Abstract
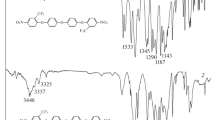
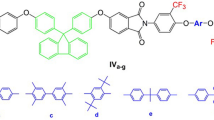
A novel fluorinated diamine monomer, 9,9-bis[4-(4-amino-2-trifluoromethylphenoxy)phenyl]fluorene (II) was prepared via the nucleophilic substitution reaction of 2-chloro-5-nitrobenzotrifluoride with 9,9-bis(4-hydroxyphenyl)fluorene in the presence of potassium carbonate, followed by catalytic reduction with hydrazine and Pd/C. Polyimides V were synthesized from diamine II and various aromatic dianhydrides III a-f via thermal imidization. These polymers had inherent viscosities ranging from 0.84 to 1.03 dL/g and were soluble in a variety of organic solvents such as NMP, DMAc, DMF, and DMSO, and some could even be dissolved in less polar solvents such as m-cresol, pyridine, and dioxane. Polyimide films V a-f had tensile strengths of 85–105 MPa, elongations to break of 7–9%, and initial moduli of 2.13–2.42 GPa. The glass transition temperature of these polymers were in the range of 277–331 °C, their 10% weight loss temperatures were in the range of 539–594 °C in nitrogen and above 544 °C in air, and their char yields at 800 °C in nitrogen ranged between 55–65 wt%. Compared with nonfluorinated polyimides VI, V showed better solubility and lower color intensity. Low dielectric constants (2.68–2.85 at 1 MHz) and low moisture absorptions (0.12–0.24 wt%) were also observed. In particular, V c-f afforded lightly-colored films, which had cutoff wavelengths lower than 385 nm and b* values ranging from 6 to 22.






Similar content being viewed by others
References
Feger C, Khojasteh MM, Htoo MS (eds)(1993) Advances in polyimide science and technology. Technomic, Lancaster, UK
Abadie MJM, Sillion B (eds)(1991) Polyimides and other high-temperature polymers. Elsevier, Amsterdam
Mittal KL (ed)(1984) Polyimides: synthesis, characterization, and application, vols I,II. Plenum, New York
Feger C, Khojasteh MM, McGrath JE (eds)(1989) Polyimides: materials, chemistry, and characterization. Elsevier, Amsterdam
Wilson D, Stenzenberger HD, Hergenrother PM (eds)(1990) Polyimides. Chapman and Hall, New York
Cassidy PE (1980) Thermally stable polymers. Marcel Dekker, New York
Yang HH (1989) Aromatic high-strength fibers. Wiley, New York
Dupont BS, Bilow N, U. S. Pat. 1986;4592925, and Landis AL, Naselow AB, U. S. Pat. 1987;4645824
Higashi K, Noda Y, Eur. Pat. 1986;240249, and Tamai S, Ohta M, Kawashima S, Oikawa H, Ohkoshi K, Yamaguchi A, Eur. Pat. 1987;234882
Matsuura T, Ando S, Sasaki S, Yamamoto F (1993) Electron Lett 29:2107
Ando S, Sawada T, Inoue Y (1993) Electron Lett 29:2143
Ghosh MK, Mittal KL (eds)(1996) Polyimides: fundamentals and applications. Marcel Dekker, New York
Li F, Fang S, Ge JJ, Honigfort PS, Chen JC, Harris FW, Cheng SZD (1999) Polymer 40:4571
Li F, Ge JJ, Honigfort PS, Fang S, Chen JC, Harris FW, Cheng SZD (1999) Polymer 40:4987
Wang CS, Yang RW (1997) J Appl Polym Sci 66:609
Yang CP, Hsiao SH, Hsu MF (2002) J Polym Sci A1 40:524
Xie K, Zhang SY, Liu JG, He MH, Yang SY (2001) J Polym Sci A1 39:2581
Yang CP, Lin JH (1993) J Polym Sci A1 31:2153
Hsiao SH, Yang CP, Lin WL (1999) Macromol Chem Physic 200:1428
Yang CP, Lin JH (1996) In: Salamone JC (ed) Polymeric materials encyclopedia, vol 9. CRC, Boca Raton, pp 6214
Acknowledgements
The authors are grateful to the National Science Council of the Republic of China for the support of this work (Grant NSC 91-2216-E-036-008).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, CP., Chiang, HC. Organosoluble and light-colored fluorinated polyimides based on 9,9-bis[4-(4-amino-2-trifluoromethylphenoxy)phenyl]fluorene and aromatic dianhydrides. Colloid Polym Sci 282, 1347–1358 (2004). https://doi.org/10.1007/s00396-003-1036-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-003-1036-5