Abstract
Background
According to the current guidelines, implantable cardioverter-defibrillators (ICD) for primary prevention in patients with heart failure and reduced ejection fraction (HFrEF) should not be considered until optimal guideline-directed medical therapy (GDMT) has been achieved for a minimum of 3 months. Optimization of GDMT often needs time beyond 3 months after diagnosis. The aim of the Heart Failure Optimization Study (HF-OPT) is to evaluate the recovery of left ventricular function beyond 3 months after diagnosis of newly diagnosed HFrEF.
Methods
The HF-OPT multicenter study is comprised of two non-randomized phases (registry and study). During the first 90 days a wearable cardioverter-defibrillator (WCD) is prescribed and patients are enrolled in an observational pre-study registry. Registry subjects meeting inclusion criteria for the study portion at day 90 have ongoing left ventricular ejection fraction (LVEF) reassessment at 90, 180 and 360 days after the index hospital discharge, regardless of continued WCD use. Approximately 600 subjects will be enrolled in the study portion. Of those, one-third are anticipated to start the study phase at day 90 with reduced LVEF. The primary objective of this study is to observe the rate of recovery of LVEF > 35% between 90 and 180 days, while key secondary endpoints include mortality and WCD recorded arrhythmias and shocks.
Discussion
The HF-OPT study will provide important information on the rate of additional recovery of LVEF > 35%, between 90 and 180 days, in newly diagnosed HF with reduced LVEF patients being titrated with GDMT. The results of the study may impact indications for primary prophylactic ICD implantation.
Zusammenfassung
Hintergrund
Den aktuellen Leitlinien nach ist die primärprophylaktische Implantation eines implantierbaren Kardioverter-Defibrillators (ICD) bei Patienten mit Herzinsuffizienz und reduzierter linksventrikulärer Ejektionsfraktion (HFrEF) erst indiziert, wenn für mindestens 3 Monate eine optimierte leitliniengerechte medikamentöse Herzinsuffizienztherapie (LGMT) durchgeführt wurde. Die Optimierung der LGMT nach Erstdiagnose dauert allerdings häufig länger als 3 Monate. Ziel der Heart Failure Optimization Study (HF-OPT) ist es, die Erholung der linksventrikulären Funktion im Zeitraum jenseits von 3 Monaten nach Erstdiagnose einer HFrEF zu evaluieren.
Methoden
Die multizentrische HF-OPT-Studie besteht aus zwei nichtrandomisierten Phasen: einer Register- und einer sich daran anschließenden Studienphase. In den ersten 90 Tagen wird den Patienten ein tragbarer Kardioverter-Defibrillator („wearable cardioverter-defibrillator“ [WCD]) verschrieben und sie werden in ein Beobachtungsregister aufgenommen. Bei Erfüllung der Einschlusskriterien für die Studienphase an Tag 90 erfolgen weitere Bestimmungen der linksventrikulären Ejektionsfraktion (LVEF) 90, 180 und 360 Tage nach erstmaliger Klinikentlassung, unabhängig von einer weiteren WCD-Verordnung. Ziel ist der Einschluss von etwa 600 Patienten in die Studienphase. Es wird erwartet, dass davon ein Drittel bei Tag 90 mit reduzierter LVEF in die Studienphase eintritt. Primärer Endpunkt der Studie ist die Erholungsrate mit LVEF > 35 % zwischen Tag 90 und 180. Zu den wichtigsten sekundären Endpunkten zählen die Mortalität sowie die durch den WCD dokumentierten Rhythmusstörungen und Schockabgaben.
Diskussion
Die HF-OPT-Studie wird wertvolle Daten zur Erholung der LVEF auf > 35 % zwischen Tag 90 und 180 bei Patienten liefern nach Erstdiagnose einer HFrEF unter einer LGMT. Die Ergebnisse können die Indikationsstellung zur primärprophylaktischen ICD-Implantation beeinflussen.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Implantable cardioverter-defibrillators (ICDs) have been shown to reduce both arrhythmic and total mortality in stable heart failure (HF) patients [1] with reduced ejection fraction (EF). However, these implantable devices contain a certain level of risk, and according to the ACC/AHA/ESC guidelines ICDs for primary prevention HF patients should not be considered for implantation until optimal guideline-directed medical therapy (GDMT) has been achieved for a minimum of 3 months [2, 3]. As medication titration is challenging to achieve GDMT levels within 90 days after discharge, ICD implantation during this period does not meet the current GDMT standards.
Besides ICDs and medical therapy, another option of sudden cardiac death (SCD) protection frequently used after HF hospitalization is the wearable cardioverter defibrillator (WCD) [4]. WCDs have been commercially approved for use by the Food and Drug Administration (FDA) and have been CE-marked for Europe since 2001. Since a WCD is non-invasive it makes an ideal device for protecting HF patients in the case of a temporary high SCD risk, but recovery is still possible. After medications have been titrated to appropriate target doses and the EF has been given time to improve, the WCD can be removed and an assessment for ICD implantation due to ongoing SCD risk can be performed. Titration to therapeutic levels is generally not achieved within three months of initiation [5, 6] for some drugs known to improve ventricular dysfunction, and improvements in heart failure symptoms and ventricular function may occur after optimization of medical therapy over time. Recent findings of the PROLONG registry show that 33% of patients with non-improved low LVEF at 3 months may indeed improve LVEF to ≥ 35% by extending heart failure optimization therapy beyond the 3‑month waiting period [7].
The primary objective of this study is to observe the rate of recovery of left ventricular function (defined as EF > 35%) between this later time period of 90 and 180 days in newly diagnosed HF with reduced EF patients who were prescribed the WCD while being initiated on HF medications. This will be measured from LVEF at hospital discharge, and then regularly at 90, 180 and 360 days after WCD start, regardless of continued WCD use, prior LVEF improvement, or ICD implantation. While approximately one-third of the patients will likely experience LVEF recovery in the first 90 days [7], we hypothesize that an additional 5–10% of patients will experience EF improvement between 90 and 180 days as GDMT is achieved. This would be considered clinically significant in terms of the potentially avoided primary preventive ICD implantations.
Methods
Study design
The HF-OPT study is a multi-center prospective observational study of newly diagnosed HF with reduced EF patients to test the hypothesis that additional LVEF recovery occurs between 90 and 180 days as GDMT is achieved (ClinicalTrials.gov Identifier: NCT03016754). This study is being conducted according to US and international standards of good clinical practice and has been approved by all relevant medical ethics committees. A total of 67 centers with WCD prescribing physicians were selected for the study based upon expressed interest. All sites were in the US or Europe and included both academic and community centers.
Although the study phase does not start until day 90, all eligible, consenting patients will be entered into a registry at the start of WCD use. The pre-study registry will allow the collection of early (90 day) outcomes and data in those patients that are likely to be eligible for the study at day 90, or that are eligible but refuse the study at day 90. While the primary endpoint analysis is at day 180, subjects will be followed for 360 days. See Fig. 1 for study flow diagram. The study is sponsored by the WCD manufacturer (ZOLL, Pittsburgh, PA, USA).
Objectives
Primary endpoint
Descriptive or summary statistics of serial LVEF measurements will be used to analyze the rate of recovery of ventricular dysfunction following discharge. LVEF at 180 days will be compared to the means of LVEF at baseline and 90 days. The percentage of patients reaching the goal of LVEF > 35% will be compared at 90 days and 180 days. Also, the time taken to reach LVEF > 35% will be calculated for those who improve and compared between groups (based on baseline characteristics). The core lab determinations will be used to verify local estimates of LVEF and may be used in secondary analyses of the study data.
Descriptive statistics will be used to assess the degree GDMT (% of target dose) was reached at 90 and 180 days, and how GDMT relates to LVEF recovery. We will also assess how self-reported adherence to the GDMT regimen affects LVEF recovery and overall clinical improvement.
Secondary endpoint: treatments by the WCD or ICD
Effectiveness of the WCD in treating ventricular arrhythmias will be evaluated through descriptive means. Clinical outcomes and device data from any defibrillation or cardiac event will be reviewed for success as defined by appropriate detection and defibrillation, conversion out of the detected ventricular arrhythmia, and consciousness when medically evaluated after treatment.
For the safety endpoint, descriptive statistics will be used to analyze the incidence of unnecessary shocks from WCD and ICD in this population. Causes of prolonged false detections and lack of response button use in WCD users will be reviewed.
Secondary endpoint: incidence of all untreated sustained VT/VF events during WCD use not associated with a shock event
Descriptive statistics will be used to analyze the incidence of untreated sustained (≥ 30 s) episodes of VT or VF. All possible reasons for a lack of shock will be reviewed, including response button use, rate threshold settings, or ECG artifact. ECG recordings will be adjudicated by a committee independent of the sponsor.
Secondary endpoint: incidence of all sudden cardiac arrest (SCA) events
Descriptive statistics will be used to analyze the incidence and etiology of SCA from the beginning of WCD use to end of study. “Sudden Cardiac Death” will be defined as an unexpected, non-traumatic, non-self-inflicted fatality in otherwise stable subjects who die within 1 h of the onset of the terminal symptoms. Subjects dying more than 1 h after a sudden cardiac arrest (SCA) from a ventricular arrhythmia will be designed as non-sudden death due to ventricular arrhythmia. For unwitnessed deaths participants will meet the definition of sudden cardiac death if they are found dead within 24 h of being well, assuming there is no evidence of another cause of death during that time period. Autopsy results may be used when available.
Secondary endpoint: incidence and etiology of other WCD recorded arrhythmias, such as asystole and sustained (≥ 30 s) supra-ventricular arrhythmias
Descriptive statistics will be used to analyze the incidence of non-VT/VF events that are recorded by the WCD. Such arrhythmic events will be evaluated for correlation with the other endpoints. ECG recordings will be adjudicated by a committee independent of the sponsor.
Secondary endpoint: mortality
Descriptive statistics will be used to analyze the effect of WCDs on 180-day and 360-day mortality following discharge from HF. Kaplan-Meier curves and survival analysis will be performed both for aggregate mortality and independently for sudden and non-sudden cardiac deaths. Cause of mortality will be adjudicated by a committee independent of the sponsor. The influence of baseline characteristics and EF recovery status on mortality will be evaluated. Comparisons will be made using the Seattle Heart Failure model as a predictive tool of post-discharge mortality.
Safety endpoint: complication data from WCD or ICD
Descriptive statistics will be used to analyze the incidence of complications from WCDs and ICDs in this population. This will include inappropriate treatments, as well as any other complications that may lead to hospitalizations or death.
Population and recruitment
Eligibility and recruitment.
Patients were enrolled from March 2017 through January 2021. Prior to 90 days, eligible patients with the WCD are enrolled into a registry. Registry patients are enrolled during the first 30 days of WCD use. The population for this study group are subjects who had an LVEF ≤ 35% following hospitalization for newly diagnosed HF, and who have already been prescribed a WCD for 90 days. Main inclusion and exclusion criteria are listed in Table 1. The study target is to enroll approximately 1400 subjects in the registry portion in order to achieve 600 subjects for the study period at 90 days. Subjects will be enrolled from study sites in the US, Germany, Austria, and France.
Study procedures and follow-up
The study procedures timeline is outlined in Table 2.
Pre-study registry
Patient demographics, limited medical history, medications, laboratory results, imaging, invasive and non-invasive cardiac examinations were collected upon registry entry. WCD device records were interrogated to determine compliance with use, defibrillation events, and arrhythmia detection. Reasons for terminating WCD use were also collected (Table 2).
Study period
Follow-ups were made in person or via phone at days 90, 120, 150, 180, 270 and 360 post WCD start.
Data collection
Data will be collected at the investigational sites using the electronic data capture (EDC) system ClindexLIVE (Fortress Medical Systems, LLC, Hopkins, MN, USA). Data will be entered at the investigational sites by trained staff. Entered data will be reviewed by the site investigator, who will affirm its accuracy and completeness by electronic sign-off. The sponsor will manage and monitor the data for quality.
Statistical analysis
Sample size estimation
This dataset will be used to evaluate among all patients whether more than 5% of them will experience EF improvement between 90 and 180 days (n5 / n > 5%) (Fig. 2).
To calculate the number of samples (n), the authors first calculated n4, then calculated n based on the following formula:
where pwcd is the percentage of patients that will wear WCD beyond 90 days (n1 / (n1 + n2 + n3)), and pfollowup1 and pfollowup2 are the percentage of patients with proper follow up from 0–90 days and from 90–180 days, respectively.
To calculate sample size for n4, the authors performed a superiority test. The statistical hypothesis to be tested is:
vs:
The sample size of the superiority test is calculated as the following formula:
where p is the actual proportion of patients that will have EF improvement (n5 / n4). p0 is the baseline proportion and specified as 0.001 (close to 0). δ is the test margin, since the hypothesis is that more than 5% of them will experience EF improvement between 90 and 180 days (n5 / n > 5%), δ depends on pwcd as \(\delta =0.05/P_{\mathrm{wcd}}\). The authors also specify α as 0.05, β as 0.1.
As shown in Fig. 2, sample size (n) depends both on p, the actual proportion of patients that will have EF improvement (p = n5 / n4) and pwcd, the percentage of patients that will wear WCD beyond 90 days (\(p_{\mathrm{wcd}}=\) n1 / (n1 + n2 + n3)). In this study, the authors specify α as 0.05, β as 0.1. Based on their assumptions regarding patients that will have continued WCD wear from 90–180 days, and the proportion of WCD patients at 90 days to have further EF improvement, the authors plan to prescribe WCD to 1400 patients (initial registry sample size), in order to have approximately 600 patients eligible for the study at day 90.
Discussion
Previous clinical data reveals that the following questions remain controversial subjects in newly diagnosed HF patients:
-
1.
How often is optimal medical therapy achieved in a real-world setting?
-
2.
When to implant an ICD in a newly diagnosed patient?
-
3.
How often and when to measure LVEF recovery?
Titration to therapeutic levels is generally not achieved within 3 months of initiation. For some drugs known to improve ventricular dysfunction, improvements in heart failure symptoms and ventricular dysfunction may occur after optimization of medical therapy over time [8].
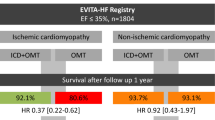
A predecessor of the HF-OPT Study was the Prolongation of Reverse remOdelling period to avoid untimely ICD impLantation in newly diagnOsed heart failure usiNG the wearable cardioverter defibrillator (PROLONG) study. The aim of the retrospective PROLONG study was to analyze evolution of LVEF after first diagnosis of a reduced LVEF ≤ 35% during elaborate optimization of heart failure therapy using the WCD to provide a secured prolongation of the observation period. They found 33% of patients that still had low LVEF at 3 month follow-up had improved during the prolongation period beyond 3 months after diagnosis, emphasizing the need for elaborate optimization of heart failure therapy [7]. Importantly, none of the patients not receiving an ICD experienced SCD or malignant arrhythmias during an extended follow-up of several years [9]. The authors sought to determine whether these results could be confirmed in a large, multi-center, prospective study, with set time-points for LVEF assessment and follow-up.
One potential reason for a slow or inadequate recovery of LVEF may be a lack of reaching target doses of GDMT during the first 3 months [10,11,12]. A retrospective study of Medicare patients linked to the National Cardiovascular Data ICD Registry showed a very low rate of GDMT being achieved in the 90 days before ICD implantation [13]. Only 61.1% had filled their prescriptions for an HF beta-blocker plus an angiotensin-converting enzyme inhibitor or angiotensin receptor blocker at least once during the 90 days before ICD implantation, and only 28.3% had a supply for ≥ 80% of the 90 days. Interestingly, those with the shortest duration of HF and most recent evaluation of LVEF were the least likely to receive GDMT. Death within 1 year occurred more frequently in those patients not receiving any GDMT even after adjusting for patient characteristics, HF severity, and comorbidities. More recent registries, such as the CHAMP-HF and CHECK-HF, have confirmed that GDMT is still being prescribed at lower than recommended doses [5, 6]. This may become even more of a problem as newer HF medications are approved and added on top of this list of GDMT medications. Also, just because the proper dosage may be prescribed does not mean the patient is taking it as directed.
Although LVEF is typically measured in the beginning, and usually at some follow-up period, it is unknown how often LVEF should be measured and what “recovery” of LV dysfunction means in terms of outcomes. To support our primary hypothesis that an additional 5% of patients will experience LVEF improvement between 90–180 days, LVEF measurement for all patients at 90 (±14), 180 (±14) and 360 (±14)-days after WCD start will be requested and recorded. For this study, LVEF > 35% will be considered as LVEF improved. This study is unique in that the LVEF trajectory, GDMT as a percent of target dose, and outcomes will be tracked for all subjects, regardless of ICD implantation. This will allow us to observe how the GDMT regimen, and self-reported adherence, relates to longitudinal LVEF recovery and clinical outcomes.
Conclusion
The HF-OPT study will provide important information on the rate of additional recovery of LVEF > 35%, especially between 90 and 180 days, in newly diagnosed HF with HFrEF patients being treated with GDMT, and will hopefully translate to better management of GDMT in newly diagnosed HF patients.
Change history
14 February 2023
An Erratum to this paper has been published: https://doi.org/10.1007/s00399-023-00928-5
References
Bardy GH, Lee KL, Mark DB, Poole JE, Packer DL, Boineau R, Domanski M, Troutman C, Anderson J, Johnson G, McNulty SE, Clapp-Channing N, Davidson-Ray LD, Fraulo ES, Fishbein DP, Luceri RM, Ip JH (2005) Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N Engl J Med 352:225–237
Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE Jr., Drazner MH, Fonarow GC, Geraci SA, Horwich T, Januzzi JL, Johnson MR, Kasper EK, Levy WC, Masoudi FA, McBride PE, McMurray JJ, Mitchell JE, Peterson PN, Riegel B, Sam F, Stevenson LW, Tang WH, Tsai EJ, Wilkoff BL (2013) ACCF/AHA guideline for the management of heart failure: executive summary: a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. Circulation 128:1810–1852
McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Bohm M, Burri H, Butler J, Celutkiene J, Chioncel O, Cleland JGF, Coats AJS, Crespo-Leiro MG, Farmakis D, Gilard M, Heymans S, Hoes AW, Jaarsma T, Jankowska EA, Lainscak M, Lam CSP, Lyon AR, McMurray JJV, Mebazaa A, Mindham R, Muneretto C, Piepoli FM, Price S, Rosano GMC, Ruschitzka F, Skibelund KA, Group ESCSD (2021) ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J 42:3599–3726
Feldman AM, Klein H, Tchou P, Murali S, Hall WJ, Mancini D, Boehmer J, Harvey M, Heilman MS, Szymkiewicz SJ, Moss AJ (2004) Use of a wearable defibrillator in terminating tachyarrhythmias in patients at high risk for sudden death: results of the WEARIT/BIROAD. Pacing Clin Electrophysiol 27:4–9
Greene SJ, Butler J, Albert NM, DeVore AD, Sharma PP, Duffy CI, Hill CL, McCague K, Mi X, Patterson JH, Spertus JA, Thomas L, Williams FB, Hernandez AF, Fonarow GC (2018) Medical therapy for heart failure with reduced ejection fraction: the CHAMP-HF registry. J Am Coll Cardiol 72:351–366
Brunner-La Rocca HP, Linssen GC, Smeele FJ, van Drimmelen AA, Schaafsma HJ, Westendorp PH, Rademaker PC, van de Kamp HJ, Hoes AW, Brugts JJ, Investigators C‑H (2019) Contemporary drug treatment of chronic heart failure with reduced ejection fraction: the CHECK-HF registry. JACC Heart Fail 7:13–21
Duncker D, Konig T, Hohmann S, Bauersachs J, Veltmann C (2017) Avoiding Untimely Implantable Cardioverter/Defibrillator Implantation by Intensified Heart Failure Therapy Optimization Supported by the Wearable Cardioverter/Defibrillator-The PROLONG Study. J Am Heart Assoc. https://doi.org/10.1161/JAHA.116.004512
Greene SJ, Fonarow GC, DeVore AD, Sharma PP, Vaduganathan M, Albert NM, Duffy CI, Hill CL, McCague K, Patterson JH, Spertus JA, Thomas L, Williams FB, Hernandez AF, Butler J (2019) Titration of medical therapy for heart failure with reduced ejection fraction. J Am Coll Cardiol 73:2365–2383
Mueller-Leisse J, Brunn J, Zormpas C, Hohmann S, Hillmann HAK, Eiringhaus J, Bauersachs J, Veltmann C, Duncker D (2021) Extended follow-up after wearable cardioverter-defibrillator period: the PROLONG-II study. Esc Heart Fail 8:5142–5148
Fonarow GC, Abraham WT, Albert NM, Stough WG, Gheorghiade M, Greenberg BH, O’Connor CM, Sun JL, Yancy CW, Young JB (2008) Dosing of beta-blocker therapy before, during, and after hospitalization for heart failure (from organized program to initiate lifesaving treatment in hospitalized patients with heart failure). Am J Cardiol 102:1524–1529
Gustafsson F, Schou M, Videbaek L, Nielsen T, Ulriksen H, Markenvard J, Svendsen TL, Ryde H, Vigholt E, Hildebrandt P, Danish Heart Failure Clinics N (2007) Treatment with beta-blockers in nurse-led heart failure clinics: titration efficacy and predictors of failure. European J of Heart Fail 9:910–916
Ouwerkerk W, Voors AA, Anker SD, Cleland JG, Dickstein K, Filippatos G, van der Harst P, Hillege HL, Lang CC, Ter Maaten JM, Ng LL, Ponikowski P, Samani NJ, van Veldhuisen DJ, Zannad F, Metra M, Zwinderman AH (2017) Determinants and clinical outcome of uptitration of ACE-inhibitors and beta-blockers in patients with heart failure: a prospective European study. Eur Heart J 38:1883–1890
Roth GA, Poole JE, Zaha R, Zhou W, Skinner J, Morden NE (2016) Use of guideline-directed medications for heart failure before cardioverter-defibrillator implantation. J Am Coll Cardiol 67:1062–1069
Acknowledgements
We would like to thank all the investigators and centers that participated in the HF-OPT study.
Funding
This research and the fees for open access publication were sponsored by ZOLL.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
B. Colley: Honoraria for lectures or consulting from Abbott and Medtronic. C. Perings: Honoraria for lectures or consulting from Bayer, Boehringer Ingelheim, Zoll. C. Veltmann: Honoraria for lectures or consulting from Abbott, AstraZeneca, Bayer, Biotronik, Boehringer Ingelheim, Boston Scientific, CVRx, Medtronic, Pfizer, and Zoll. D. Duncker: Honoraria for lectures, travel grants and/or a fellowship grant from Abbott, AstraZeneca, Bayer, Biotronik, Boehringer Ingelheim, Boston Scientific, Bristol Myers Squibb, CVRx, Medtronic, Microport, Pfizer, Zoll. R. Sanchez, M. Doering, S. Gummadi, M. Robertson, G. Shroff, and Heart Failure Optimization Study (HF-OPT) Investigators declare that they have no competing interests.
All procedures performed in studies involving human participants or on human tissue were in accordance with the ethical standards of the institutional and/or national research committee and with the 1975 Helsinki declaration and its later amendments or comparable ethical standards. The study was approved by the ethics committee of Hannover Medical School (No.: 7292) and by the ethics committee by each participating center. Clinical trial registration: NCT03016754. All patients gave written informed consent to HF-OPT.
Additional information
The authors R. Sanchez and D. Duncker contributed equally to the manuscript.

Scan QR code & read article online
The original online version of this article was revised: please note that only C. Veltmann writes on behalf of Heart Failure Optimization Study (HF-OPT) Investigators.
Appendix
Appendix
Trial organization
-
ZOLL Cardiac Management Solutions, Pittsburgh, PA
Participating countries
-
Austria,
-
France,
-
Germany,
-
USA.
Core laboratories
-
Echo core lab Europe: Department of Cardiology, Hannover Medical School, Carl-Neuberg-Str. 1, 30625 Hannover, Germany.
-
Echo core lab USA: Medpace, Inc., 5375 Medpace Way, Cincinnati, Ohio 45227 USA
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sanchez, R., Duncker, D., Colley, B. et al. The Heart Failure Optimization Study (HF-OPT): rationale and design. Herzschr Elektrophys 34, 52–58 (2023). https://doi.org/10.1007/s00399-022-00920-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00399-022-00920-5
Keywords
- Implantable cardioverter-defibrillator
- Wearable cardioverter-defibrillator
- Guideline-directed medical therapy
- Left ventricular ejection fraction