Abstract
Introduction
Most adult cases of bacterial–septic–arthritis of a native joint are effectively managed with a single surgical debridement, but some cases may require more than one debridement to control the infection. Consequently, this study assessed the failure rate of a single surgical debridement in adults with bacterial arthritis of a native joint. Additionally, risk factors for failure were assessed.
Materials and Methods
The review protocol was registered on PROSPERO (CRD42021243460) before data collection and conducted in line with the ‘Preferred Reporting Items for Systematic Reviews and Meta-Analyses’ (PRISMA) guidelines. Multiple libraries were systematically searched to identify articles including patients reporting on the incidence of failure (i.e. persistence of infection requiring reoperation) of the treatment of bacterial arthritis. The quality of individual evidence were assessed using the Quality in Prognosis Studies (QUIPS) tool. Failure rates were extracted from included studies and pooled. Risk factors for failure were extracted and grouped. Moreover, we evaluated which risk factors were significantly associated with failure.
Results
Thirty studies (8,586 native joints) were included in the final analysis. The overall pooled failure rate was 26% (95% CI 20 to 32%). The failure rate of arthroscopy and arthrotomy was 26% (95% CI 19 to 34%) and 24% (95% CI 17 to 33%), respectively. Seventy-nine potential risk factors were extracted and grouped. Moderate evidence was found for one risk factor (synovial white blood cell count), and limited evidence was found for five risk factors (i.e. sepsis, large joint infection, the volume of irrigation, blood urea nitrogen-test, and blood urea nitrogen/creatinine ratio).
Conclusion
A single surgical debridement fails to control bacterial arthritis of a native joint in approximately a quarter of all adult cases. Limited to moderate evidence exists that risk factors associated with failure are: synovial white blood cell count, sepsis, large joint infection, and the volume of irrigation. These factors should urge physicians to be especially receptive to signs of an adverse clinical course.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bacterial arthritis, also known as septic arthritis, of a native joint is a clinical emergency that requires prompt treatment [1,2,3,4]. Most patients are effectively managed with a single surgical debridement of the joint in combination with systemic antibiotics, but some cases may require more than one debridement to control the infection.
Reported failure rates of a single surgical debridement vary widely [5, 6], and a structured assessment of risk factors for failure is lacking. Identification of risk factors may help to create a more uniform and evidence-based treatment approach to bacterial arthritis in clinical practice, which now relies predominantly on convention and local preferences [7, 8].
Consequently, the purpose of this systematic review is to assess the failure rate of a single –arthroscopic or open– surgical debridement in adults with bacterial arthritis of a native joint and to identify risk factors (e.g. demographics, medical history, lab markers, immunosuppression, and initial treatment approach) for failure of a single surgical debridement.
Materials and methods
Study design
The review protocol was registered on PROSPERO (CRD42021243460) before data collection. This study was conducted in line with the ‘Preferred Reporting Items for Systematic Reviews and Meta-Analyses’ (PRISMA) guidelines [9].
In consult with a medical librarian, we searched eligible studies using the PubMed, Embase, and Cochrane libraries between January 1980 and January 2021 using the following keywords: “bacterial arthritis”, “treatment failure”, “native joint”, and “risk factors”, including synonyms, related words, and MeSH Terms (appendix A).
Studies were included if they included 10 or more patients (16 years or older) with bacterial arthritis of a native appendicular joint, who underwent either open or arthroscopic surgical debridement. Furthermore, the studies had to report the incidence of failure (i.e. persistence of infection requiring reoperation or mortality) of the treatment. We excluded studies that did not present original data, meeting abstracts, case reports, animal or cadaveric studies, studies reporting on arthroplasty or with a foreign body in the affected joint (e.g. anchor), and studies published in a language other than English.
Two reviewers (AW, TS) independently screened titles and abstracts for eligibility using predefined criteria. Subsequently, full texts of selected papers were obtained and screened. Discordant judgment in study selection was resolved by consensus discussion together with a third reviewer (SJ). Bibliographies of included studies were screened to assess whether eligible studies were missed by our search.
Data were extracted using Microsoft Excel v. 16.52 (Microsoft Inc., Redmond, WA, USA).
Two authors (AW, TS) assessed the risk of bias independently and the Quality in Prognostic Studies (QUIPS) tool was used [10]. The QUIPS tool guides quality assessment in 6 domains: study participation, study attrition, prognostic factor measurement, outcome measurement, study confounding, and statistical analysis and reporting. The risk of bias is reported as low, moderate, or high for each domain and then an overall risk of bias is assigned based on the ratings in each domain (appendix B1-B2).
Variables and outcome measures
The primary outcome variable was the rate of failure. Additionally, the following variables were extracted per study: year of publication, country, study type, years of inclusion, number of patients and joints with bacterial arthritis, sex distribution, age, microorganism profile, surgical technique, number of total operations, and potential risk factors associated with failure of a single surgical debridement.
We also extracted the criteria used to define failure of a single surgical debridement.
Statistical analysis
We performed a meta-analysis by pooling the failure rate from individual studies using a random-effects model with an inverse variance method and logit transformation. 95%-confidence intervals were assessed using the Clopper-Pearson interval. We separately analysed the failure rate of arthroscopy and arthrotomy, and the failure rate of shoulder and knee bacterial arthritis to increase homogeneity. Heterogeneity among studies was evaluated by calculating I2 and a chi-square Q test [11]. A p-value of < 0.05 was considered to indicate significant heterogeneity. Analyses were conducted using the meta package in R version 3.04 (R studio, Northern Ave, Boston, USA) [12].
Best-evidence-synthesis
The strength of the evidence for risk factors was evaluated using a best-evidence synthesis in absence of reported effect sizes, which did not allow pooling of data. Only the risk factors with significant association in at least one included study were categorized. The strength of evidence for each identified risk factor was based on the guidelines by Furlan et al. [13], additional details on the guidelines are provided in Appendix B.
The overall risk of bias was reported as low, moderate, or high. Studies reported as low risk of bias were categorized as high-quality studies, and studies reported as moderate and high risk of bias were reported as low-quality studies).
Results
Study characteristics and quality appraisal
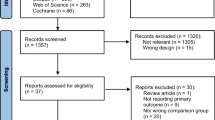
The search yielded 1,836 potentially eligible studies (Fig. 1). After screening titles and abstracts, 1,739 studies were excluded. Subsequent full-text screening resulted in 30 studies that were included in this review. Twenty-nine studies (97%) had a retrospective design. The majority of the included studies (n = 26, 87%) were published after 2010.
Twelve of the studies [5, 14,15,16,17,18,19,20,21,22,23,24] were at low risk of bias, five [6, 25,26,27,28] were at high risk of bias and 13 [29,30,31,32,33,34,35,36,37,38,39,40,41] were at moderate risk of bias (Fig. 2 and Appendix B1-B2).
Patient characteristics
The 30 studies reported on a total of 8,569 patients with bacterial arthritis of 8,586 native joints. The average age of patients was 59 years (range: 42 to 72 years) and 61% were men (range: 31 to 89%; Table 1).
Bacterial arthritis was most commonly reported in the shoulder (70%), followed by the knee (21%) (Appendix C). The most commonly identified microorganism was Staphylococcus Aureus (44%) (Table 2). Of the 8,586 surgical debridements, 5,680 (66%) were performed via an arthrotomy and 2,855 (33%) arthroscopically. One study (n = 51, 1%) did not report the surgical approach [35].
The failure rate
The overall pooled failure rate of a single surgical debridement was 25.5% (range among studies: 8.3% to 72% (95% CI 20 to 32%, Fig. 3). The heterogeneity was high with I2 = 96% (p < 0.01). Therefore, additional analyses were conducted in an attempt to parse out this heterogeneity. The study of Jiang et al. [33] (5154 shoulders, with a failure rate of 12.4%) had a relatively high weight of 60% in the model. However, the removal of the study from the model did not affect the pooled failure rate (26.2%; 95% CI 21 to 33%, I2 = 93%).
A sub-group analysis of the failure rate of the separate surgical approaches, which was reported in 25 studies, showed a failure rate of arthroscopy of 26% (95% CI 19 to 34%, I2 = 92%, ranging from 4.9 to 72%, Fig. 4), and a failure rate of arthrotomy of 24% (95% CI 17 to 33%, I2 = 96%, ranging from 8.3% to 70%). Thirteen studies specifically compared arthroscopy and arthrotomy as potential risk factors. Four out of thirteen (31%) studies identified arthrotomy as a statistically significant risk factor for failure of a single debridement. In contrast, two out of these thirteen (15%) studies identified arthroscopy as a significant risk factor (Table 3).
A sub-group analysis of the failure rate of the shoulder, which was reported in 11 studies, showed an overall failure rate of 19% (95% 13 to 26%, I2 = 84%, ranging from 8.3 to 56%, appendix D). The failure rate of shoulder arthroscopy was 22% (95% CI 13 to 34%, I2 = 89%, ranging from 8.4 to 56%), and for arthrotomy it was 14% (95% CI 12 to 17%, I2 = 0%, ranging from 8.3 to 18%). A sub-group analysis of the failure rate of the knee, which was reported in eight studies, showed an overall failure rate of 26% (95% CI 17 to 44%, I2 = 93%, ranging from 4.9 to 72%, appendix D). The failure rate of knee arthroscopy was 26% (95% CI 14 to 44%, ranging from 4.9 to 72%), and for arthrotomy it was 25% (95% CI 13 to 44%, ranging from 14 to 70%). The failure rate of the hip was reported in three studies and showed a failure rate of 10% (n = 51), 45% (n = 421), and 53% (n = 17). Only one of these studies reported the failure rate of arthroscopy (38%) and arthrotomy (46%), separately.
Risk factors for failure of single surgical debridement
The included studies investigated 79 different factors for association with failure. Twenty-six of these factors were found to be a statistically significant predictor of failure of a single debridement in one or more studies (Table 3). Based on the best-evidence-syntheses of risk factors for failure, there were no positive risk factors classified as strong evidence, one risk factor (i.e. synovial white blood cell count) classified as moderate evidence, and five risk factors (i.e. systemic sepsis, large joint infection, the volume of irrigation, blood urea nitrogen-test (BUN), and BUN/creatinine ratio) were classified as limited evidence. Twelve risk factors were classified as conflicting evidence and four as inconclusive evidence (Table 3 and Appendix C).
Definitions of failure of single surgical debridement (Table 4)
The most commonly used definition of failure was a combination of clinical findings, elevated laboratory signs of systemic inflammation, and/or positive/purulent fluids analysis requiring reoperation (n = 14, 47%).
Discussion
The main finding of this study is that bacterial arthritis of a native joint can be treated successfully by a single surgical debridement in combination with systemic antibiotic therapy in the majority of patients. However, one should be vigilant, as in about a quarter, additional or repeated treatment is necessary to control the infection. Based on the best-evidence-synthesis; synovial white blood cell count, systemic sepsis, large joint infection, the volume of irrigation, blood urea nitrogen test, and blood urea nitrogen/creatinine ratio can be considered as risk factors for failure with limited to moderate evidence.
A wide range of failures from 8 to 72% was found in the included studies. The three studies with the lowest score in the risk of bias analyses and inclusion of several joints, Besnard et al., Hunter et al., and, Jung et al. [15, 18, 21] showed that after a single surgical debridement, 33% to 38% of patients require a reoperation. This is slightly higher than the 95% confidence interval of the pooled overall failure rates, which could indicate underreporting in higher risk-of-bias studies.
Over the years, various treatments have been advocated for bacterial arthritis, including repeated non-operative needle aspiration, open synovectomy, and arthroscopy with or without synovectomy. Early and aggressive intervention is essential in eradicating the infection and preventing joint damage [42]. In our study, we encompassed a wide timeframe for the inclusion of studies, ranging from 1980 to 2021. It is worth noting that four studies included in our analysis had an inclusion period predating 2000. This factor raises the possibility of potential influence on the results due to subsequent advancements in techniques. Furthermore, arthroscopic techniques have gained popularity as an alternative to arthrotomy in the treatment of bacterial arthritis. They allow for minimally invasive joint lavage and debridement, effectively reducing bacterial load and improving treatment outcomes [43]. Moreover, needle arthroscopy, an even less invasive approach, has emerged as a promising option in the management of bacterial arthritis, providing visualization and irrigation of the infected area, under local anaesthesia, through the insertion of a small arthroscope [44, 45].
In terms of failure rate, our systematic review does not support one surgical approach (i.e., arthrotomy versus arthroscopy) over another. Also, there was no clear difference found in the reported risk factor analyses that included the surgical approach. However, it should be noted that (1) these results are likely affected by selection bias; i.e. a longstanding or more severely ill patient is perhaps more likely to undergo arthrotomy versus arthroscopy, and (2) this systematic review was not designed to directly compare arthroscopy and arthrotomy in terms of other outcomes. A recent systematic review that did focus on these outcomes found a significantly lower complication rate and duration of hospital stay after arthroscopy [46].
In the shoulder, we found a pooled failure rate of 19%. Memon et al.[47] found –in a systematic review in 2018 including 121 shoulders– a failure rate of 30%. This difference might be explained by the fact that they included all ages (including children), did not focus on isolated bacterial arthritis, and included case reports which are often exceptional cases that may relate to higher reoperation and/or persistent infection rates. Furthermore, large studies, including one study with 5,154 cases [33], were published recently and were additionally included in our review. Therefore, the results of this review probably better reflect the failure rate in adult patients with bacterial arthritis of the native shoulder joint.
In the knee, we found an overall pooled failure rate of 26% (26% for arthroscopy, and 25% for arthrotomy). In a systematic review and meta-analysis –including seven studies with 1,089 knees– by Panjwani et al. [48], a failure rate of 17% in the arthroscopy group and 22% in the arthrotomy group was described. The authors concluded that arthroscopy results in a significantly lower re-operation rate than arthrotomy [48]. These findings should be interpreted with care as to the earlier mentioned potential selection bias inherent to non-experimental retrospective studies. Our results contradict their conclusion. Besides, in a randomized clinical trial by Peres et al. [49], no significant difference was shown between arthrotomy and arthroscopy; albeit both groups were small. Highly-quality experimental (randomized) controlled studies are necessary to confirm superiority or non-inferiority. Such study should not only focus on reoperation rate, but also on Patient-Reported Outcomes Measures, functional outcomes, hospital stay, and complications.
In the hip, we found a failure rate ranging from 10 to 53%. Because only three studies focused on the hip, we abstained from data pooling. According to a recent systematic review (n = 25 patients), arthroscopy was found to be both safe and effective in treating bacterial hip arthritis; however, there was no superiority of arthroscopy over arthrotomy (or vice-versa) [50]. In our review, we analysed a study involving 421 patients, out of which 387 received an arthrotomy and 34 underwent an arthroscopy. This study’s findings indicate that patients with bacterial arthritis of the hip demonstrated comparable rates of short-term complications and reoperations [36]. Based on this limited evidence there is no preference for hip arthroscopy over arthrotomy (or vice-versa). This is an important finding as hip arthroscopy is challenging and requires specific expertise that is not always available.
There were 26 risk factors associated with failure. None of these risk factors were classified as strong evidence. Six risk factors were classified as moderate or limited evidence: synovial white blood cell count, sepsis, large joint infection, the volume of irrigation, blood urea nitrogen (BUN) test, and BUN/creatinine ratio. The BUN test and BUN/creatinine ratio are tests that are not routinely used [51]. Besides, these two parameters were only investigated by one small retrospective study (n = 63) [23], and hence should be interpreted with care. Synovial white blood cell count, sepsis, large joint infection, and the volume of irrigation are associated with the severity of the infection and virulent organisms. Another interesting finding of this review is that commonly assumed risk factors such as specific micro-organisms (e.g., Methicillin-resistant Staphylococcus aureus (MRSA), but also rheumatoid arthritis, diabetes, and other comorbidities were not found to be associated with failure.
We found five different criteria that helped define failure of a single surgical debridement among included studies. A combination of clinical findings, laboratory signs of systemic inflammation, and/or positive/purulent fluid analyses was the most commonly used criterion to prompt additional intervention. Nonetheless, none of the studies described clear cut-off points for failure of a single debridement and it was presumably ultimately at the discretion of the treating physician, which is challenging due to the lack of good quality diagnostic tests [52].
Limitations
This study has several limitations. First, it is limited by the quality of included studies, which were all prone in varying degrees to bias. Study confounding and prognostic factor measurement were the most commonly identified biases (Fig. 2). Second, most of the meta-analyses presented significant heterogeneity among included studies. This can be explained by a wide range of populations, joints, and surgical techniques between studies. Therefore, random-effects models and sub-analyses by surgical technique and affected joint were conducted. No major differences were found between surgical techniques. However, in the studies on the shoulder joint, slightly lower failure rates were found than in the knee. The differences in joints between studies may therefore have been a source of heterogeneity. Third, a wide variety in number and definition of risk factors made it impossible to pool the risk factors for failure. Future well-designed high-quality studies are merited to confirm these results. Fourth, in contrast to what we expected based on our clinical practice, the shoulder was the most commonly affected joint in this systematic review–- mainly driven by the study of Jiang et al. [33] that added 5154 shoulder joints to our study. A sensitivity analysis that excluded these shoulders did not affect the overall pooled failure rate. Fifth, it should be noted that this review included all joints of the appendicular skeleton; nevertheless, a mere 0.9% (78 joints) of the arthritis pertained to small joints, including: the wrist, elbow, and ankle. It is important to note that inclusion of these cases does not significantly impact the overall pooled failure rate. To clarify, whether all these joints had either failed or succeeded, the overall failure rate would have only increased or decreased by no more than 1%. Sixth, although we had a wide timeframe for inclusion of studies (1980–2021), we only found 4 studies with an inclusion period predating 2000, which may influence the results due to the subsequent advancements in technique. However, it is important to note that the majority of the studies included in this review (n = 26) had an inclusion period after 2000, and thus generally reflect current practice.
Conclusion
In conclusion, this systematic review found that a single surgical debridement fails to control the infection in native joint bacterial arthritis in 26% of cases. No difference in failure rates was found between arthroscopy and arthrotomy. Limited to moderate evidence exists that risk factors associated with failure are synovial white blood cell count, sepsis, large joint infection, and the volume of irrigation. These factors should urge physicians to be especially receptive to signs of an adverse clinical course.
Data availability
Not applicable.
References
Riegels-Nielsen P, Frimodt-Moller N, Sorensen M, Jensen JS (1989) Antibiotic treatment insufficient for established septic. Staphylococcus aureus experiments in rabbits. Acta Orthop Scand 60(1):113–5
Mathews CJ, Coakley G (2008) Septic arthritis: current diagnostic and therapeutic algorithm. Curr Opin Rheumatol 20(4):457–462
Mathews CJ, Kingsley G, Field M, Jones A, Weston VC, Phillips M et al (2008) Management of septic arthritis: a systematic review. Postgrad Med J 84(991):265–270
Mathews CJ, Weston VC, Jones A, Field M, Coakley G (2010) Bacterial septic arthritis in adults. Lancet 375(9717):846–855
Al-Nammari SS, Bobak P, Venkatesh R (2007) Methicillin resistant Staphylococcus aureus versus methicillin sensitive Staphylococcus aureus adult haematogenous septic arthritis. Arch Orthop Trauma Surg. 127:537–42
Kuo CL, Chang JH, Wu CC, Shen PH, Wang CC, Lin LC et al (2010) Treatment of septic knee arthritis: comparison of arthroscopic debridement alone or combined with continuous closed irrigation-suction system. J Trauma. 71(1):454–9
Coakley G, Mathews C, Field M, Jones A, Kingsley G, Walker D et al (2006) BSR & BHPR, BOA, RCGP and BSAC guidelines for management of the hot swollen joint in adults. Rheumatology (Oxford) 45(8):1039–1041
Weston V, Coakley G (2006) British Society for Rheumatology Standards G, Audit Working G, British Society for Antimicrobial C, British Orthopaedic A et al guideline for the management of the hot swollen joint in adults with a particular focus on septic arthritis. J Antimicrob Chemother. 58(3):492–3
Moher D, Liberati A, Tetzlaff J (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 339:b2535
Hayden JA, van der Windt DA, Cartwright JL, Cote P, Bombardier C (2013) Assessing bias in studies of prognostic factors. Ann Intern Med 158(4):280–286
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21(11):1539–1558
Balduzzi S, Rucker G, Schwarzer G (2019) How to perform a meta-analysis with R: a practical tutorial. Evid Based Ment Health 22(4):153–160
Furlan AD, Pennick V, Bombardier C, van Tulder M, Editorial Board CBRG (2009) 2009 updated method guidelines for systematic reviews in the Cochrane Back Review Group. Spine (Phila Pa 1976) 34(18):1929–41
Assunção JH, Noffs GG, Malavolta EA, Gracitelli MEC, Lima ALM, Ferreira Neto AA (2018) Septic arthritis of the shoulder and elbow: one decade of epidemiological analysis at a tertiary referral hospital. Rev Bras Ortop. 53(6):707–13
Besnard M, Babusiaux D, Garaud P, Rosset P, Bernard L, Le Nail LR et al (2018) Impact of setting up a bone and joint infection referral center on arthroscopic treatment of septic arthritis of the knee and shoulder: Retrospective study. Orthop Traumatol Surg Res. 104(8):1265–9
Bovonratwet P, Fu MC, Pathak N, Ondeck NT, Bohl DD, Nho SJ et al (2019) Surgical Treatment of septic shoulders: a comparison between arthrotomy and arthroscopy. Arthroscopy. 35(7):1984–91
Bovonratwet P, Nelson SJ, Bellamkonda K, Ondeck NT, Shultz BN, Medvecky MJ et al (2017) Similar 30-Day Complications for Septic Knee Arthritis Treated With Arthrotomy or Arthroscopy: An American College of Surgeons National Surgical Quality Improvement Program Analysis. Arthroscopy. 34(1):213–9
Hunter JG, Gross JM, Dahl JD, Amsdell SL, Gorczyca JT (2015) Risk factors for failure of a single surgical debridement in adults with acute septic arthritis. J Bone Joint Surg Am. 97(7):558–64
Johns BP, Loewenthal MR, Dewar DC (2017) Open compared with arthroscopic treatment of acute septic arthritis of the native knee. J Bone Joint Surg Am. 99(6):499–505
Joo YB, Lee WY, Shin HD, Kim KC, Kim YK (2019) Risk factors for failure of eradicating infection in a single arthroscopic surgical procedure for septic arthritis of the adult native shoulder with a focus on the volume of irrigation. J Shoulder Elbow Surg. 29(3):497–501
Jung SW, Kim DH, Shin SJ, Kang BY, Eho YJ, Yang SW (2017) Septic arthritis associated with systemic sepsis. Int Orthop. 42(1):1–7
Sammer DM, Shin AY (2009) Comparison of arthroscopic and open treatment of septic arthritis of the wrist. J Bone Joint Surg Am. 91(6):1387–93
Stake S, Scully R, Swenson S, Lee D, Lee R, Sparks A et al (2020) Repeat irrigation & debridement for patients with acute septic knee arthritis: Incidence and risk factors. J Clinical Orthopaedics Trauma. 11(177):S177–S83
Abdou MA, Jo A, Choi IS, Iim CJ, Park HK, Oh HK, et al. (2019) Shoulder Joint Infections with Negative Culture Results: Clinical Characteristics and Treatment Outcomes. BioMed Research International.
Aim F, Delambre J, Bauer T, Hardy P (2015) Efficacy of arthroscopic treatment for resolving infection in septic arthritis of native joints. Orthopaedics Traumato: Surg Res. 101(1):61–4
Faour M, Sultan AA, George J, Samuel LT, Curtis GL, Molloy R et al (2019) Arthroscopic irrigation and debridement is associated with favourable short-term outcomes vs open management: an ACS-NSQIP database analysis. Knee Surg Sports Traumatol Arthrosc. 27(10):3304–10
Jeon IH, Choi CH, Seo JS, Seo KJ, Ko SH, Park JY (2006) Arthroscopic management of septic arthritis of the shoulder joint. J Bone Joint Surg Series A. 88(8):1802–6
Matsuhashi T, Suenaga N, Oizumi N, Iwasaki N, Minami A (2011) Arthroscopic debridement with continuous irrigation for septic arthritis of the shoulder joint. Europ Orthopaedics Traumatology. 2(4):87–92
Bohler C, Dragana M, Puchner S, Windhager R, Holinka J (2016) Treatment of septic arthritis of the knee: a comparison between arthroscopy and arthrotomy. Knee Surg, Sports Traumatology, Arthroscopy : off J ESSKA 24(10):3147–54
Bohler C, Pock A, Waldstein W, Staats K, Puchner SE, Holinka J et al (2017) Surgical treatment of shoulder infections: a comparison between arthroscopy and arthrotomy. J Shoulder Elbow Surg. 26(11):1915–21
Cho CH, Oh GM (2016) Prognostic factors affecting the clinical outcome of septic arthritis of the shoulder. J Hand Surg Asian Pac 21(3):339–44
Jaffe D, Costales T, Greenwell P, Christian M, Henn IR (2017) 3rd Methicillin-resistant staphylococcus aureus infection is a risk factor for unplanned return to the operating room in the surgical treatment of a septic knee. J Knee Surg. 30(9):872–8
Jiang JJ, Piponov HI, Mass DP, Angeles JG, Shi LL (2017) Septic arthritis of the shoulder: a comparison of treatment methods. J Am Acad Orthop Surg. 25(8):e175–e84
Kang T, Lee JK (2018) Host factors affect the outcome of arthroscopic lavage treatment of septic arthritis of the knee. Orthopedics. 41(2):e184–e8
Kao FC, Hsu YC, Liu PH, Tu YK, Jou IM (2019) High 2-year mortality and recurrent infection rates after surgical treatment for primary septic arthritis of the hip in adult patients: An observational study. Medicine (Baltimore) 98(32):e16765
Khazi ZM, Cates WT, An Q, Duchman KR, Wolf BR, Westermann RW (2019) Arthroscopy versus open arthrotomy for treatment of native hip septic arthritis: an analysis of 30-day complications. Arthroscopy 36(4):1048–52
Khazi ZM, Cates WT, Shamrock AG, An Q, Duchman KR, Westermann RW et al (2020) Arthroscopic débridement has similar 30-day complications compared with open arthrotomy for the treatment of native shoulder septic arthritis: a population-based study. J Shoulder Elbow Surg. 29(6):1121–6
Lee DK, Rhee SM, Jeong HY, Ro K, Jeon YS, Rhee YG (2019) Treatment of acute shoulder infection: can osseous lesion be a rudder in guideline for determining the method of débridement? J Shoulder Elbow Surg. 28(12):2317–25
Mabille C, El Samad Y, Joseph C, Brunschweiler B, Goeb V, Grados F, et al. (2008) Medical versus surgical treatment in native hip and knee septic arthritis. Med Mal Infect.
Rhee SM, Lee DK, Park JS, Rhee YG (2020) The prognostic value of a novel magnetic resonance imaging-based classification for septic arthritis of the shoulder. J Bone Joint Surg Am. 102(15):1321–8
Stutz G, Kuster MS, Kleinstück F, Gächter A (2000) Arthroscopic management of septic arthritis: stages of infection and results. Knee Surg Sports Traumatol Arthrosc. 8(5):270–4
Tarkowski A (2006) Infection and musculoskeletal conditions: Infectious arthritis. Best Pract Res Clin Rheumatol 20(6):1029–1044
Aim F, Delambre J, Bauer T, Hardy P (2015) Efficacy of arthroscopic treatment for resolving infection in septic arthritis of native joints. Orthop Traumatol Surg Res 101:61–64
Stornebrink T, Janssen SJ, Kievit AJ, Mercer NP, Kennedy JG, Stufkens SAS et al (2021) Bacterial arthritis of native joints can be successfully managed with needle arthroscopy. J Exp Orthop 8(1):67
Walinga AB, Stornebrink T, Janssen SJ, Dalmau-Pastor M, Kievit AJ, Kerkhoffs GMM (2022) Needle arthroscopy for bacterial arthritis of a native joint: surgical technique for the shoulder, elbow, wrist, knee, and ankle under local Anesthesia. Arthrosc Tech 11(9):e1641–e1648
Acosta-Olivo C, Vilchez-Cavazos F, Blazquez-Saldana J, Villarreal-Villarreal G, Pena-Martinez V, Simental-Mendia M (2021) Comparison of open arthrotomy versus arthroscopic surgery for the treatment of septic arthritis in adults: a systematic review and meta-analysis. Int Orthop 45(8):1947–1959
Memon M, Kay J, Ginsberg L, de Sa D, Simunovic N, Samuelsson K et al (2018) Arthroscopic management of septic arthritis of the native shoulder: a systematic review. Arthroscopy. 34(2):625–46
Panjwani T, Wong KL, Tan SHS, Liau G, Vaidya N, Krishna L (2019) Arthroscopic debridement has lower re-operation rates than arthrotomy in the treatment of acute septic arthritis of the knee: a meta-analysis. J ISAKOS. 4(1):307–12
Peres LR, Marchitto RO, Pereira GS, Yoshino FS, de Castro FM, Matsumoto MH (2016) Arthrotomy versus arthroscopy in the treatment of septic arthritis of the knee in adults: a randomized clinical trial. Knee Surg Sports Traumatol Arthrosc 24(10):3155–3162
D’Angelo F, Monestier L, Zagra L (2021) Active septic arthritis of the hip in adults: what’s new in the treatment? A Syst Rev EFORT Open Rev 6(3):164–172
Lyman JL (1986) Blood urea nitrogen and creatinine. Emerg Med Clin North Am 4(2):223–233
Walinga AB, Stornebrink T, Langerhuizen DWG, Struijs PAA, Kerkhoffs GMMJ, Janssen SJ (2021) What are the best diagnostic tests for diagnosing bacterial arthritis of a native joint? : a systematic review of 27 studies. Bone Joint J. 103(12):1745–53
Acknowledgements
We thank Faridi S. van Etten–Jamaludin, Medical Librarian Amsterdam University Medical Center for help in developing our search syntax.
Funding
No funding was received to support conduct of this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors received payments of any kind related to the subject of this study.
Ethical approval
This study is in accordance with the ethical standards in the 1964 Declaration of Helsinki.
Informed consent
Not applicable
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Walinga, A.B., Stornebrink, T., Emanuel, K.S. et al. Failure rates in surgical treatment in adults with bacterial arthritis of a native joint: a systematic review of 8,586 native joints. Arch Orthop Trauma Surg 143, 6547–6559 (2023). https://doi.org/10.1007/s00402-023-04958-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00402-023-04958-z