Abstract
Purpose
Implantation rates differ according to ovulation induction agents in ART. This study investigates the different local endometrial effects of LH- versus hCG-induced ovulation.
Methods
Endometrial stromal cells from healthy patients were cultured with hCG or LH in different concentrations, supplemented with 250 ng/mL hCG and progesterone after 2 and 5 days. In addition after decidualization induction, cells were treated with hCG (50 or 250 ng/mL) or LH (10 or 50 ng/mL) for 3 days. Receptivity markers expression was evaluated by real-time quantitative PCR on day 3 and 6.
Results
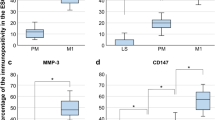
On day 3, non-decidualized cells treated with LH showed an increased expression of IGFBP1, IL-8 and CXCL12 compared to hCG. The expression pattern changed on day 6, where cells treated with hCG showed higher expression of implantation markers compared to LH-treated cells. Furthermore, on day 3, decidualized cells treated with hCG250 showed an increased IL8 and CXCL12 expression compared to LH10.
Conclusions
LH seems to modulate the local endometrial expression of receptivity markers earlier compared to hCG; however, the effect is not sustained over time in cells without prior decidualization. Though, in decidualized cells, pattern changed and an earlier positive effect of hCG was shown on IL-8 and CXCL12.






Similar content being viewed by others
Abbreviations
- IGFBP1:
-
Insulin-like growth factor binding protein 1
- IL8:
-
Interleukin 8
- CXCL1:
-
CXC-Motif-Chemokin ligand 1
- CXCL12:
-
CXC-Motif-Chemokin ligand 12, also stromal cell-derived factor 1 (SDF-1) or pre-B cell growth-stimulating factor (PBSF)
- LH:
-
Luteinizing hormone
- hCG:
-
Human chorionic gonadotropin
References
Bourdiec A, Shao R, Rao CV, Akoum A (2012) Human chorionic gonadotropin triggers angiogenesis via the modulation of endometrial stromal cell responsiveness to interleukin 1: a new possible mechanism underlying embryo implantation. Biol Reprod 87(3):66. https://doi.org/10.1095/biolreprod.112.100370
Horcajadas JA, Minguez P, Dopazo J, Esteban FJ, Dominguez F, Giudice LC, Pellicer A, Simon C (2008) Controlled ovarian stimulation induces a functional genomic delay of the endometrium with potential clinical implications. J Clin Endocrinol Metab 93(11):4500–4510. https://doi.org/10.1210/jc.2008-0588
Castro-Rendon WA, Castro-Alvarez JF, Guzman-Martinez C, Bueno-Sanchez JC (2006) Blastocyst-endometrium interaction: intertwining a cytokine network. Braz J Med Biol Res 39(11):1373–1385
Casarini L, Santi D, Simoni M, Poti F (2018) ‘Spare’ luteinizing hormone receptors: facts and fiction. Trends Endocrinol Metab 29(4):208–217. https://doi.org/10.1016/j.tem.2018.01.007
Theofanakis C, Drakakis P, Besharat A, Loutradis D (2017) Human chorionic gonadotropin: the pregnancy hormone and more. Int J Mol Sci. https://doi.org/10.3390/ijms18051059
Nwabuobi C, Arlier S, Schatz F, Guzeloglu-Kayisli O, Lockwood CJ, Kayisli UA (2017) hCG: biological functions and clinical applications. Int J Mol Sci. https://doi.org/10.3390/ijms18102037
Horcajadas JA, Riesewijk A, Polman J, van Os R, Pellicer A, Mosselman S, Simon C (2005) Effect of controlled ovarian hyperstimulation in IVF on endometrial gene expression profiles. Mol Hum Reprod 11(3):195–205. https://doi.org/10.1093/molehr/gah150
Zarei A, Parsanezhad ME, Younesi M, Alborzi S, Zolghadri J, Samsami A, Amooee S, Aramesh S (2014) Intrauterine administration of recombinant human chorionic gonadotropin before embryo transfer on outcome of in vitro fertilization/intracytoplasmic sperm injection: a randomized clinical trial. Iran J Reprod Med 12(1):1–6
Zygmunt M, Herr F, Keller-Schoenwetter S, Kunzi-Rapp K, Munstedt K, Rao CV, Lang U, Preissner KT (2002) Characterization of human chorionic gonadotropin as a novel angiogenic factor. J Clin Endocrinol Metab 87(11):5290–5296. https://doi.org/10.1210/jc.2002-020642
Berndt S, Blacher S, Perrier d’Hauterive S, Thiry M, Tsampalas M, Cruz A, Pequeux C, Lorquet S, Munaut C, Noel A, Foidart JM (2009) Chorionic gonadotropin stimulation of angiogenesis and pericyte recruitment. J Clin Endocrinol Metab 94(11):4567–4574. https://doi.org/10.1210/jc.2009-0443
Aflatoonian A, Yousefnejad F, Eftekhar M, Mohammadian F (2012) Efficacy of low-dose hCG in late follicular phase in controlled ovarian stimulation using GnRH agonist protocol. Arch Gynecol Obstet 286(3):771–775. https://doi.org/10.1007/s00404-012-2337-z
Berkkanoglu M, Isikoglu M, Aydin D, Ozgur K (2007) Clinical effects of ovulation induction with recombinant follicle-stimulating hormone supplemented with recombinant luteinizing hormone or low-dose recombinant human chorionic gonadotropin in the midfollicular phase in microdose cycles in poor responders. Fertil Steril 88(3):665–669. https://doi.org/10.1016/j.fertnstert.2006.11.150
Hershko Klement A, Shulman A (2017) hCG triggering in ART: an evolutionary concept. Int J Mol Sci. https://doi.org/10.3390/ijms18051075
Sahin S, Ozay A, Ergin E, Turkgeldi L, Kurum E, Ozornek H (2015) The risk of ectopic pregnancy following GnRH agonist triggering compared with hCG triggering in GnRH antagonist IVF cycles. Arch Gynecol Obstet 291(1):185–191. https://doi.org/10.1007/s00404-014-3399-x
Atkinson P, Koch J, Susic D, Ledger WL (2014) GnRH agonist triggers and their use in assisted reproductive technology: the past, the present and the future. Womens Health 10(3):267–276. https://doi.org/10.2217/whe.14.14
Craciunas L, Tsampras N, Raine-Fenning N, Coomarasamy A (2018) Intrauterine administration of human chorionic gonadotropin (hCG) for subfertile women undergoing assisted reproduction. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.cd011537.pub3
Maslova MA, Smol’nikova VY, Savilova AM, Burmenskaya OV, Bystritskii AA, Tabolova VK, Korneeva IE, Demura TA, Donnikov AE (2015) Profiles of mRNA expression for genes involved in implantation, early and middle phases of secretion stage in human endometrium. Bull Exp Biol Med 158(6):781–784. https://doi.org/10.1007/s10517-015-2861-5
Kao LC, Tulac S, Lobo S, Imani B, Yang JP, Germeyer A, Osteen K, Taylor RN, Lessey BA, Giudice LC (2002) Global gene profiling in human endometrium during the window of implantation. Endocrinology 143(6):2119–2138. https://doi.org/10.1210/endo.143.6.8885
Riesewijk A, Martin J, van Os R, Horcajadas JA, Polman J, Pellicer A, Mosselman S, Simon C (2003) Gene expression profiling of human endometrial receptivity on days LH + 2 versus LH + 7 by microarray technology. Mol Hum Reprod 9(5):253–264
Rinaldi L, Lisi F, Selman H (2014) Mild/minimal stimulation protocol for ovarian stimulation of patients at high risk of developing ovarian hyperstimulation syndrome. J Endocrinol Invest 37(1):65–70. https://doi.org/10.1007/s40618-013-0021-1
Choi J, Smitz J (2014) Luteinizing hormone and human chorionic gonadotropin: origins of difference. Mol Cell Endocrinol 383(1–2):203–213. https://doi.org/10.1016/j.mce.2013.12.009
Wirleitner B, Schuff M, Vanderzwalmen P, Stecher A, Okhowat J, Hradecky L, Kohoutek T, Kralickova M, Spitzer D, Zech NH (2015) Intrauterine administration of human chorionic gonadotropin does not improve pregnancy and life birth rates independently of blastocyst quality: a randomised prospective study. Reprod Biol Endocrinol 13:70. https://doi.org/10.1186/s12958-015-0069-1
Eftekhar M, Mojtahedi MF, Miraj S, Omid M (2017) Final follicular maturation by administration of GnRH agonist plus HCG versus HCG in normal responders in ART cycles: an RCT. Int J Reprod Biomed 15(7):429–434
Ganeff C, Chatel G, Munaut C, Frankenne F, Foidart JM, Winkler R (2009) The IGF system in in vitro human decidualization. Mol Hum Reprod 15(1):27–38. https://doi.org/10.1093/molehr/gan073
Germeyer A, Jauckus J, Zorn M, Toth B, Capp E, Strowitzki T (2011) Metformin modulates IL-8, IL-1beta, ICAM and IGFBP-1 expression in human endometrial stromal cells. Reprod Biomed Online 22(4):327–334. https://doi.org/10.1016/j.rbmo.2010.11.006
Karpovich N, Klemmt P, Hwang JH, McVeigh JE, Heath JK, Barlow DH, Mardon HJ (2005) The production of interleukin-11 and decidualization are compromised in endometrial stromal cells derived from patients with infertility. J Clin Endocrinol Metab 90(3):1607–1612. https://doi.org/10.1210/jc.2004-0868
Lathi RB, Hess AP, Tulac S, Nayak NR, Conti M, Giudice LC (2005) Dose-dependent insulin regulation of insulin-like growth factor binding protein-1 in human endometrial stromal cells is mediated by distinct signaling pathways. J Clin Endocrinol Metab 90(3):1599–1606. https://doi.org/10.1210/jc.2004-1676
Damewood MD, Shen W, Zacur HA, Schlaff WD, Rock JA, Wallach EE (1989) Disappearance of exogenously administered human chorionic gonadotropin. Fertil Steril 52(3):398–400
Humaidan P, Van Vaerenbergh I, Bourgain C, Alsbjerg B, Blockeel C, Schuit F, Van Lommel L, Devroey P, Fatemi H (2012) Endometrial gene expression in the early luteal phase is impacted by mode of triggering final oocyte maturation in recFSH stimulated and GnRH antagonist co-treated IVF cycles. Hum Reprod 27(11):3259–3272. https://doi.org/10.1093/humrep/des279
Licht P, von Wolff M, Berkholz A, Wildt L (2003) Evidence for cycle-dependent expression of full-length human chorionic gonadotropin/luteinizing hormone receptor mRNA in human endometrium and decidua. Fertil Steril 79(Suppl 1):718–723
Dekel N, Gnainsky Y, Granot I, Mor G (2010) Inflammation and implantation. Am J Reprod Immunol 63(1):17–21. https://doi.org/10.1111/j.1600-0897.2009.00792.x
Gnainsky Y, Granot I, Aldo PB, Barash A, Or Y, Schechtman E, Mor G, Dekel N (2010) Local injury of the endometrium induces an inflammatory response that promotes successful implantation. Fertil Steril 94(6):2030–2036. https://doi.org/10.1016/j.fertnstert.2010.02.022
Han VK, Bassett N, Walton J, Challis JR (1996) The expression of insulin-like growth factor (IGF) and IGF-binding protein (IGFBP) genes in the human placenta and membranes: evidence for IGF-IGFBP interactions at the feto-maternal interface. J Clin Endocrinol Metab 81(7):2680–2693. https://doi.org/10.1210/jcem.81.7.8675597
Szklarczyk D, Franceschini A, Wyder S, Forslund K, Heller D, Huerta-Cepas J, Simonovic M, Roth A, Santos A, Tsafou KP, Kuhn M, Bork P, Jensen LJ, von Mering C (2015) STRING v10: protein–protein interaction networks, integrated over the tree of life. Nucleic Acids Res 43(Database issue):D447–D452. https://doi.org/10.1093/nar/gku1003
Szklarczyk D, Morris JH, Cook H, Kuhn M, Wyder S, Simonovic M, Santos A, Doncheva NT, Roth A, Bork P, Jensen LJ, von Mering C (2017) The STRING database in 2017: quality-controlled protein-protein association networks, made broadly accessible. Nucleic Acids Res 45(D1):D362–D368. https://doi.org/10.1093/nar/gkw937
Zhang Q, Zhang PW, Cai YD (2016) The use of protein–protein interactions for the analysis of the associations between PM2.5 and some diseases. Biomed Res Int 2016:4895476. https://doi.org/10.1155/2016/4895476
Teh WT, McBain J, Rogers P (2016) What is the contribution of embryo-endometrial asynchrony to implantation failure? J Assist Reprod Genet 33(11):1419–1430. https://doi.org/10.1007/s10815-016-0773-6
Kol S, Humaidan P (2013) GnRH agonist triggering: recent developments. Reprod Biomed Online 26(3):226–230. https://doi.org/10.1016/j.rbmo.2012.11.002
Casarini L, Lispi M, Longobardi S, Milosa F, La Marca A, Tagliasacchi D, Pignatti E, Simoni M (2012) LH and hCG action on the same receptor results in quantitatively and qualitatively different intracellular signalling. PLoS ONE 7(10):e46682. https://doi.org/10.1371/journal.pone.0046682
Mostajeran F, Godazandeh F, Ahmadi SM, Movahedi M, Jabalamelian SA (2017) Effect of intrauterine injection of human chorionic gonadotropin before embryo transfer on pregnancy rate: a prospective randomized study. J Res Med Sci 22:6. https://doi.org/10.4103/1735-1995.199096
Craciunas L, Tsampras N, Coomarasamy A, Raine-Fenning N (2016) Intrauterine administration of human chorionic gonadotropin (hCG) for subfertile women undergoing assisted reproduction. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.cd011537.pub2
Youssef MA, Van der Veen F, Al-Inany HG, Mochtar MH, Griesinger G, Nagi Mohesen M, Aboulfoutouh I, van Wely M (2014) Gonadotropin-releasing hormone agonist versus HCG for oocyte triggering in antagonist-assisted reproductive technology. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.cd008046.pub4
Satyaswaroop PG, Bressler RS, de la Pena MM, Gurpide E (1979) Isolation and culture of human endometrial glands. J Clin Endocrinol Metab 48(4):639–641. https://doi.org/10.1210/jcem-48-4-639
Ujvari D, Jakson I, Babayeva S, Salamon D, Rethi B, Gidlof S, Hirschberg AL (2017) Dysregulation of in vitro decidualization of human endometrial stromal cells by insulin via transcriptional inhibition of forkhead box protein O1. PLoS ONE 12(1):e0171004. https://doi.org/10.1371/journal.pone.0171004
Acknowledgements
We acknowledge financial support by Ruprecht-Karls-Universität Heidelberg for open-access publishing.
Funding
This research was funded by Dres. Majic/Majic-Schlez-Stiftung, University of Heidelberg.
Author information
Authors and Affiliations
Contributions
Conceptualization, AF, AG, AMW; and JJ; methodology, AF, AG, JJ, AMW; formal analysis, AF, AG, EC, AMW; investigation, JJ, AMW, MZ, AF; resources, AG, AF, TS; data curation, JJ, AMW; writing—original draft preparation, AF, AMW; writing—review and editing, AF, AG, AMW, EC; visualization, AF, AMW; supervision, AG; project administration, AG; funding acquisition, AG, AF.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest regarding this paper.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. All patients provided written consent for the use of their tissue samples. The study was approved by the Ethics Committee of Ruprecht Karls University Heidelberg (S239/2005).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Freis, A., Germeyer, A., Jauckus, J. et al. Endometrial expression of receptivity markers subject to ovulation induction agents. Arch Gynecol Obstet 300, 1741–1750 (2019). https://doi.org/10.1007/s00404-019-05346-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-019-05346-y