Abstract
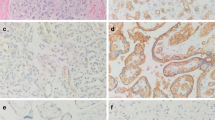
In previous studies, the expression and the role of proangiogenic factors in infantile hemangiomas have been well studied. However, the role of angiogenic inhibitors has been revealed rarely. The expression of PEDF, as the strongest and safe endogenous inhibitor, is still unrecognized until the current study. In order to investigate the expression and significance of the pigment epithelium-derived factor (PEDF) in the proliferating and regressing phases of infantile hemangiomas, the expression of PEDF, VEGF, Ki-67, and CD34 protein in hemangioma tissues was examined with immunohistochemical polymer HRP method in 42 cases during the proliferative phase, 40 cases during the regressing phase, and 11 cases of non-involuting congenital hemangiomas (NICHs). Meanwhile, the mRNA expression of these factors was detected with quantitative realtime RT-PCR. We found the protein and mRNA expression of PEDF in regressing phase was significantly higher than those in proliferative phase and NICHs (P < 0.001), while the protein and mRNA expression of VEGF were much lower (P < 0.001). The microvessel density (MVD), Ki-67 changes, and the expression of PEDF and VEGF were found significantly correlated. These results indicated that the reduction of VEGF and increase in PEDF are causative to the evolution of infantile hemangioma. PEDF may play a key role in the spontaneous regression of infantile hemangioma and may become an important potential therapeutic agent for infantile hemangioma.






Similar content being viewed by others
References
Akhavan A (2010) Current treatments for infantile hemangiomas. J Drugs Dermatol 9:176–180
Alcantara MB, Dass CR (2013) Regulation of MT1-MMP and MMP-2 by the serpin PEDF: a promising new target for metastatic cancer. Cell Physiol Biochem 31:487–494
Becerra SP (2006) Focus on molecules: pigment epithelium-derived factor (PEDF). Exp Eye Res 82:739–740
Belkacemi L, Zhang SX (2016) Anti-tumor effects of pigment epithelium—derived factor (PEDF): implication for cancer therapy. A mini-review. J Exp Clin Cancer Res 35(1):1–4
Boscolo E, Bischoff J (2009) Vasculogenesis in infantile hemangioma. Angiogenesis 12:197–207
Bruckner AL, Frieden IJ (2003) Hemangiomas of infancy. J Am Acad Dermatol 48:477–493
Cai J, Parr C, Watkins G et al (2006) Decreased pigment epithelium-derived factor expression in human breast cancer progression. Clin Cancer Res 12:3510–3517
Cheung LW, Au SC, Cheung AN et al (2006) Pigment epithelium-derived factor is estrogen sensitive and inhibits the growth of human ovarian cancer and ovarian surface epithelial cells. Endocrinology 147:4179–4191
Dawson D, Volpert O, Gillis W et al (1999) Pigment epithelium derived factor: a potent inhibitor of angiogenesis. Science 285:245–248
Demestre M, Terzi MY, Mautner V et al (2013) Effects of pigment epithelium derived factor (PEDF) on malignant peripheral nerve sheath tumours (MPNSTs). J Neurooncol 115:391–399
Drolet BA, Swanson EA, Frieden IJ et al (2008) Infantile hemangiomas: an emerging health issue linked to an increased rate of low birth weight infants. J Pediatr 153:712–715
Eivazi B, Ardelean M, Baumler W et al (2009) Update on haemangiomas and vascular malformations of the head and neck. Eur Arch Otorhinolaryngol 266:187–197
Ek ET, Dass CR, Choong PF (2006) Pigment epithelium-derived factor: a multimodal tumor inhibitor. Mol Cancer Ther 57:1641
Fernandez-Garcia NI, Volpert OV, Jimenez B (2007) Pigment epithelium-derived factor as a multifunctional antitumor factor. J Mol Med 85:15–22
Gaudreault J, Shiu V, Bricarello A et al (2005) Concomitant administration of bevacizumab, irinotecan, 5-fluorouracil, and leucovorin: nonclinical safety and pharmacokinetics. Int J Toxicol 24:357–363
Guan M, Yam HF, Su B et al (2003) Loss of pigment epithelium derived factor expression in glioma progression. J Clin Pathol 56:277–282
Guo T, Gu C, Li B (2012) PEDF inhibits growth and invasiveness of endometrial cancer cells in vitro. Panminerva Med 54(4):299–304
Halin S, Wikstrom P, Rudolfsson SH et al (2004) Decreased pigment epithelium-derived factor is associated with metastatic phenotype in human and rat prostate tumors. Cancer Res 64:5664–5671
Haurigot V, Villacampa P, Ribera A et al (2012) Long-term retinal PEDF overexpression prevents neovascularization in a murine adult model of retinopathy. PLoS One 7:e41511
Hoshina D, Abe R, Yamagishi SI et al (2010) The role of PEDF in tumor growth and metastasis. Curr Mol Med 10:292–295
Ide H, Yamagishi S, Lu Y (2015) Circulating pigment epithelium- derived factor (PEDF) is associated with pathological grade of prostate cancer. Anticancer Res 35(3):1703–1708
Janmohamed SR, Madern GC, de Laat PC (2015) Therapy of infantile haemangioma–history and current state. Eur J Pediatr 174(2):259–266
Jinnin M, Ishihara T, Boye E et al (2010) Recent progress in studies of infantile hemangioma. J Dermatol 37(4):283–298
Kubicka-Trzaska A, Wilanska J, Romanowska-Dixon B et al (2012) Circulating antiretinal antibodies predict the outcome of anti-VEGF therapy in patients with exudative age-related macular degeneration. Acta phthalmol 90:e21–e24
Léauté-Labrèze C, Dumas de la Roque E, Hubiche T et al (2008) Propranolol for severe hemangiomas of infancy. N Engl J Med. 358(24): 2649–2651
MacIsaac ZM, Nayar HS, Gehris R (2016) Treatment for infantile hemangiomas: selection criteria, safety, and outcomes using oral propranolol during the early phase of propranolol use for hemangiomas. J Craniofac Surg 27(1):159–162
Manley PW, Martiny-Baron G, Schlaeppi JM et al (2002) Therapies directed at vascular endothelial growth factor. Expert Opin Investig Drugs 11:1715–1736
Matsumoto K, Ishikawa H, Nishimura D et al (2004) Antiangiogenic property of pigment epithelium—derived factor in hepatocellular carcinoma. Hepatology 40:252–259
Mei LT, Peter FM, Crispin RD et al (2010) Anti-chondrosarcoma effects of PEDF mediated via molecules important to apoptosis, cell cycling, adhesion and invasion. Biochem Biophys Res Commun 398:613–618
Mori K, Gehtbach P, Yammnoto S et al (2002) AAV-mediated gene transfer of pigment epithelium-derived factor inhibits choroidal neovascularization. Invest Ophthalmol Vis Sci 43:1994–2000
Neri I, Balestri R, Patrizi A et al (2012) Hemangiomas: new insight and medical treatment. Dermatol Ther 25:322–334
Rosen LS (2002) Clinical experience with angiogenesis signaling inhibitors: focus on vascular endothelial growth factor (VEGF) blockers. Cancer Control 9(2 Suppl):36–44
Stellmach V, Crawford SE, Zhou W et al (2001) Prevention of ischemia-induced retinopathy by the natural ocular antiangiogenic agent pigment epithelium-derived factor. Proc Natl Acad Sci USA 98:2593–2597
Takanori M, Yuri N, Sayaka M et al (2012) PEDF-derived peptide inhibits corneal angiogenesis by suppressing VEGF expression. Microvasc Res 84:105–108
Takenak K, Yamagishi S, Jinnouchi Y, et al. (2005) Pigment epithelium-derived factor(PEDF) induced apoptosis and inhibition of vascular endothelial growth factor(VEGF) expression in MG63 human osteosarcoma cells. Life Sci 77:3231–3241
Wang Q, Zhang Z, Ding T et al (2013) Mesenchymal stem cells overexpressing PEDF decreased the angiogenesis of gliomas. Biosci Rep 33:e00019
Wu QJ, Gong CY, Luo ST et al (2012) AAV-mediated human PEDF inhibits tumor growth and metastasis in murine colorectal peritoneal carcinomatosis model. BMC Cancer 12:129
Yang H, Cheng R, Liu G et al (2009) PEDF inhibits growth of retinoblastoma by anti-angiogenic activity. J. Cancer Sci 100:2419–2425
Yang LP, Cheng P, Peng XC et al (2009) Anti-tumor effect of adenovirus-mediated gene transfer of pigment epithelium-derived factor on mouse B16-F10 melanoma. J Exp Clin Cancer Res 28:75
Zhang SX, Wang JJ, Gao G et al (2006) Pigment epithelium- derived factor downregulates vascular endothelial growth factor (VEGF) expression and inhibits VEGF-VEGF receptor 2 binding in diabetic retinopathy. J Mol Endocrinol 37:1–12
Zheng D, Chen H, Davids J et al (2013) Serpins for diagnosis and therapy in cancer. Cardiovasc Hematol Disord Drug Targets 13:123–132
Acknowledgements
Sincere thanks are due to Fangmeng Fu for his valuable academic suggestion and his warm encouragement, and to Yuanjing Chen for his assistance with the experiments.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
This research was supported by Fujian Youth Foundation of Fujian Health Department (2013-1-28) and the National Natural Science Foundation of China (81470125).
Ethical approval
All procedures performed in the study involving human participants were in accordance with the ethical standards of the Bioethical Committee of Affiliated Fujian Union Hospital of Fujian Medical University and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from parents of all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Li, M., Chen, Y., Guo, Z. et al. The pigment epithelium-derived factor (PEDF): an important potential therapeutic agent for infantile hemangioma. Arch Dermatol Res 309, 169–178 (2017). https://doi.org/10.1007/s00403-017-1716-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00403-017-1716-5