Abstract
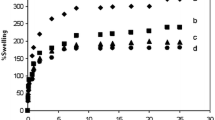
The complex formation of soluble poly (N-vinylimidazole) (PVIm) with trivalent metal ions in aqueous solution was studied by using UV–vis spectroscopy. Formation constants of PVIm–metal complexes were calculated by applying the “molar ratio” method. It was found that the interaction between PVIm and trivalent metal ions follows 4(base unit):1(metal ion) stoichiometry. The stability constants for the complexes of PVIm with trivalent transition-metal ions were in agreement with the Irving– William series. The biggest formation constant was found for the PVIm–Fe3+ complex system. The capacity of adsorption for these metal ions was investigated using cross-linked PVIm. Cross-linked PVIm hydrogels were prepared by irradiating binary mixture of N-vinylimidazole–water with a 60Co γ-ray source having a dose rate of 4.5 kGy/h. Adsorption studies were performed at different pH and metal ion concentrations at room temperature. It was observed that the same sequence for the metal ions was verified by adsorption studies.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 28 May 2001 Accepted: 29 July
Rights and permissions
About this article
Cite this article
Pekel, N., Savaş, H. & Güven, O. Complex formation and adsorption of V3+, Cr3+ and Fe3+ ions with poly(N-vinylimidazole). Colloid Polym Sci 280, 46–51 (2002). https://doi.org/10.1007/s003960200006
Issue Date:
DOI: https://doi.org/10.1007/s003960200006