Abstract
Objective
To investigate the influence of two types of tumor-associated macrophages (TAMs) on the biological function of human ovarian cell lines in vitro.
Methods
(1) M2 macrophage release was induced by IL-4, and M1 macrophage release by phorbol myristate acetate (PMA) in vitro. Flow cytometry was used to distinguish these two types; (2) transwell culture system was used to establish a non-contact co-culture model of macrophage and ovarian cancer cells (SKOV3, HEY, HO8910 and A2780) in vitro. The microenvironment of ovarian cancer was simulated in vitro. (3) The proliferation, apoptosis, migration and invasion of ovarian cancer cells SKOV3, HEY, HO8910 and A2780 were analyzed after co-culture. Their proliferation was detected by CCK8 method, apoptosis by flow cytometry, Annexin V-FITC/PI double staining, invasion by Transwell assay, and migration by wound healing test.
Results
(1) IL-4-induced macrophages (M2) overexpressed CD163, and PMA-induced macrophages (M1) overexpressed HLA-DR. After co-culturing primary macrophages with ovarian cancer cells (SKOV3, HEY, HO8910, A2780), macrophage CD163 was highly expressed. (2) Proliferation and apoptosis of ovarian cancer cells (SKOV3, HEY, HO8910, A2780): the proliferation of ovarian cancer cells in M2 co-culture group increased compared to that in M1 co-culture group and primary co-culture group (p < 0.05); the apoptosis of ovarian cancer cells in M2 co-culture group decreased compared to that in M1 co-culture group and primary co-culture group (p < 0.05). (3) Migration and invasion of ovarian cancer cells (SKOV3, HEY, HO8910, A2780): the invasion of ovarian cancer cells in M2 co-culture group increased compared to that in M1 co-culture group and primary co-culture group (p < 0.05); the migration of ovarian cancer cells in M2 co-culture group increased compared to that in M1 co-culture group and the primary co-culture group (p < 0.05).
Conclusion
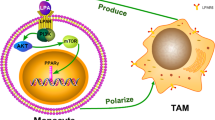
In the simulated in vitro tumor microenvironment, co-cultured ovarian cancer cells polarized macrophages to the M2 phenotype. Furthermore, M2 macrophages enhanced the proliferation, invasion and migration, and inhibited the apoptosis of ovarian cancer cells.





Similar content being viewed by others
References
Mantovani A (2009) Cancer: inflaming metastasis. Nature 457(7225):36–37
Qian BZ, Pollard JW (2010) Macrophage diversity enhances tumor progression and metastasis. Cell 141(1):39–51
Forssell J, Oberg A, Henriksson ML, Stenling R, Jung A, Palmqvist R (2007) High macrophage infiltration along the tumor front correlates with improved survival in colon cancer. Clin Cancer Res 13(5):1472–1479
Wan T, Liu JH, Zheng LM, Cai MY, Ding T (2009) Prognostic significance of tumor-associated macrophage infiltration in advanced epithelial ovarian carcinoma. Ai Zheng 28(3):323–327
Shah CA, Allison KH, Garcia RL, Gray HJ, Goff BA, Swisher EM (2008) Intratumoral T cells, tumor-associated macrophages, and regulatory T cells: association with p53 mutations, circulating tumor DNA and survival in women with ovarian cancer. Gynecol Oncol 109(2):215–219
Tanaka Y, Kobayashi H, Suzuki M, Kanayama N, Suzuki M, Terao T (2004) Upregulation of bikunin in tumor-infiltrating macrophages as a factor of favorable prognosis in ovarian cancer. Gynecol Oncol 94(3):725–734
Bonecchi R, Locati M, Mantovani A (2011) Chemokines and cancer: a fatal attraction. Cancer Cell 19(4):434–435
Mosser DM (2003) The many faces of macrophage activation. J Leu-koc Biol 73:209–212
Hagemann T, Wilson J (2006) Ovarian cancer cells polarize macrophages toward a tumor-associated phenotype. J Immunol 176:5023–5032
Mnatovani A, Schiopa T, Porta C, Allavena P, Sica A (2006) Role of tumor-associated macrophages in tumor progression and invasion. Cancer Metastasis Rev 25:315–322
Balkwill F (2004) Cancer and the chemokine network. Nat Rev Cancer 4:540–550
Pollard JW (2004) Tumor-educated macrophages promote tumor progression and metastasis. Nat Rev Cancer 4:71–78
Condeelis J, Pollard JW (2006) Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell 124(2):263–266
Ryder M, Ghossein RA, Ricarte-Filho JC, Knauf JA, Fagin JA (2008) Increased density of tumor-associated macrophages is associated with decreased survival in advanced thyroid cancer. Endocr Relat Cancer 15:1069–1074
Zhu XD, Zhang JB, Zhuang PY, Zhu HG, Zhang W, Xiong YQ, Wu WZ, Wang L, Tang ZY, Sun HC (2008) High expression of macrophage colony-stimulating factor in peritumoral liver tissue is associated with poor survival after curative resection of hepatocellular carcinoma. J Clin Oncol 26(16):2707–2717
Zabuawala T, Taffany DA, Sharma SM, Merchant A, Adair B, Srinivasan R, Rosol TJ, Fernandez S, Huang K, Leone G, Ostrowski MC (2010) An ets2-driven transcriptional program in tumor-associated macrophages promotes tumor metastasis. Cancer Res 70(4):1323–1333
Ojalvo LS, Whittaker CA, Condeelis JS, Pollard JW (2010) Gene expression analysis of macrophages that facilitate tumor invasion supports a role for Wnt-signaling in mediating their activity in primary mammary tumors. J. Immunol 184(2):702–712
Sica A, Schioppa T, Mantovani A, Allavena P (2006) Tumor-associated macrophages are a distinct M2 polarised population promoting tumor progression: potential targets of anti-cancer therapy. Eur. J. Cancer 42(6):717–727
An H, Rim HK, Chung HS, Choi IY, Kim NH, Kim SJ, Moon PD, Myung NY, Jeong HJ, Jeong CH, Chung SH, Um JY, Hong SH, Kim HM (2009) Expression of inducible nitric oxide synthase by corydalis turtschaninovii on interferon-gamma stimulated macrophages. J Ethnopharmacol 122(3):573–578
Ma J, Liu L, Che G, Yu N, Dai F, You Z (2010) The M1 form of tumor-associated macrophages in non-small cell lung cancer is positively associated with survival time. BMC Cancer 10:112–116
Niino D, Komohara Y, Murayama T, Aoki R, Kimura Y, Hashikawa K, Kiyasu J, Takeuchi M, Suefuji N, Sugita Y, Takeya M, Ohshima K (2010) Ratio of M2 macrophage expression is closely associated with poor prognosis for Angioimmunoblastic T-cell lymphoma(AITL). Pathol Int 60(4):278–283
Acknowledgements
Yi Jiang was supported by the National Natural Science Foundation of China (No. 81702566) and Wenjun Cheng was supported by the National Natural Science Foundation of China (No. 81872119).
Author information
Authors and Affiliations
Contributions
Yi Jiang is responsible for subject experiment and article writing. Lin Yuan is responsible for part of the subject experiment, such as cell proliferation and apoptosis. Yicong Wan is responsible for part of the article writing. Chengyan Luo is responsible for cell co-culture. Shulin Zhou is responsible for part of the subject experiment, such as transwell assay. Wenjun Cheng is responsible for Subject guidance.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that there is no conflict of interest.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors.
Informed consent statement
The informed consent statement is not applicable to this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yi, J., Lin, Y., Yicong, W. et al. Effect of macrophages on biological function of ovarian cancer cells in tumor microenvironment in vitro. Arch Gynecol Obstet 302, 1009–1017 (2020). https://doi.org/10.1007/s00404-020-05719-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-020-05719-8