Abstract.
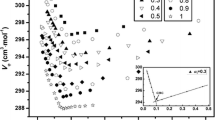
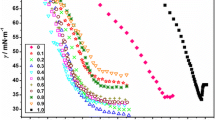
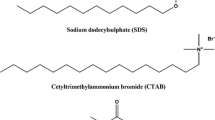
The dodecyltrimethylammonium bromide (DTAB)–didodecyldimethylammonium bromide (DDAB)–water system was studied to elucidate whether the mixed surfactant aggregates are mainly micelles or vesicles, and whether the surfactant–surfactant interaction is ideal or not. The regular solution theory of mixed micelles and the Israelachvili–Mitchell–Ninham (IMN) packing parameter of the aggregated mixture were used. The interaction between dissimilar surfactant species was found to be nonideal, with a positive value of the regular solution theory interaction parameter. The steric hindrance interaction parameter of Huang and Somasundaran theory indicated that the introduction of DTAB into DDAB layers was favoured at very low DTAB content, but became highly unfavourable when the proportion of DTAB increased. The aggregates were systematically much richer in DDAB than the solution of interaggregates. The IMN values systematically were compatible with flat surfaces, i.e., lamellar liquid crystals and vesicles. The results may be explained by a different composition of the inner and outer monolayers in the bilayer structure of the vesicles.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Proverbio, Z.E., Schulz, P.C. & Puig, J.E. Aggregation of the aqueous dodecyltrimethylammonium bromide–didodecyldimethylammonium bromide system at low concentration. Colloid Polym Sci 280, 1045–1052 (2002). https://doi.org/10.1007/s00396-002-0731-y
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00396-002-0731-y