Abstract
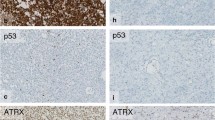
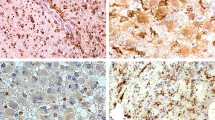
Oligoastrocytomas are glial tumours consisting of a mixture of neoplastic astrocytic and oligodendroglial cells. Genetic alterations of oligoastrocytomas include loss of heterozygosity of chromosomes 1p and/or 19q (LOH 1p/19q), typically occurring in oligodendrogliomas, and mutations of TP53, frequently occurring in astrocytomas. To investigate whether these neoplastic cell types in oligoastrocytomas have different genetic profiles, we examined the two different components of oligoastrocytomas in comparison with the histological diagnosis of the specific tumour area for LOH 1p/19q and TP53 mutations by using microdissection technique. We found a variety of lost markers for 1p and 19q, and the presence of two different TP53 mutations in the tumour samples. In the majority of cases (9/11), the oligodendroglial and astrocytic components of an individual oligoastrocytoma displayed the same genotype. We present two cases of biphasic oligoastrocytomas with aberrant findings, suggesting the coexistence of genetically and morphologically distinct tumour cell clones in these tumours. In one case, the oligodendroglial part of the tumour showed LOH19q, whereas the astrocytic part showed TP53 mutation (codon 273). In another case, we found LOH 1p/19q in the oligodendroglial component, but two retained areas on chromosome 1p in the astrocytic component of the tumour. No evidence was found for the coexistence of tumour cells with the two genotypical changes within the same morphological region of one individual tumour. The two cases of biphasic oligoastrocytomas in our sample that display a different genotype in the astrocytic and oligodendroglial part of the tumour show that different components of an oligoastrocytoma may be derived from different cell clones during neoplastic transformation.





Similar content being viewed by others
References
Berg C, Hedrum A, Holmberg A, Ponten F, Uhlen M, Lundeberg J (1995) Direct solid-phase sequence analysis of the human p53 gene by use of multiplex polymerase chain reaction and alpha-thiotriphosphate nucleotides. Clin Chem 41:1461–1466
Chompret A, Brugieres L, Ronsin M, Gardes M, Dessarps-Freichey F, Abel A, Hua D, Ligot L, Dondon MG, Bressac-de Paillerets B, Frebourg T, Lemerle J, Bonaiti-Pellie C, Feunteun J (2000) P53 germline mutations in childhood cancers and cancer risk for carrier individuals. Br J Cancer 82:1932–1937
Coons SW, Johnson PC, Shapiro JR (1995) Cytogenetic and low cytometric DNA analysis of regional heterogeneity in a low grade glioma. Cancer Res 55:1569–1577
von Deimling A, Eibl RH, Ohgaki H, Louis DN, von Ammon K, Petersen I, Kleihues P, Chung RY, Wiestler OD, Seizinger BR (1992) p53 mutations are associated with 17p allelic loss in grade II and grade III astrocytoma. Cancer Res 52:2987–2990
von Deimling A, Fimmers R, Schmidt MC, Bender B, Fassbender F, Nagel J, Jahnke R, Kaskel P, Duerr EM, Koopmann J, Maintz D, Steinbeck S, Wick W, Platten M, Muller DJ, Przkora R, Waha A, Blumcke B, Wellenreuther R, Meyer-Puttlitz B, Schmidt O, Mollenhauer J, Poustka A, Stangl AP, Lenartz D, von Ammon K (2000) Comprehensive allelotype and genetic anaysis of 466 human nervous system tumors. J Neuropathol Exp Neurol 59:544–558
Dong ZQ, Pang JC, Tong CY, Zhou LF, Ng HK (2002) Clonality of oligoastrocytomas. Hum Pathol 33:528–535
Hart MN, Petito CK, Earle KM (1974) Mixed gliomas. Cancer 33:134–140
Hedrum A, Ponten F, Ren Z, Lundeberg J, Ponten J, Uhlen M (1994) Sequence-based analysis of the human p53 gene based on microdissection of tumor biopsy samples. Biotechniques 17:118–119, 122–124, 126–129
Kim SH, Kim H, Kim TS (2005) Clinical, histological, and immunohistochemical features predicting 1p/19q loss of heterozygosity in oligodendroglial tumors. Acta Neuropathol 110:27–38
Kleihues P, Cavenee WK (2000) World Health Organization classification of tumors, pathology & genetics, tumours of the nervous system. International Agency for Research on Cancer (IARC) Press, Lyon
Kleihues P, Louis DN, Scheithauer BW, Rorke LB, Reifenberger G, Burger PC, Cavenee WK (2002) The WHO classification of tumors of the nervous system. J Neuropathol Exp Neurol 61:215–225; discussion 26–29
Konig EA, Koves I, Rasinariu A, Popp AR, Kusser WC, Soyonki K, Kovacs A, Glickman BW, Jeney A, Marcsek ZL (2001) Alterations of K-ras and p53 mutations in colorectal cancer patients in Central Europe. J Toxicol Environ Health A 62:333–347
Kraus JA, Koopmann J, Kaskel P, Maintz D, Brandner S, Schramm J, Louis DN, Wiestler OD, von Deimling A (1995) Shared allelic losses on chromosomes 1p and 19q suggest a common origin of oligodendroglioma and oligoastrocytoma. J Neuropathol Exp Neurol 54:91–95
Krouwer HG, van Duinen SG, Kamphorst W, van der Valk P, Algra A (1997) Oligoastrocytomas: a clinicopathological study of 52 cases. J Neurooncol 33:223–238
Leitner T, Halapi E, Scarlatti G, Rossi P, Albert J, Fenyo EM, Uhlen M (1993) Analysis of heterogeneous viral populations by direct DNA sequencing. Biotechniques 15:120–127
Maintz D, Fiedler K, Koopmann J, Rollbrocker B, Nechev S, Lenartz D, Stangl AP, Louis DN, Schramm J, Wiestler OD, von Deimling A (1997) Molecular genetic evidence for subtypes of oligoastrocytomas. J Neuropathol Exp Neurol 56:1098–1104
Mueller W, Hartmann C, Hoffmann A, Lanksch W, Kiwit J, Tonn J, Veelken J, Schramm J, Weller M, Wiestler OD, Louis DN, von Deimling A (2002) Genetic signature of oligoastrocytomas correlates with tumor location and denotes distinct molecular subsets. Am J Pathol 161:313–319
van Meyel DJ, Ramsay DA, Casson AG, Keeney M, Chambers AF, Cairncross JG (1994) p53 mutation, expression, and DNA ploidy in evolving gliomas: evidence for two pathways of progression. J Natl Cancer Inst 86:1011–1017
Okamoto Y, Di Patre PL, Burkhard C, Horstmann S, Jourde B, Fahey M, Schuler D, Probst-Hensch NM, Yasargil MG, Yonekawa Y, Lutolf UM, Kleihues P, Ohgaki H (2004) Population-based study on incidence, survival rates, and genetic alterations of low-grade diffuse astrocytomas and oligodendrogliomas. Acta Neuropathol (Berl) 108:49–56
Peraud A, Kreth FW, Wiestler OD, Kleihues P, Reulen HJ (2002) Prognostic impact of TP53 mutations and P53 protein overexpression in supratentorial WHO grade II astrocytomas and oligoastrocytomas. Clin Cancer Res 8:1117–1124
Perry JR (2001) Oligodendrogliomas: clinical and genetic correlations. Curr Opin Neurol 14:705–710
Ren ZP, Hedrum A, Ponten F, Nister M, Ahmadian A, Lundeberg J, Uhlen M, Ponten J (1996) Human epidermal cancer and accompanying precursors have identical p53 mutations different from p53 mutations in adjacent areas of clonally expanded non-neoplastic keratinocytes. Oncogene 12:765–773
Ren ZP, Ahmadian A, Ponten F, Nister M, Berg C, Lundeberg J, Uhlen M, Ponten J (1997) Benign clonal keratinocyte patches with p53 mutations show no genetic link to synchronous squamous cell precancer or cancer in human skin. Am J Pathol 150:1791–1803
Ren ZP, Sallstrom J, Sundstrom C, Nister M, Olsson Y (2000) Recovering DNA and optimizing PCR conditions from microdissected formalin-fixed and paraffin-embedded materials. Pathobiology 68:215–217
Reifenberger J, Reifenberger G, Liu L, James CD, Wechsler W, Collins VP (1994) Molecular genetic analysis of oligodendroglial tumors shows preferential allelic deletions on 19q and 1p. Am J Pathol 145:1175–1190
Reifenberger G, Louis DN (2003) Oligodendroglioma: toward molecular definitions in diagnostic neuro-oncology. J Neuropathol Exp Neurol 62:111–126
Smith JS, Alderete B, Minn Y, Borell TJ, Perry A, Mohapatra G, Hosek SM, Kimmel D, O’Fallon J, Yates A, Feuerstein BG, Burger PC, Scheithauer BW, Jenkins RB (1999) Localization of common deletion regions on 1p and 19q in human gliomas and their association with histological subtype. Oncogene 18:4144–4152
Smith JS, Jenkins RB (2000) Genetic alterations in adult diffuse glioma: occurrence, significance, and prognostic implications. Front Biosci 5:D213–231
Smith JS, Perry A, Borell TJ, Lee HK, O’Fallon J, Hosek SM, Kimmel D, Yates A, Burger PC, Scheithauer BW, Jenkins RB (2000) Alterations of chromosome arms 1p and 19q as predictors of survival in oligodendrogliomas, astrocytomas, and mixed oligoastrocytomas. J Clin Oncol 18:636–645
Walker C, Joyce KA, Thompson-Hehir J, Davies MP, Gibbs FE, Halliwell N, Lloyd BH, Machell Y, Roebuck MM, Salisbury J, Sibson DR, Du Plessis D, Broome J, Rossi ML (2001) Characterisation of molecular alterations in microdissected archival gliomas. Acta Neuropathol (Berl) 101:321–333
Watanabe T, Nakamura M, Kros JM, Burkhard C, Yonekawa Y, Kleihues P, Oghaki H (2002) Phenotype versus genotype correlation in oligodendrogliomas and low-grade diffuse astrocytomas. Acta Neuropathol 103:267–275
Acknowledgments
We thank Ms Inger Jonasson and Ms Lila Shokohideh at the Rudbeck Laboratory, Uppsala University, for their devoted assistance in this project, and Harry Smits for linguistic revision of the manuscript. This study is financially supported by grants from the Swedish Cancer Foundation (Z-PR, MN), the Uppsala University Hospital (AS) and the Lions Cancer Research Fund at the Uppsala University Hospital (Z-PR, AS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qu, M., Olofsson, T., Sigurdardottir, S. et al. Genetically distinct astrocytic and oligodendroglial components in oligoastrocytomas. Acta Neuropathol 113, 129–136 (2007). https://doi.org/10.1007/s00401-006-0142-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-006-0142-0