interaction terms. In the model without imputation, where 61.99% of the BMI values were missings, female neonatal sex was a non-significant risk factor, and two significant interactions justified the increased risk of ECV failure associated with female fetal sex, found in the model without interaction terms. In particular, there was a significant interaction between young maternal age and female neonatal sex and between nulliparity and female neonatal sex (Supplemental Table 2C). However, in supplemental Table 2D with the imputation of missing values, fetal neonatal sex was a significant risk factor for ECV failure. Considering that the BMI at delivery missings were 61.99% of the assessed cohort, the models were evaluated with 1000 different random imputations, and the p values of the 1000 models with the interaction terms were constantly significant for the female fetal sex as a risk factor for failed ECV. Although not considered in the multivariate models, because of the high rate of missing values, weight gain during pregnancy was also associated with an increased ECV failure (OR 1.01, CI 95 1.01–1.01, p < 0.001) (Table 1). Considering the false discovery rate adjusted p-values, the same pattern of significant differences was observed (Supplemental Table 1).
The chart exhibits the multivariate logistic regression analysis (dependent variable failed ECV). The plot shows the adjusted odds ratio (aOR), its 95% confidence interval (CI 95), and p value. The reported effects size magnitude is based on Cohen’s effect size interpretation [51]
Discussion
Main findings
We found that 64.23% of the procedures were successful. Moreover, among the successful ECV procedures, the majority were delivered vaginally and only 23.66% delivered by CS. The prevalence of spontaneous vaginal delivery in successful ECV was 71.63%. Among the failed ECV procedures, 24.74% had a cephalic presentation at delivery, but 63.11% of these pregnancies were delivered by CS. Nulliparity, female sex, low fetal weight centile (10–49th centile and small for gestational age fetuses < 10th centile), high pre-pregnancy BMI, high BMI at delivery, and high weight gain during pregnancy were associated with an increased ECV failure. African American, AIAN race categories, younger maternal age (< 25 years), and older maternal age (> 40 years) were protective factors against ECV failure.
Results in the context of what is known
Implementing ECV procedures seems feasible and, in case of a successful procedure, brings high chances of delivering spontaneously and vaginally. Hence reducing the burden related to the high rate of CS in breech presenting fetuses at delivery [2,3,4,5]. In the previous literature, the prevalence of CS after a successful ECV was 18.71% (CI 95 13.01–26.16%), which is similar to the prevalence found in our study 23.66% [25]. Moreover, women with a failed procedure can be reassured that in 8.48% of the cases, fetuses are presenting anyway cephalic, and the delivery can be a vaginal spontaneous cephalic delivery (Fig. 2A and B). Recently Birene and coworkers found that performing ECV did not reduce the number of CS [26]. However, in their control group, 33% of pregnancies had breech vaginal delivery [26]. Our study found that vaginal breech delivery is performed only in 1.06% after the failed ECV procedures and in 0.42% after the successful procedures.
The reasons that can lead to ECV failure can be multiple. In addition to the mechanical difficulty during the ECV maneuvers, other reasons related to ECV failure are the spontaneous repositioning of the fetus in the breech position and the occurrence of complications (e.g., placental abruption, vaginal bleeding, or abnormal cardiotocography) [27]. Although the overall prevalence of serious complications is low, abnormal fetal heartbeat, including bradycardia, are among the most frequent reasons to discontinue the procedure [27].
Nulliparity
In the previous literature, parity was consistently associated with ECV failure or success, and in particular, nulliparity was associated with ECV failure [13,14,15,16, 25, 28,29,30,31,32]. Our data confirm the strong association between nulliparity and ECV procedure failure. Probably the difference in comparison to other risk factors, which showed varying outcomes, is the magnitude of the effect size of parity that allows a better classification of the association also with small sample sizes.
Female fetal sex
Even though many different explanations related to the sex differences between females and males are possible, there is no definitive answer. It is known that there are significant anthropometric differences between female and male fetuses. Female fetuses have significantly shorter crown-rump, crown-heel distances, and lower weight than males [33]. They also have a higher relative fat content in respect to the muscle mass [33]. This different ratio between muscle and fat masses can lead to different behaviors during the ECV maneuvers, and it can explain the differences in the ECV success rate. Even, López-Pérez and coworkers found an increased rate of ECV failures associated with female neonatal sex [16].
Fetal weight
Previous studies found a higher prevalence of fetal weight < 2500 g and lower birth weight or estimated fetal weight in the groups of failed ECV procedures [14, 30]. We found a significant correlation between low fetal weight centile (10-49th centile and small for gestational age fetuses < 10th centile) and ECV procedure failure confirming that a low fetal weight can impair the procedure’s success. We also found an increased ECV success rate in fetal weight > 90th centile compared to the reference category. However, this difference was mainly non-significant after adjusting for the false discovery rate test in the models with the interactions. The only significant difference was found in the model considering the BMI at delivery and comprising the imputed values. However, further evidence is required because of this model’s high number of imputed BMI values.
Maternal anthropometric characteristics
This study confirms high maternal BMI is a factor that impedes the ECV success [13, 15, 17, 18, 34]. In previous literature, there was a broad heterogeneity in the way to consider maternal anthropometric features in the prediction of ECV success [15]. Also, the results were heterogeneous [6]. Some authors found that maternal weight was not a significant predictor of ECV failure or success [16, 30, 31, 35,36,37]. Meanwhile, two recent studies found that an increased pre-pregnancy BMI was a significant risk factor for ECV failure [17, 34]. Moreover, the high maternal BMI at the time of the procedure was also found to be a significant predictor of ECV failure [34]. To our knowledge, only limited data are presented in previous literature about the association between weight gain during pregnancy and ECV failure. López-Pérez and coworkers showed a non-significant higher weight gain among the group of pregnancies with a successful ECV procedure. Otherwise, we found an increased risk of failure associated with high weight gain during pregnancy. However, this study can not be conclusive because of this factor’s high number of missings. Furthermore, the BMI at delivery or at the time of the procedure partly also considers the effect of weight gain during pregnancy, and our results are in accordance with the findings of Dong and coworkers [34].
Maternal race
We also found that the African American race to influence the ECV success positively. Previous studies hypothesized that the increased chance of ECV success in the African American race was because presenting part usually remains high until the onset of labor [18, 19]. Hence, the high presenting part facilitates the ECV maneuvers. Also, AIAN was found to be a favoring factor for successful ECV. The AIAN group is also known to have a lower incidence of CS than the other race strata [38].
Maternal age
The previous literature found no significant correlation between maternal age and ECV success or failure. It was only found that older maternal age is a significant risk factor for breech presentation [39]. Furthermore, Dong and coworkers found that mothers younger than 35 significantly correlated to a vaginal delivery after a successful ECV procedure [34]. Our study found a U-shape association between maternal age and ECV success. In particular, younger maternal age (< 25 years) and older maternal age (> 40 years) were associated with an increased rate of ECV success. The increased success rate in younger women can be related to the reduction of soft tissue elasticity correlated with older women’s age [40]. It is also known that the increased rigidity of the abdominal and uterine wall was found to be a risk factor for ECV failure [15, 41]. However, this cannot explain the reduced risk in women older than 40. In this case, we believe that a possible explanation can be a more stringent selection of candidates for the procedure in this age group. In addition, there are many significant interaction factors with maternal age. For example, a mother younger than 25 years carrying a female fetus will have an odds of 0.81 instead of 0.65 (Supplemental Table 2). Furthermore, a mother younger than 25 years and nulliparous will have an odds of 1.10 instead of 0.65 (Supplemental Table 2).
Strengths and weaknesses
The main strength of this study is the vast observational cohort analyzed. However, several potential limitations should be discussed. The retrospective nature of this study limits the value of the results because of the possible inherited biases from the data collection planning and procedures.
The absence of information about methodological issues of the procedures limits the study. It was impossible to correct for some factors known to be correlated to the procedure’s success. Gestational age at the procedure was not available, and it was previously found to be a significant predictive factor for the procedure success [34]. However, in the US, it is suggested for the recruitment and procedure timing the gestational period between 36 and 37 weeks and 6 days [6]. This period is within a relatively small timeframe and is associated with a high probability of procedure success [34]. Another critical issue is the unavailable information about the operator’s experience that is known to favor the procedure’s success [42]. Repeated procedures were not recorded. The practice of repeating procedures can further reduce the number of breech fetuses at delivery [43]. However, due to the broad cohort included and the nationwide nature of the study, it is unlikely that these features will influence the actual results of our analysis. Another missing information in the original dataset was the time interval between the ECV procedure and the delivery. However, it is unlikely that this factor will alter the results of our analysis as in previous literature, was found no significant association between the time-interval and the mode of delivery [44].
In addition to information on the methodology of the technique, other information was missing. The estimated fetal weight at the time of the procedure was not recorded. As this is an essential factor in predicting the procedure failure or success, we assumed that the same weight centile registered at birth was effectively the weight centile at the procedure time. In addition, according to this assumption, we used a prenatal growth curve to assess fetal centiles. In all our analyses, including the multivariate models, we considered the fetal/newborn weight centile. Also, the estimated amount of amniotic fluid was not recorded in the original dataset. Previous studies indicate an association between amniotic fluid quantity and successful ECV [13, 14, 16, 18, 34,35,36, 45, 46], whereas additional studies do not [30, 37, 47]. Due to the heterogeneity of the literature about this issue and the possible errors related to the ultrasound estimation of the amniotic fluid quantity, it is unlikely that this information would have changed the results of our analysis. Even the placenta location was not present in the analyzed dataset. Some previous reports showed an association between successful ECV and placenta location [13, 19, 30, 32, 34], but others failed to see an association [16, 35,36,37, 47]. Other factors were not present in the original dataset, such as the characteristics of the maternal abdominal and uterine wall [15, 41, 48], the persistence of breech presentation [49], the type of presentation at the time of ECV maneuver [13, 18, 47], and the engagement of the fetal presenting part into the pelvis [15, 16, 18, 50]. Moreover, the association between ECV failure and female fetal sex reflects a small effect size. Generally, a Cohen h effect size is defined as small as 0.20; in this case, the observed effect size of female fetal sex is 0.06 [51]. Previous literature developed different predictive instruments for ECV failure or success with an accuracy ranging from 70 to 80% [16, 34]. However, these models were developed using small cohorts underpowered to identify potential predictors with a small effect size that can improve the model’s discrimination capacity. The advantage of this cohort is to better depict the effect size of some parameters such as BMI, maternal weight gain during pregnancy, or fetal sex, which can be useful to plan future studies to develop accurate prediction algorithms for ECV success or failure.
Generalisability, relevance of the findings, and unanswered questions
Accurate knowledge of the factors involved in the ECV procedure and subsequent delivery is fundamental in making appropriate counseling for women who access this service. Moreover, these results increase the knowledge about the factors involved in ECV success or failure. Greater knowledge means being able to face this path in the best and most conscious way, which can reduce the number of cesarean sections, thus reducing the subsequent morbidity related to them.
Conclusions
This study found a high prevalence of successful ECV procedures and spontaneous vaginal delivery after successful ECV procedures. Furthermore, the present results found nulliparity, female fetal sex, low fetal weight centile, high pre-pregnancy BMI, high BMI at delivery, and increased maternal weight gain during pregnancy negatively correlated to ECV success. African American women and AIAN race categories were significant protective factors against ECV failure. Maternal age presented a U-shape risk profile, with younger and older maternal ages found to be significant protective factors against ECV failure. These findings can potentially improve the knowledge about the factors involved in ECV, allowing more informed counseling to the women undergoing this procedure.
Availability of data and materials
All datasets are freely available at https://www.cdc.gov/nchs/data_access/Vitalstatsonline.htm.
Abbreviations
- AIAN:
-
American Indian and Alaska Native
- BMI:
-
Body mass index
- CI.95:
-
95% Confidence interval
- CS:
-
Cesarean section
- ECV:
-
External cephalic version
- IQR:
-
Interquartile range
- NHOPI:
-
Native Hawaiian and Other Pacific Islanders
- OR:
-
Odds ratio
- STROBE:
-
Strengthening the Reporting of Observational Studies in Epidemiology
- US:
-
United States
References
Weiniger CF, Lyell DJ, Tsen LC, Butwick AJ, Shachar B, Callaghan WM et al (2016) Maternal outcomes of term breech presentation delivery: impact of successful external cephalic version in a nationwide sample of delivery admissions in the United States. BMC Pregnancy Childbirth 16:150
Macharey G, Toijonen A, Hinnenberg P, Gissler M, Heinonen S, Ziller V (2020) Term cesarean breech delivery in the first pregnancy is associated with an increased risk for maternal and neonatal morbidity in the subsequent delivery: a national cohort study. Arch Gynecol Obstet 302:85–91
Hannah ME, Hannah WJ, Hewson SA, Hodnett ED, Saigal S, Willan AR (2000) Planned caesarean section versus planned vaginal birth for breech presentation at term: a randomised multicentre trial. Term Breech Trial Collaborative Group. Lancet 356:1375–1383
Hinkson L, Schauer M, Latartara E, Alonso-Espias M, Rossetti E, Gebert P et al (2021) The Charité external cephalic version for leading twin breech without regional anesthesia and tocolysis. A prospective study on feasibility, sonographic assessment and outcomes. Eur J Obstet Gynecol Reprod Biol 268:62–67
ACOG Committee on Obstetric Practice (2018) ACOG Committee Opinion No 745: mode of term singleton breech delivery. Obstet Gynecol 132:e60–e63
ACOG (2020) External Cephalic Version: ACOG Practice Bulletin, Number 221. Obstet Gynecol 135:e203–e212
RCGOG (2017) External cephalic version and reducing the incidence of term breech presentation: green-top guideline No 20a. BJOG 124:e178–e192
Schauer M, Latartara E, Alonso-Espias M, Rossetti E, Gebert P, Henrich W et al (2022) Depression, anxiety and stress in women with breech pregnancy compared to women with cephalic presentation-a cross-sectional study. Arch Gynecol Obstet
Toijonen AE, Heinonen ST, Gissler MVM, Macharey G (2020) A comparison of risk factors for breech presentation in preterm and term labor: a nationwide, population-based case-control study. Arch Gynecol Obstet 301:393–403
Martin JA, Hamilton BE, Osterman MJK, Driscoll AK (2021) Births: final data for 2019. Natl Vital Stat Rep 70:1–51
Soneji S, Beltrán-Sánchez H (2019) Association of maternal cigarette smoking and smoking cessation with preterm birth. JAMA Netw Open 2:e192514
Hadlock FP, Harrist RB, Martinez-Poyer J (1991) In utero analysis of fetal growth: a sonographic weight standard. Radiology 181:129–133
Ben-Meir A, Erez Y, Sela HY, Shveiky D, Tsafrir A, Ezra Y (2008) Prognostic parameters for successful external cephalic version. J Matern Fetal Neonatal Med 21:660–662
Ebner F, Friedl TWP, Leinert E, Schramm A, Reister F, Lato K et al (2016) Predictors for a successful external cephalic version: a single centre experience. Arch Gynecol Obstet 293:749–755
Kok M, Cnossen J, Gravendeel L, van der Post J, Opmeer B, Mol BW (2008) Clinical factors to predict the outcome of external cephalic version: a metaanalysis. Am J Obstet Gynecol 199(630):e1-7 (discussion e1–5)
López-Pérez R, Lorente-Fernández M, Velasco-Martínez M, Martínez-Cendán JP (2020) Prediction model of success for external cephalic version. Complications and perinatal outcomes after a successful version. J Obstet Gynaecol Res 46:2002–2009
Chaudhary S, Contag S, Yao R (2019) The impact of maternal body mass index on external cephalic version success. J Matern Fetal Neonatal Med 32:2159–2165
Fortunato SJ, Mercer LJ, Guzick DS (1988) External cephalic version with tocolysis: factors associated with success. Obstet Gynecol 72:59–62
Hofmeyr GJ, Sadan O, Myer IG, Galal KC, Simko G (1986) External cephalic version and spontaneous version rates: ethnic and other determinants. Br J Obstet Gynaecol 93:13–16
Velzel J, de Hundt M, Mulder FM, Molkenboer JFM, Van der Post JAM, Mol BW et al (2015) Prediction models for successful external cephalic version: a systematic review. Eur J Obstet Gynecol Reprod Biol 195:160–167
Burgos J, Melchor JC, Pijoán JI, Cobos P, Fernández-Llebrez L, Martínez-Astorquiza T (2011) A prospective study of the factors associated with the success rate of external cephalic version for breech presentation at term. Int J Gynaecol Obstet 112:48–51
Kok M, Cnossen J, Gravendeel L, Van Der Post JA, Mol BW (2009) Ultrasound factors to predict the outcome of external cephalic version: a meta-analysis. Ultrasound Obstet Gynecol 33:76–84
Dahl CM, Zhang Y, Ong JX, Yeh C, Son M, Miller ES et al (2021) A multivariable predictive model for success of external cephalic version. Obstet Gynecol 138:426–433
R Core Team (2022) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, p 2022
Chan LYS, Leung TY, Fok WY, Chan LW, Lau TK (2002) High incidence of obstetric interventions after successful external cephalic version. BJOG 109:627–631
Birene B, Ishaque U, Chrusciel J, Bonneau S, Gabriel R, Graesslin O (2021) Influence of the external cephalic version attempt on the Cesarean section rate: experience of a type 3 maternity hospital in France. Arch Gynecol Obstet 303:443–454
Grootscholten K, Kok M, Oei SG, Mol BWJ, van der Post JA (2008) External cephalic version-related risks: a meta-analysis. Obstet Gynecol 112:1143–1151
Salzer L, Nagar R, Melamed N, Wiznitzer A, Peled Y, Yogev Y (2015) Predictors of successful external cephalic version and assessment of success for vaginal delivery. J Matern Fetal Neonatal Med 28:49–54
Mowat A, Gardener G (2014) Predictors of successful external cephalic version in an Australian maternity hospital. Aust N Z J Obstet Gynaecol 54:59–63
Newman RB, Peacock BS, VanDorsten JP, Hunt HH (1993) Predicting success of external cephalic version. Am J Obstet Gynecol 169:245–249 (discussion 249–250)
Tan GW, Jen SW, Tan SL, Salmon YM (1989) A prospective randomised controlled trial of external cephalic version comparing two methods of uterine tocolysis with a non-tocolysis group. Singapore Med J 30:155–158
Brocks V, Philipsen T, Secher NJ (1984) A randomized trial of external cephalic version with tocolysis in late pregnancy. Br J Obstet Gynaecol 91:653–656
Palti H, Adler B (1975) Anthropometric measurements of the newborn, sex differences, and correlations between measurements. Hum Biol 47:523–530
Dong T, Chen X, Zhao B, Jiang Y, Chen Y, Lv M et al (2022) Development of prediction models for successful external cephalic version and delivery outcome. Arch Gynecol Obstet 305:63–75
Hellström AC, Nilsson B, Stånge L, Nylund L (1990) When does external cephalic version succeed? Acta Obstet Gynecol Scand 69:281–285
Shalev E, Battino S, Giladi Y, Edelstein S (1993) External cephalic version at term–using tocolysis. Acta Obstet Gynecol Scand 72:455–457
Hutton EK, Saunders CA, Tu M, Stoll K, Berkowitz J (2008) Early External Cephalic Version Trial Collaborators Group n. Factors associated with a successful external cephalic version in the early ECV trial. J Obstet Gynaecol Can 30:23–28
Valdes EG (2021) Examining Cesarean delivery rates by race: a population-based analysis using the Robson ten-group classification system. J Racial Ethn Health Disparities 8:844–851
Fruscalzo A, Londero AP, Salvador S, Bertozzi S, Biasioli A, Della Martina M et al (2014) New and old predictive factors for breech presentation: our experience in 14,433 singleton pregnancies and a literature review. J Matern Fetal Neonatal Med 27:167–172
Radivojevic K, Rudelstorfer R (1988) The older primipara: labor induction and rigidity of the soft tissues. Geburtshilfe Frauenheilkd 48:246–248
Westgren M, Edvall H, Nordström L, Svalenius E, Ranstam J (1985) Spontaneous cephalic version of breech presentation in the last trimester. Br J Obstet Gynaecol 92:19–22
Thissen D, Swinkels P, Dullemond RC, van der Steeg JW (2019) Introduction of a dedicated team increases the success rate of external cephalic version: a prospective cohort study. Eur J Obstet Gynecol Reprod Biol 236:193–197
Reicher L, Lavie A, Fouks Y, Isakov O, Attali E, Yogev Y et al (2021) Efficacy of a second external cephalic version (ECV) after a successful first external cephalic version with subsequent spontaneous reinversion to breech presentation: a retrospective cohort study. Arch Gynecol Obstet 303:911–916
Cahan T, Levin G, Moran O, Weill Y, Pollack R, Meyer R (2020) Successful vaginal delivery after external cephalic version (ECV): does time interval from ECV to delivery make a difference? A multicenter study. Arch Gynecol Obstet 302:1361–1367
Healey M, Porter R, Galimberti A (1997) Introducing external cephalic version at 36 weeks or more in a district general hospital: a review and an audit. Br J Obstet Gynaecol 104:1073–1079
Boucher M, Bujold E, Marquette GP, Vezina Y (2003) The relationship between amniotic fluid index and successful external cephalic version: a 14-year experience. Am J Obstet Gynecol 189:751–754
Donald WL, Barton JJ (1990) Ultrasonography and external cephalic version at term. Am J Obstet Gynecol 162:1542–1545 (discussion 1545–1547)
Buhimschi CS, Buhimschi IA, Wehrum MJ, Molaskey-Jones S, Sfakianaki AK, Pettker CM et al (2011) Ultrasonographic evaluation of myometrial thickness and prediction of a successful external cephalic version. Obstet Gynecol 118:913–920
Lauterbach R, Bachar G, Ben-David C, Matanes E, Ginsberg Y, Beloosesky R et al (2021) Association of persistent breech presentation with external cephalic version success. Obstet Gynecol 137:258–262
Wise MR, Sadler L, Ansell D (2008) Successful but limited use of external cephalic version in Auckland. Aust N Z J Obstet Gynaecol 48:467–472
Cohen J (1988) Statistical power analysis for the behavioral sciences, 2nd edn. L. Erlbaum Associates, Hillsdale
Acknowledgements
The authors would like to thank the whole staff collaborating in article collection, selection, reading and in paper writing and reviewing.
Funding
Open access funding provided by Università degli Studi di Genova within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Contributions
Substantial contributions to conception and design or acquisition of data or to analysis and interpretation of data (APL, AX, CM, AF, AC). Drafting the article or revising it critically for important intellectual content (APL, AX, CM, AF, AC).All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no potential conflicts of interest relevant to this article. This study has had no financial support.
Ethical approval and consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Londero, A.P., Xholli, A., Massarotti, C. et al. Factors influencing the effect of external cephalic version: a retrospective nationwide cohort analysis. Arch Gynecol Obstet 308, 1127–1137 (2023). https://doi.org/10.1007/s00404-022-06763-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-022-06763-2