Abstract
Purpose
Soybean glycinin (11S) and β-conglycinin (7S) are major antigenic proteins in soybean and can induce a variety of allergic reactions in the young animals. This study aimed to investigate the effect of 7S and 11S allergens on the intestine of piglets.
Methods
Thirty healthy 21-day-old weaned “Duroc × Long White × Yorkshire” piglets were randomly divided into three groups fed with the basic diet, the 7S supplemented basic diet, or the 11S supplemented basic diet for 1 week. Allergy markers, intestinal permeability, oxidative stress, and inflammatory reactions were detected, and we observed different sections of intestinal tissue. The expressions of genes and proteins related to NOD-like receptor thermal protein domain associated protein 3 (NLRP-3) signaling pathway were detected by IHC, RT-qPCR, and WB.
Results
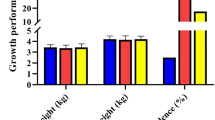
Severe diarrhea and decreased growth rate were observed in the 7S and 11S groups. Typical allergy markers include IgE production and significant elevations of histamine and 5-hydroxytryptamine (5-HT). More aggressive intestinal inflammation and barrier dysfunction were observed in the experimental weaned piglets. In addition, 7S and 11S supplementation increased the levels of 8-hydroxy-2 deoxyguanosine (8-OHdG) and nitrotyrosine, triggering oxidative stress. Furthermore, higher expression levels of NLRP-3 inflammasome ASC, caspase-1, IL-1β, and IL-18 were observed in the duodenum, jejunum, and ileum.
Conclusion
We confirmed that 7S and 11S damaged the intestinal barrier of weaned piglets and may be associated with the onset of oxidative stress and inflammatory response. However, the molecular mechanism underlying these reactions deserves further study.







Similar content being viewed by others
Data availability
The data used to support the findings of this study are included within the article.
References
Kim S, Lee J, Kwon Y, Kim W, Jung G, Kim D, Lee C, Lee Y, Kim M, Kim Y et al (2013) Introduction and nutritional evaluation of germinated soy germ. Food Chem 136(2):491–500. https://doi.org/10.1016/j.foodchem.2012.08.022
Kim D, Quang T, Yoon C, Ngan N, Lim S, Lee S, Kim Y (2016) Anti-neuroinflammatory activities of indole alkaloids from kanjang (Korean fermented soy source) in lipopolysaccharide-induced BV2 microglial cells. Food Chem 213(2016):69–75. https://doi.org/10.1016/j.foodchem.2016.06.068
Suo H, Feng X, Zhu K, Wang C, Zhao X, Kan J (2015) Shuidouchi (Fermented Soybean) fermented in different vessels attenuates HCl/ethanol-induced gastric mucosal injury. Molecules 20(11):19748–19763. https://doi.org/10.3390/molecules201119654
He L, Han M, Qiao S, He P, Li D, Li N (2015) Soybean antigen proteins and their intestinal sensitization activities. Curr Protein Pept Sci 16:613–621. https://doi.org/10.2174/1389203716666150630134602
Krishnan H, Kim W, Jang S, Kerley M (2009) All three subunits of soybean beta-conglycinin are potential food allergens. J Agric Food Chem 57(3):938–943. https://doi.org/10.1021/jf802451g
Tang C (2017) Emulsifying properties of soy proteins: a critical review with emphasis on the role of conformational flexibility. Crit Rev Food Sci Nutr 57(12):2636–2679. https://doi.org/10.1080/10408398.2015.1067594
Yaklich RW (2001) β-Conglycinin and glycinin in high-protein soybean seeds. J Agric Food Chem 49(2):729–735. https://doi.org/10.1021/jf001110s
Chen J, Wang J, Song P, Ma X (2014) Determination of glycinin in soybean and soybean products using a sandwich enzyme-linked immunosorbent assay. Food Chem 162:27–33. https://doi.org/10.1016/j.foodchem.2014.04.065
Dick K, Pattang A, Hooker J, Nissan N, Sadowski M, Barnes B, Tan LH, Burnside D, Phanse S, Aoki H, Babu M, Dehne F, Golshani A, Cober ER, Green JR, Samanfar B (2021) Human-soybean allergies: elucidation of the seed proteome and comprehensive protein-protein interaction prediction. J Proteome Res 20(11):4925–4947. https://doi.org/10.1021/acs.jproteome.1c00138
He Y, Liang J, Dong X, Liu H, Yang Q, Zhang S, Chi S, Tan B (2022) Soybean β-conglycinin and glycinin reduced growth performance and the intestinal immune defense and altered microbiome in juvenile pearl gentian groupers Epinephelus fuscoguttatus♀ × Epinephelus lanceolatus♂. Anim Nutr 8(9):193–203. https://doi.org/10.1016/j.aninu.2021.11.0012405-6545
Zheng S, Qin G, Tian H, Sun Z (2014) Role of soybean β-conglycinin subunits as potential dietary allergens in piglets. Vet J 199(3):434–438. https://doi.org/10.1016/j.tvjl.2013.11.020
Moneret-Vautrin D, Morisset M, Flabbee J, Beaudouin E, Kanny G (2005) Epidemiology of life-threatening and lethal anaphylaxis: a review. Allergy 60:443–451. https://doi.org/10.1111/j.1398-9995.2005.00785.x
Peng C, Cao C, He M, Shu Y, Tang X, Wang Y (2018) Soybean Glycinin- and β-conglycinin-induced intestinal damage in piglets via the p38/JNK/NF-κB signaling pathway. J Agric Food Chem 66:9534–9541. https://doi.org/10.1021/acs.jafc.8b03641
Aviello G, Knaus U (2017) ROS in gastrointestinal inflammation: rescue or sabotage? Br J Pharmaco 174:1704–1718. https://doi.org/10.1111/bph.13428
Kinoshita Y, Ishimura N, Ishihara S (2018) Advantages and disadvantages of long-term proton pump inhibitor use. J Neurogastroenterol Motil 24:182–196. https://doi.org/10.5056/jnm18001
Tourkochristou E, Aggeletopoulou I, Konstantakis C, Triantos C (2019) Role of NLRP-3 inflammasome in inflammatory bowel diseases. World J Gastroenterol 25:4796–4804. https://doi.org/10.3748/wjg.v25.i33.4796
Zhen Y, Zhang H (2019) NLRP-3 inflammasome and inflammatory bowel disease. Front Immunol 10:276. https://doi.org/10.3389/fimmu.2019.00276
Ma H, Shu Q, Li Z, Song X, Xu H (2023) Formaldehyde aggravates allergic contact dermatitis by facilitating NLRP3 inflammasome activation in macrophages. Int Immunopharmacol 117:109904. https://doi.org/10.1016/j.intimp.2023.109904
Wang L, Sun Z, Xie W, Peng C, Ding H, Li Y (2022) 11S glycinin up-regulated NLRP-3-induced pyroptosis by triggering reactive oxygen species in porcine intestinal epithelial cells. Front Vet Sci 9:890978. https://doi.org/10.3389/fvets.2022.890978
Parrish A, Boudaud M, Kuehn A, Ollert M, Desai M (2022) Intestinal mucus barrier: a missing piece of the puzzle in food allergy. Trends Mol Med 28(1):36–50. https://doi.org/10.1016/j.molmed.2021.10.004
Acevedo N, Alashkar Alhamwe B, Caraballo L, Ding M, Ferrante A, Garn H, Garssen J, Hii CS, Irvine J, Llinás-Caballero K, López J, Miethe S, Perveen K, Pogge von Strandmann E, Sokolowska M, Potaczek DP (2021) Perinatal and early-life nutrition, epigenetics, and allergy. Nutrients 13(3):724. https://doi.org/10.3390/nu13030724
Thorpe D, Stringer A, Butler R (2013) Chemotherapy-induced mucositis: the role of mucin secretion and regulation, and the enteric nervous system. Neurotoxicology 38:101–105. https://doi.org/10.1016/j.neuro.2013.06.007
Zhang KY, Hornef MW, Dupont A (2015) The intestinal epithelium as guardian of gut barrier integrity. Cell Microbiol 17:1561–1569. https://doi.org/10.1111/cmi.12501
Shi L, Xun W, Peng W, Hu H, Cao T, Hou G (2020) Effect of the single and combined use of curcumin and piperine on growth performance, intestinal barrier function, and antioxidant capacity of weaned Wuzhishan Piglets. Front Vet Sci 7:418. https://doi.org/10.3389/fvets.2020.00418
Henrichs B, Brost K, Hayes C, Campbell J, Drackley J (2021) Effects of spray-dried bovine plasma protein in milk replacers fed at a high plane of nutrition on performance, intestinal permeability, and morbidity of Holstein calves. J Dairy Sci 104:7856–7870. https://doi.org/10.3168/jds.2020-20104
Choi J, Jin S, Park B, Kim H, Khanal T (2013) Cultivated ginseng inhibits 2,4-dinitrochlorobenzene-induced atopic dermatitis-like skin lesions in NC/Nga mice and TNF-α/IFN-γ-induced TARC activation in HaCaT cells. Food Chem Toxicol 56:195–203. https://doi.org/10.1016/j.fct.2013.02.037
Wang Y, Lin J, Shu J, Li H, Ren Z (2018) Oxidative damage and DNA damage in lungs of an ovalbumin-induced asthmatic murine model. J Thorac Dis 10:4819–4830. https://doi.org/10.21037/jtd.2018.07.74
Meng Y, Xu X, Xie G, Zhang Y, Chen S, Qiu Y (2022) Alkyl organophosphate flame retardants (OPFRs) induce lung inflammation and aggravate OVA-simulated asthmatic response via the NF-кB signaling pathway. Environ Int 163:107209. https://doi.org/10.1016/j.envint.2022.107209
Wang J (2016) Utility of component diagnostic testing in guiding oral food challenges to milk and egg. Allergy Asthma Proc 37(6):439–442. https://doi.org/10.2500/aap.2016.37.3997
Qu Q, Song X, Lin L, Gong Z, Xu W, Xiao W (2022) L-theanine modulates intestine-specific immunity by regulating the differentiation of CD4+ T cells in ovalbumin-sensitized mice. J Agric Food Chem 70(47):14851–14863. https://doi.org/10.1021/acs.jafc.2c06171
Johansson S (2016) The discovery of IgE. J Allergy Clin Immunol 137:1671–1673. https://doi.org/10.1016/j.jaci.2016.04.004
Gould H, Sutton B (2008) IgE in allergy and asthma today. Nat Rev Immunol 8(3):205–217. https://doi.org/10.1038/nri2273
Mandola A, Nozawa A, Eiwegger T (2019) Histamine, histamine receptors, and anti-histamines in the context of allergic responses. LymphoSign J 6:35–51. https://doi.org/10.14785/lymphosign-2018-0016
Bischoff S (2009) Physiological and pathophysiological functions of intestinal mast cells. Semin Immunopathol 31(2):185–205. https://doi.org/10.1007/s00281-009-0165-4
Shulpekova Y, Nechaev V, Popova I, Deeva T, Kopylov A, Malsagova K, Kaysheva A, Ivashkin V (2021) Food intolerance: the role of histamine. Nutrients 13(9):3207. https://doi.org/10.3390/nu13093207
Ma B, Athari SS, Mehrabi Nasab E, Zhao L (2021) PI3K/AKT/mTOR and TLR4/MyD88/NF-κB signaling inhibitors attenuate pathological mechanisms of allergic asthma. Inflammation 44(5):1895–1907. https://doi.org/10.1007/s10753-021-01466-3
Vivinus-Nébot M, Dainese R, Anty R, Saint-Paul M, Nano J, Gonthier N (2012) Combination of allergic factors can worsen diarrheic irritable bowel syndrome: role of barrier defects and mast cells. Am J Gastroenterol 107(1):75–81. https://doi.org/10.1038/ajg.2011.315
Fischer R, Maier O (2015) Interrelation of oxidative stress and inflammation in neurodegenerative disease: role of TNF. Oxid Med Cell Longev 2015:610813. https://doi.org/10.1155/2015/610813
Xie J, Li B, Yao B, Zhang P, Wang L, Lu H, Song X (2020) Transforming growth factor-β1-regulated Fas/FasL pathway activation suppresses nucleus pulposus cell apoptosis in an inflammatory environment. Biosci Rep 40(2):BSR20191726. https://doi.org/10.1042/BSR20191726
Naji K, Al-Shaibani E, Alhadi F, Al-Soudi S, D’souza M (2017) Hepatoprotective and antioxidant effects of single clove garlic against CCl-induced hepatic damage in rabbits. BMC Complem Altern M 17:411. https://doi.org/10.1186/s12906-017-1916-8
Ruart M, Chavarria L, Camprecios G, Suarez-Herrera N, Montironi C, Guixe-Muntet S, Bosch J (2019) Impaired endothelial autophagy promotes liver fibrosis by aggravating the oxidative stress response during acute liver injury. J Hepatol 70(3):458–469. https://doi.org/10.1016/j.jhep.2018.10.015
Hong M, Xu A, Zhou H, Wu L, Randers-Pehrson G, Santella RM, Yu Z, Hei TK (2010) Mechanism of genotoxicity induced by targeted cytoplasmic irradiation. Br J Cancer 103(8):1263–1268. https://doi.org/10.1038/sj.bjc.6605888
Zhuang A, Yang C, Liu Y, Tan Y, Bond S, Walker S, Sikora T, Laskowski A (2021) SOD2 in skeletal muscle: new insights from an inducible deletion model. Redox Biol 47(undefined):102135. https://doi.org/10.1016/j.redox.2021.102135
Yuan L, Mishra R, Patel H, Alanazi S, Wei X, Ma Z, Garrett J (2020) BRAF mutant melanoma adjusts to BRAF/MEK inhibitors via dependence on increased antioxidant SOD2 and increased reactive oxygen species levels. Cancers (Basel). https://doi.org/10.3390/cancers12061661
Zhang H, Zhao F, Gai X, Cai J, Zhang X, Chen X, Zhu Y, Zhang Z (2022) Astilbin attenuates apoptosis induced by cadmium through oxidative stress in carp (Cyprinus carpio L.) head kidney lymphocyte. Fish Shellfish Immunol 125:230–237. https://doi.org/10.1016/j.fsi.2022.05.021
Wang F, Hong Y, Jiang W, Wang Y, Chen M, Zang D, Zhu Q (2022) ROS-mediated inflammatory response in liver damage via regulating the Nrf2/HO-1/NLRP3 pathway in mice with trichloroethylene hypersensitivity syndrome. J Immunotoxicol 19(1):100–108. https://doi.org/10.1080/1547691X.2022.2111003
Lee T, Song H, Park C (2014) Role of inflammasome activation in development and exacerbation of asthma. Asia Pac Allergy 4:187–196. https://doi.org/10.5415/apallergy.2014.4.4.187
Liston A, Masters S (2017) Homeostasis-altering molecular processes as mechanisms of inflammasome activation. Nat Rev Immunol 17(3):208–214. https://doi.org/10.1038/nri.2016.151
Fernandes-Alnemri T, Wu J, Yu J, Datta P, Miller B, Jankowski W, Rosenberg S, Zhang J (2007) The pyroptosome: a supramolecular assembly of ASC dimers mediating inflammatory cell death via caspase-1 activation. Cell Death Differ 14:1590–1604. https://doi.org/10.1038/sj.cdd.4402194
Rajatava B, Sarah W, Suniti B, Zindl C (2015) IL-1 signaling modulates activation of STAT transcription factors to antagonize retinoic acid signaling and control the T(H)17 cell-iT(reg) cell balance. Nat Immunol 16(3):286–295. https://doi.org/10.1038/ni.3099
Kim D, Jung W, Kim M, Lee H, Youn H, Seon J, Lee H, Lee J (2014) Genistein inhibits pro-inflammatory cytokines in human mast cell activation through the inhibition of the ERK pathway. Int J Mol Med 34(6):1669–1674. https://doi.org/10.3892/ijmm.2014.1956
Acknowledgements
We gratefully acknowledge the professors at the College of Animal Science and Technology of Anhui Agricultural University.
Funding
This research was supported by the National Natural Science Foundation of China (No. 31972750).
Author information
Authors and Affiliations
Contributions
JW coordinated the project funding and technical support. LW and CP conceived and designed this research. LW conducted experiments and wrote manuscript. WL, SX, and SW collected experimental samples. JW, DH, YL, SF, XW, and CZ reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
All authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical approval
All experiments were performed in accordance with the Regulations on the Administration of Laboratory Animals (Ministry of Science and Technology of China; revised in June 2004) and approved by the Ethics Committee of Anhui Agricultural University (Approval number 20210512).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, L., Li, W., Xin, S. et al. Soybean glycinin and β-conglycinin damage the intestinal barrier by triggering oxidative stress and inflammatory response in weaned piglets. Eur J Nutr 62, 2841–2854 (2023). https://doi.org/10.1007/s00394-023-03188-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-023-03188-8