Abstract
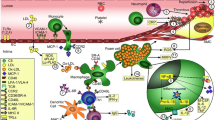
A chronic (auto)immune response is the critical mechanism in atherosclerosis. Interleukin-17A is a pivotal effector cytokine, which modulates immune cell trafficking and initiates inflammation in (auto)immune and infectious diseases. However, expression of IL-17A in the context of human atherosclerosis has hardly been explored. Carotid artery plaques were collected from 79 patients undergoing endarterectomy. Patients were grouped according to their symptomatic status (TIA, stroke), plaque morphology and medication. Quantitative RT-PCR was used to analyze tissue inflammation and immunohistochemistry to assess cellular source of IL-17A expression and lesion morphology. Carotid plaques from patients with ischemic symptoms were characterized by a highly activated inflammatory milieu including accumulation of T cells (p = 0.04) and expression of IL-6 and VCAM1 (p = 0.02, 0.01). Expression of IL-17A and its positive regulators IL-21 and IL-23 was present in atherosclerotic lesions, significantly upregulated in atheromas of symptomatic patients (p = 0.005, 0.004, 0.03), and expression of IL-17A and IL-21 showed a strong correlation (p = 0.002, r = 0.52). The cellular sources of lesional IL-17A expression are T cells, macrophages, B cells and plasma cells. Vulnerable/ruptured (complicated) plaques were significantly associated with IL-17A expression levels (p = 0.003). In addition, IL-17A showed a marked negative correlation with the potent anti-inflammatory/atheroprotective cytokine IL-10 (p = 0.0006, r = −0.46). Furthermore, treatment with a HMG-CoA reductase inhibitor or acetylsalicylic acid showed reduced levels of IL-21, IL-23 and VCAM1 (all p < 0.05), but did not influence IL-17A. The association of IL-17A with ischemic symptoms and vulnerable plaque characteristics suggests that the pro-inflammatory cytokine IL-17A may contribute to atherosclerosis und plaque instability.





Similar content being viewed by others
References
Beckman JA, Ganz J, Creager MA, Ganz P, Kinlay S (2001) Relationship of clinical presentation and calcification of culprit coronary artery stenoses. Arterioscler Thromb Vasc Biol 21:1618–1622
Bose D, von Birgelen C, Zhou XY, Schmermund A, Philipp S, Sack S, Konorza T, Mohlenkamp S, Leineweber K, Kleinbongard P, Wijns W, Heusch G, Erbel R (2008) Impact of atherosclerotic plaque composition on coronary microembolization during percutaneous coronary interventions. Basic Res Cardiol 103:587–597
Chapman MJ (2007) From pathophysiology to targeted therapy for atherothrombosis: a role for the combination of statin and aspirin in secondary prevention. Pharmacol Ther 113:184–196
Cordell JL, Falini B, Erber WN, Ghosh AK, Abdulaziz Z et al (1984) Immunoenzymatic labeling of monoclonal antibodies using immune complexes of alkaline phosphatase and monoclonal anti-alkaline phosphatase (APAAP complexes). J Histochem Cytochem 32:219–229
Davies MJ (1990) A macro and micro view of coronary vascular insult in ischemic heart disease. Circulation 82:38–46
de Boer OJ, van der Meer JJ, Teeling P, van der Loos CM, Idu MM et al (2010) Differential expression of interleukin-17 family cytokines in intact and complicated human atherosclerotic plaques. J Pathol 220:499–508
Erbel C, Chen L, Bea F, Wangler S, Celik S et al (2009) Inhibition of IL-17A attenuates atherosclerotic lesion development in ApoE-deficient mice. J Immunol 183:8167–8175
Erbel C, Sato K, Meyer FB, Kopecky SL, Frye RL et al (2007) Functional profile of activated dendritic cells in unstable atherosclerotic plaque. Basic Res Cardiol 102:123–132
Fuster V, Badimon L, Badimon JJ, Chesebro JH (1992) The pathogenesis of coronary artery disease and the acute coronary syndromes (1). N Engl J Med 326:242–250
Gravallese EM (2003) Osteopontin: a bridge between bone and the immune system. J Clin Invest 112:147–149
Hansson GK, Libby P (2006) The immune response in atherosclerosis: a double-edged sword. Nat Rev Immunol 6:508–519
Huang H, Virmani R, Younis H, Burke AP, Kamm RD et al (2001) The impact of calcification on the biomechanical stability of atherosclerotic plaques. Circulation 103:1051–1056
Jander S, Sitzer M, Schumann R, Schroeter M, Siebler M et al (1998) Inflammation in high-grade carotid stenosis: a possible role for macrophages and T cells in plaque destabilization. Stroke 29:1625–1630
Kamimura M, Viedt C, Dalpke A, Rosenfeld ME, Mackman N et al (2005) Interleukin-10 suppresses tissue factor expression in lipopolysaccharide-stimulated macrophages via inhibition of Egr-1 and a serum response element/MEK-ERK1/2 pathway. Circ Res 97:305–313
Kendrach MG, Kelly-Freeman M (2004) Approximate equivalent rosuvastatin doses for temporary statin interchange programs. Ann Pharmacother 38:1286–1292
Kleinbongard P, Heusch G, Schulz R (2010) TNFalpha in atherosclerosis, myocardial ischemia/reperfusion and heart failure. Pharmacol Ther 127:295–314
Konstandin MH, Aksoy H, Wabnitz GH, Volz C, Erbel C et al (2009) Beta2-integrin activation on T cell subsets is an independent prognostic factor in unstable angina pectoris. Basic Res Cardiol 104:341–351
Korn T, Bettelli E, Gao W, Awasthi A, Jager A et al (2007) IL-21 initiates an alternative pathway to induce proinflammatory T(H)17 cells. Nature 448:484–487
Langrish CL, Chen Y, Blumenschein WM, Mattson J, Basham B et al (2005) IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J Exp Med 201:233–240
Matsui Y, Rittling SR, Okamoto H, Inobe M, Jia N et al (2003) Osteopontin deficiency attenuates atherosclerosis in female apolipoprotein E-deficient mice. Arterioscler Thromb Vasc Biol 23:1029–1034
Naylor AR (2009) The importance of initiating “best medical therapy” and intervening as soon as possible in patients with symptomatic carotid artery disease: time for a radical rethink of practice. J Cardiovasc Surg (Torino) 50:773–782
Stary HC (2000) Natural history and histological classification of atherosclerotic lesions: an update. Arterioscler Thromb Vasc Biol 20:1177–1178
Van Herck JL, De Meyer GR, Martinet W, Bult H, Vrints CJ, Herman AG (2010) Proteasome inhibitor bortezomib promotes a rupture-prone plaque phenotype in ApoE-deficient mice. Basic Res Cardiol 105:39–50
Van Herck JL, De Meyer GR, Martinet W, Salgado RA, Shivalkar B, De Mondt R, Van De Ven H, Ludwig A, Van Der Veken P, Van Vaeck L, Bult H, Herman AG, Vrints CJ (2010) Multi-slice computed tomography with N1177 identifies ruptured atherosclerotic plaques in rabbits. Basic Res Cardiol 105:51–59
Virmani R, Burke AP, Farb A, Kolodgie FD (2006) Pathology of the vulnerable plaque. J Am Coll Cardiol 47:C13–C18
Waxman S, Ishibashi F, Muller JE (2006) Detection and treatment of vulnerable plaques and vulnerable patients: novel approaches to prevention of coronary events. Circulation 114:2390–2411
Weaver CT, Hatton RD, Mangan PR, Harrington LE (2007) IL-17 family cytokines and the expanding diversity of effector T cell lineages. Annu Rev Immunol 25:821–852
Zhang C (2008) The role of inflammatory cytokines in endothelial dysfunction. Basic Res Cardiol 103:398–406
Acknowledgments
This work was supported by a research stipend from the German Society of Cardiology to Dr. C. Erbel.
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Erbel, C., Dengler, T.J., Wangler, S. et al. Expression of IL-17A in human atherosclerotic lesions is associated with increased inflammation and plaque vulnerability. Basic Res Cardiol 106, 125–134 (2011). https://doi.org/10.1007/s00395-010-0135-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00395-010-0135-y