Abstract
Purpose
To assess the association between a multi-dimensional Macronutrient Quality Index (MQI) and the risk of cardiovascular disease (CVD) in a Mediterranean cohort.
Methods
Prospective analyses among 18,418 participants (mean age 36 years, 60.8% women) of the Seguimiento Universidad de Navarra (SUN) cohort. Dietary intake information was obtained through a validated semi-quantitative food-frequency questionnaire (FFQ). The MQI (expressing high-quality macronutrient intake) was calculated based on three previously reported quality indices: the Carbohydrate Quality Index (CQI), the Fat Quality Index (FQI), and the Healthy Plate Protein source Quality Index (HPPQI). Adherence to the Mediterranean diet (MedDiet) and Provegetarian Diet was evaluated using the Trichopoulou index and the score proposed by Martínez-González, respectively. CVD was defined as new-onset stroke, myocardial infarction, or CVD death.
Results
After a median follow-up time of 14 years (211,744 person-years), 171 cases of CVD were identified. A significant inverse association was found between the MQI and CVD risk with multivariable-adjusted HR for the highest vs. the lowest quartile of 0.60 (95% IC, 0.38–0.96; Ptrend = 0.024).
Conclusion
In this Mediterranean cohort, we found a significant inverse relationship between a multidimensional MQI (expressing high-quality macronutrient intake) and a lower risk of CVD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cardiovascular disease (CVD) was the leading cause of death in the world, contributing to around 18 million deaths in 2019 [1]. In Europe, it is estimated that CVD is responsible for one in four deaths [2], accounting for 2.2 million deaths in women and 1.9 million in men [3]. These numbers represent a global health concern and highlight the need for prevention strategies.
Reducing the risk of major modifiable factors such as unhealthy diet, smoking, alcohol consumption, and physical inactivity represents one of the main strategies for the prevention of non-communicable diseases [4, 5]. The American College of Cardiology/American Heart Association (ACC/AHA) emphasizes the importance of healthy dietary patterns rather than their isolated components [6], suggesting a greater magnitude of beneficial effects [7]. Healthy dietary patterns are characterized by high consumption of fruits, vegetables, legumes, whole grains, and fish while limiting the consumption of whole dairy products, red meats, processed meats, and sugars [8]. Diet quality is widely used in nutritional epidemiology to develop national nutrition guidelines, evaluate compliance to predefined healthy dietary patterns, or assess the risk of developing chronic diseases such as CVD [9, 10].
In the last few decades, the dietary approach used for CVD prevention has traditionally focused on diets with low fat intake—particularly on reduction in saturated fatty acids (SFA) and cholesterol—and promotion of high intake of unsaturated fatty acids—such as polyunsaturated fatty acids (PUFA) [11,12,13,14]. However, the emerging evidence on fats contradicts such approach [15,16,17,18]. Appropriate distribution of macronutrients with respect to total energy intake (45–65%, 10–35%, and 20–35% for carbohydrates, proteins, and fats, respectively), has been associated with lower risk of chronic diseases and adequate micronutrient intake [19]. However, macronutrient quality is likely to be even more important than macronutrient quantity [20, 21].
Most of the existing research on macronutrient quality and CVD has focused on their isolated effects, particularly on fat quality [22, 23], rather than using a more comprehensive approach. To cover this gap, our team created a novel global index, namely the Macronutrient Quality Index (MQI), based on the best scientific evidence available at that moment to evaluate its association with all-cause mortality in the “Seguimiento Universidad de Navarra (SUN)” (University of Navarra Follow-Up) Project [24]. The MQI was constructed upon three equally weighted sub-indexes, one for each macronutrient class (proteins, carbohydrates, and fats), and is an example of an a priori dietary quality index. These indexes are considered useful tools and are widely used in research and clinical practice [25]. To our knowledge, there are no prospective studies that have investigated the association between the dietary quality of macronutrients and the risk of CVD using the MQI. Hence, we aimed to prospectively assess the association of MQI and CVD risk, with a median follow-up time of 14 years, in the SUN cohort.
Materials and methods
Study population
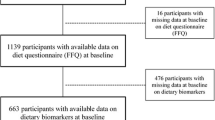
The SUN Project is a continuous, dynamic, multipurpose and prospective cohort [26]. All participants are university graduates, which reduces the potential confounding related to educational level and socioeconomic status, and increases the validity and reliability of the information [27]. Self-reported mailed and electronic questionnaires are collected at baseline and every 2 years to gather information related to socio-demographics, lifestyle, and medical history, including mortality and its causes. Participants who did not respond to any of the five follow-up mailings were contacted by email or phone. By December 2019, a total of 22,894 participants were enrolled in the SUN cohort. For these analyses, we excluded 341 participants recruited after March 2017 (to ensure a minimum follow-up of 2 years); 350 participants with prevalent CVD; 2,114 individuals with energy intake outside of predefined limits (men: < 800 or > 4,000 kcal/day; women: < 500 or > 3,500 kcal/day) [28]; and 1,671 participants without follow-up (retention rate 91.7%). Therefore, 18,418 participants were the basis for our analyses (Fig. 1).
Bioethics
Participants received written information about the information collected in the questionnaires, their privacy rights to protect their data, and future feedback of the finding of the project from the research team. Potential candidates were additionally informed about their right to refuse to participate or withdraw from the study at any time without reprisal, according to the ethical standards of the Declaration of Helsinki. Voluntary completion of the baseline questionnaire was considered as informed consent for participation in the study. The Research Ethics Committee of the University of Navarra approved the study. The SUN cohort is registered at clinicaltrials.gov as NCT02669602.
Dietary assessment
Baseline dietary information and after 10 years of follow-up was evaluated using a self-administered food frequency questionnaire (FFQ). The questionnaire has been previously validated, and the reproducibility for the majority of foods and nutrients is good [29,30,31]. The FFQ consists of 136 items and includes 9 food groups: (1) dairy products, (2) eggs, meat, and fish, (3) vegetables, (4) fruits, (5) legumes and cereals, (6) oils and fats, (7) pastries, (8) beverages and (9) miscellaneous. For each food, participants reported how often, on average, during the previous year they had consumed, specifying serving size with different options from “never or almost never” to “more than six times a day.”
Spanish food composition tables were used to calculate dietary intake, considering the daily intake of each food and the composition of nutrients [32, 33]. The ad hoc computer system was used to calculate the daily consumption of each food by multiplying the typical serving size by the frequency of consumption.
MQI assessment
As previously explained, the MQI was constructed based on three sub-indices, the Carbohydrate Quality Index (CQI), the Fat Quality Index (FQI), and the Healthy Plate Protein source Quality Index (HPPQI). The CQI has been used in previous cohort and trial studies to evaluate their association with CVD [34], plasma metabolomic profiles [35], and changes in cardiovascular risk factors [36]. The CQI is based on four equally weighted carbohydrate quality domains: glycemic index (GI), total dietary fiber intake (g/d), ratio of whole grains/ total cereals (whole grains + refined cereals + products prepared with refined flours), and the ratio of solid/total carbohydrates (liquids + solids).
The FQI has been used in nutritional adequacy [37] and CVD investigations [38]. For the calculation of FQI, monounsaturated fatty acids (MUFA), PUFA, SFA, and trans-fat acids (TFA) were taken into account as follows: FQI = (MUFA + PUFA)/(SFA + TFA), receiving equally weighting.
Lastly, the HPPQI has been used in a previous study conducted by our group [24] and it was calculated based on the following ratio: HPPQI = (seafood + poultry + pulses + nuts)/(red and processed meats + cheese), considering the first food group as healthy sources of protein and the second group as unhealthy sources, according to the Harvard’s Healthy Eating Plate [39].
To calculate the MQI, participants were classified into quintiles for each sub-index (CQI, FQI, and HPPQI), assigning values ranging from 1 (lowest quality) to 5 (highest quality). All the sub-index values were summed up, resulting in an MQI score ranging from 3 (poorest macronutrient quality) to 15 (highest macronutrient quality). Lastly, we classified participants into quartiles according to their total MQI score (Table 1).
Other dietary scores
Adherence to the Mediterranean diet (MedDiet) was assessed with the well-known score proposed by Trichopoulou et al. [40]. The total score range was from 0 to 9, with higher scores indicating greater adherence.
To assess adherence to the Provegetarian pattern, we used the score proposed by Martínez-González et al. [41]. The total score was calculated by summing up the values of the quintiles of vegetable food (1 point for the lowest quintile and 5 points for the highest quintile) and the values of the quintiles of animal food inversely weighted (1 point for the highest quintile and 5 points for the lowest quintile). The final score ranged from 12 (worst adherence) to 60 points (best adherence) [41].
Ascertainment of CVD
CVD was the primary endpoint of our study and it was included inquired in by every self-reported follow-up questionnaire collected every 2 years. When the participant reported a CVD event, we requested the medical documentation and a team of cardiologists adjudicated the event, blinded to the dietary exposures. The endpoint was a composite of acute myocardial infarction with or without ST elevation, stroke (both confirmed by a review of medical records with the prior permission of relatives), and cardiovascular death. Cardiovascular events were generally self-reported. Medical records of participants were requested to confirm cases and finally, cardiovascular events were confirmed by a cardiologist who was blind to diet and lifestyle exposure. Additionally, all potential cases were reviewed by a team of expert physicians. Nonfatal stroke was defined as a focal neurological deficit of sudden onset with a duration of more than 24 h and vascular mechanism. Diagnosis of myocardial infarction was defined using universal criteria [42]. Deceases from cardiovascular causes were confirmed by death certificates, medical records, or records linked to the National Institute of Statistics. For participants lost during follow-up, we consulted the National Death Index of Spain at least once a year, to identify any member of the cohort who may have died.
Other covariates
Additional covariates include anthropometric measurements, habits related to health and lifestyle. The validity of self-reported anthropometric information (weight and height) has been previously evaluated in a subsample of the SUN cohort [43].
Statistical analysis
We describe the baseline characteristics of participants adjusted for age and sex using the inverse probability weighting method according to quartiles of the MQI. Proportions for categorical variables and means and standard deviation (SD) for quantitative variables were calculated.
Cox proportional hazard regression models were used to estimate the association between the quartiles of MQI and CVD incidence. Hazard ratios (HRs) were calculated with their 95% confidence intervals (CIs) for each quartile, considering Q1 as the reference category. The interpretation of HR > 1 was considered a higher CVD risk, whereas HR < 1 was considered a lower probability of CVD.
Based on the existing literature and also on previous findings of the SUN cohort on [44, 45], we adjusted our models as follows: age was used as underlying time-variable in all models; model 1 was adjusted for sex, age (deciles), and stratified by year entering the cohort; model 2 was additionally adjusted for total energy intake (kcal/d, continuous), marital status (single, married, widowed, separated and others), educational level (years of higher education, continuous), smoking (never, current, and former smoker), accumulated smoking habit (pack-years, continuous), alcohol intake (never, < 5 women or < 10 men g/d, 5–25 women or 10–50 men g/d, and > 25 women or > 50 men g/d), physical activity (metabolic equivalent-h/week, continuous), snacking between meals (yes/no), body mass index (BMI [kg/m2, linear and quadratic terms, continuous]), time spent sitting (hours/week, continuous), weight gain in the previous 5 years before entering the cohort (< 3 kg and ≥ 3 kg) and following a special diet at baseline (yes/no); model 3, was additionally adjusted for family history of CVD (yes/no), and any diagnosis of diabetes (yes/no), hypertension (yes/no), hypercholesterolemia (yes/no), dyslipidemia (yes/no), depression (yes/no), cancer (yes/no); and lastly, model 4 was adjusted for total carbohydrate intake (g/d, continuous), total fat intake (g/d, continuous), and total protein intake (g/d, continuous).
Linear trend tests were performed through successive quartiles, assigning the median value of each quartile, and treating the resulting variables as continuous.
To minimize any effect of dietary variation, we used repeated measurements with updated data and cumulative diet average information of the MQI and its components, with a complete repetition of the FFQ after 10 years of follow-up. For the analysis of repeated measures, the mean between the baseline FFQ and the 10 year FFQ (i.e., cumulative average exposure) was calculated to assess a more realistic diet based on the MQI.
We additionally evaluated the combined effects of adherence to the MedDiet and the Provegetarian dietary pattern with the MQI. For both indexes, participants were categorized into two groups (below and above the median), interpreted as “low adherence” and “high adherence”, respectively, while the MQI was categorized into three groups (Q1, Q2-Q3, and Q4). We considered as reference category the Q4 of the MQI and the highest adherence to MedDiet or Provegetarian dietary pattern.
The following sensitivity analyses and subgroup analyses were additionally performed to assess the robustness of our findings: (a) selection by sex, only men or women participants, (b) only participants < 45 years or ≥ 45 years, (c) censoring participants at > 50 years, (d) only health professionals or only non-health professionals participants, (e) exclusion of participants with hypercholesterolemia and prevalent hypertension, (f) using different predefined energy intake limits (5th percentile and 95th percentile), (g) exclusion of participants with prevalent cancer, (h) exclusion of participants who followed a special diet at baseline, (i) exclusion of participants with ≥ 30 items missing in the FFQ, and (j) exclusion of participants with early CVD (≤ 2 years).
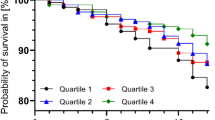
Finally, the Nelson-Aalen curves were used to represent the cumulative risk of CVD during the follow-up of the study according to tertiles of MQI (T1: < 8, T2:8–10 y T3 ≥ 11).
Statistical analyses were conducted using STATA version 16 (STATA Corporation) with the SUN database updated in December 2019. All p value were two-tailed, and statistical significance was deemed in the conventional cut-off p < 0.05.
Results
Baseline characteristics of the participants
A total of 18,418 participants were followed for a mean time of 14 years (211,744 person-years). During this time, 171 cases of prevalent CVD were identified, including 82 cases of nonfatal acute myocardial infarction, 61 cases of nonfatal strokes, and 28 CV exitus.
Table 2 presents the characteristics of the participants, according to the quartiles of the MQI adjusted for age and sex.
The mean age of the participants was 36 (SD 12.1) years, and the mean baseline BMI was 23.1 (SD 3.5) kg/m2. Around 61% of the participants were women. Participants in the highest quartile of the MQI were more likely to be single, healthcare professionals, physically active, and less likely to snack between meals. Additionally, participants with better MQI were more likely to follow a special diet, consume some type of supplementation, and have greater adherence to the MedDiet.
Regarding the dietary characteristics of participants, those with higher values of MQI consumed more vegetables, fruits, legumes, whole grains, fish, white meats, skimmed dairy products, nuts, and olive oil, but smaller quantities of whole dairy, eggs, soft drinks, and fast food. Regarding the baseline intake, participants in the highest quartile had a higher proportion of energy intake from carbohydrates, PUFA, and showed greater fiber intake, while their proportion of total fat, SFA, TFA, and cholesterol intake was lower (Table 3).
Association between MQI and CVD
Table 4 shows the results of the multivariate Cox regression analysis for the association between MQI and CVD risk. An inverse association between the MQI and the risk of CVD was observed in all models. Point estimates monotonically decreased across successive quartiles of MQI. In the fully adjusted model, the relative risk of CVD was 40% lower for participants in the highest quartile when compared with the lowest quartile, with HR of 0.60 (95% CI, 0.38 – 0.96, Ptrend = 0.024).
To have an updated dietary approach, we conducted time-dependent Cox regression models with repeated measurements of dietary exposures using cumulative average information after a 10 year follow-up. Despite all models revealed an inverse association between the MQI and the risk of CVD, none of the models reached statistical significance, with HR of 0.68 (95% CI 0.42–1.09, Ptrend = 0.101) for updated dietary information and HR of 0.65 (95% CI 0.41–1.03, Ptrend = 0.073) when comparing participants in the highest vs the lowest quartile in fully adjusted models (Table 5).
The combined analyses of MQI and other dietary scores and the incidence of CVD
Figure 2a and b represent HRs for the incidence of CVD according to the combined analysis of MQI and adherence to MedDiet and Provegetarian Diet, respectively. All participants were categorized into six groups according to the quartile of the MQI (3 groups) and adherence to MedDiet and Provegetarian Diet (two groups, below and above the median).
a Hazard ratios (HR) and confidence intervals (CI) for CVD risk according to the combined effect of the MQI categories and the grade of adherence to the MedDiet in 18,418 participants of the SUN cohort*. b Hazard ratios (HR) and confidence intervals (CI) for CVD risk according to the combined effect of the MQI categories and the grade of adherence to a provegetarian pattern in 18,418 participants of the SUN cohort*. *Adjusted for sex, age (deciles), and stratified by year entering the cohort, total energy intake, marital status (five categories), educational level, smoking (three categories), accumulated smoking habit, alcohol intake, physical activity, snacking between meals (yes/no), BMI, sitting time, weight gain in the previous 5 years before entering the cohort (< 3 kg and ≥ 3 kg), following a special diet at baseline, family history of CVD (yes/no), any diagnosis of diabetes (yes/no), hypertension (yes/no), hypercholesterolemia (yes/no), dyslipidemia (yes/no), depression (yes/no), cancer (yes/no), total carbohydrate intake, total fat intake, and total protein intake
Overall, for participants with lower adherence to the MedDiet (Fig. 2a) and Provegetarian Diet (Fig. 2b), the risk of CVD increased across higher categories of the MQI. Thus, the HR (95% CI) for participants with lower adherence to the MedDiet (≤ median) and lower MQI (Q1) was 1.71 (1.06—2.77) as compared to participants with higher adherence to the MedDiet (> median) and higher MQI (Q4). Similar results were observed for the analyses of the joint exposure to MQI and adherence to the Provegetarian Diet (Fig. 2b). Participants with lower adherence to both the MedDiet (≤ median) and lower MQI (Q1) had a HR (95% CI) of 1.77 (1.07—2.94).
Sensitivity analyses
Multiple sensitivity analyses were performed to corroborate our findings (Fig. 3). Overall, the results did not substantially change in any scenario or sub-group, observing an inverse association between MQI and the incidence of CVD. Point estimates were consistent with the HRs observed in the main analysis, except when men were excluded (HR = 1.13, 95% CI 0.34–3.76).
Hazard ratios (HR) and 95% confidence intervals (CI) for the association between MQI and the risk of CVD in the SUN cohort. Quartile 4 vs Quartile 1. Adjusted for sex, age (deciles), and stratified by year entering the cohort, total energy intake, marital status (five categories), educational level, smoking (three categories), accumulated smoking habit, alcohol intake, physical activity, snacking between meals (yes/no), BMI, sitting time, weight gain in the previous 5 years before entering the cohort (< 3 kg and ≥ 3 kg), following a special diet at baseline, family history of CVD (yes/no), any diagnosis of diabetes (yes/no), hypertension (yes/no), hypercholesterolemia (yes/no), dyslipidemia (yes/no), depression (yes/no), cancer (yes/no), total carbohydrate intake, total fat intake, and total protein intake
We used the Nelson-Aalen survival curve to graphically represent the association between CVD incidence and MQI during the follow-up of the study after controlling for confounding using inverse probability weighting (Fig. 4).
Discussion
Diet is a major determinant of health and life expectancy. Quality diets should recommend appropriate amounts and proportions of nutrient intake to help educate population about healthy choices. In the past years, a strong interest has emerged to design tools to assess the quality of the diet. The present study is the first to prospectively investigate the association between a novel and multidimensional MQI and the incidence of CVD. Unlike other previous a priori indices, this MQI does not assess adherence to an eating pattern or dietary guideline, but it specifically appraises the global macronutrient quality and quantity. Thus, the Pearson’s correlation coefficient between the MQI and MedDiet was 0.65.
As expected, participants with better MQI were healthier—had higher intakes of vegetables, fruits, legumes, whole grains, fish and seafood, white meats, skimmed dairy products, nuts, olive oil, PUFA, and fiber. These food groups contain high nutritional density and contain bioactive compounds such as flavonoids, polyphenols, and oleocanthal that confer cardioprotective effects [11, 13]. Moreover, the consumption of these food groups provides additional functional properties such as low energy density and low glycemic, which may reduce the risk of CVD [46,47,48]. Several studies have found that preference for whole grains over refined grains was associated with lower CVD risk. Whole grains preserve all the components (the bran, germ, and endosperm), which help decrease total cholesterol, LDL, body fat, and maintain adequate levels of postprandial glucose [49,50,51,52]. A previous study in this cohort demonstrated an inverse association between better carbohydrate quality or a higher proportion of energy from better quality carbohydrates and the risk of CVD and a higher proportion of carbohydrates from whole grains was strongly inversely associated with CVD. Moreover, the study suggested that replacing the amount of refined cereals with the same amount of whole grains and replacing bakery products or cookies with whole bread, the risk of CVD was significantly reduced [34].
Dietary fat has been considered one of the most important modifiable factors associated with the risk of CVD [53]. The traditional recommendations to prevent CVD have been focused on SFA [22]. Currently, the Dietary Guidelines for Americans (DGA) suggest considering fats as part of healthy dietary patterns [12]. In our study, we found participants with higher MQI showed a higher percentage of energy coming from PUFA and a lower percentage of energy from SFA. This is consistent with previous findings of multiple studies in which lower intake of SFA and higher intake of MUFA or PUFA from fish, nuts, and vegetable oils such as olive oil, were associated with a lower incidence rate of CVD [22, 53,54,55]. The lower incidence rate of CVD may be explained by the consequent decrease in LDL, which is well known to be the main causal factor in the development of atherosclerosis [56]. Previous research in the SUN cohort assessed the dietary quality of fats using the FQI. This study did not find any association between a better fat quality diet and the risk of incident CVD. The authors concluded that a “heart-healthy diet” should focus on a general dietary pattern approach rather than limiting total fat intake or certain subtypes of fat [38].
In our study, we found that participants with higher consumption of red meat, whole dairy products, and eggs, had a lower MQI. However, epidemiological evidence of the long-term effects of higher protein intake on CVD is not clear [57]. Previous findings have demonstrated that intake of vegetable protein from legumes, fruits, vegetables, and nuts was associated with a lower risk of CVD. Conversely, vegetable protein intake from cereals and potatoes was not associated with all-cause mortality and CVD [58, 59]. Animal protein, mainly red meat, has a high content of SFA that increases plasma concentrations of LDL cholesterol [58, 59], which is linked to the etiopathogenesis of CVD [17]. Therefore, the replacement of red meat by fish or poultry and the increase in the consumption of plant protein may produce positive cardioprotective effects [57]. Additionally, changing the proportion of animal protein by vegetable protein may lead to greater CVD risk reduction due to the lower energy density of these foods, and consequently, a reduction in body weight, a major modifiable factor involved in the incidence of CVD [57].
In this sense, Michas et al. [60] suggested that the total matrix of food is more important than just the content of certain nutrients to predicting the effect on CVD risk. Thus, recommendations to improve the dietary quality should be oriented towards dietary patterns that have an adequate contribution of macronutrients, such as the MedDiet, and should not focus on isolated nutrients [61].
The benefits of MedDiet in the prevention of CVD have been reported in multiple studies [62]. Strong and robust evidence exists about the beneficial effects of the MedDiet on the prevention of chronic diseases, particularly CVD due to its high content of legumes, fruits, vegetables, nuts, fish, and olive oil as the main fat, and its low content of meat products [62, 63].
The cardioprotective effects of the provegetarian pattern may be explained due to the high content of foods from plants, fruits, fiber, PUFA, lower intake of animal foods such as red and processed meats, and SFA. These foods lead to positive outcomes such as reduction in lower blood pressure, LDL, inflammatory processes, and improvements in glycemic control [64]. This is supported by a recent meta-analysis with seven studies and 15,077 cases of CVD, in which the authors concluded that greater adherence to the plant-based diet conferred CVD protection [65]. However, it is important to highlight that unhealthy plant-based diets may be associated with higher CVD risk. Lower intakes of unsaturated fats, micronutrients, antioxidants, dietary fiber, and higher intakes of added sugar and glycemic load may explain this detrimental association [66, 67].
Some limitations of the present study should be noted. First, our participants had a high level of education, and results may be considered non-representative of the general population. However, the generalizability of results should be based on biological mechanisms rather than statistical representativeness. Second, the FFQ was self-reported, which may lead to measurement errors. Nevertheless, the FFQ is a gold standard tool in nutritional epidemiology for assessing eating habits and has been repeatedly validated [23,24,25]. Third, residual confounding cannot be excluded, despite our models were adjusted for traditional CVD risk factors. Fourth, it has not been possible to independently evaluate the association of MQI with fatal cardiovascular disease, due to the small number of fatal events (only 28) in our cohort, that precluded component-specific analyses for the composite CVD outcome. Finally, the MQI has not been formally validated to date. Although some food groups with cardio-protective properties were correlated with the MQI, the novelty of our findings is the more comprehensive nature of our approach that goes beyond assessing the amounts or proportions of each macronutrient in a particular diet and considers instead the overall quality of the three major macronutrients. In addition, this index is based on the best scientific evidence and it was previously used in other studies [24].
The strengths of the study rely on its large sample size, the large follow-up period, the high retention rate (> 91%), the ability to control for a wide number of potential confounders, the blind confirmation of cardiovascular events by medical records which minimizes the potential misclassification bias, the use of repeated measurements of diet after 10 years of follow-up, the numerous sensitivity analyses, and the confirmation of deaths by the Spanish National Death Index.
Conclusions
In conclusion, in this Mediterranean cohort, we found a significant inverse relationship between a multidimensional MQI and a lower risk of CVD. Additionally, we found that better MQI with greater adherence to MedDiet or the Provegetarian diet was also associated with a lower risk of CVD.
More research is needed to establish appropriate dietary guidelines about the quality of macronutrients to reduce CVD risk based on healthy dietary patterns such as the Mediterranean or the Provegetarian.
References
Liu MB (2021) Cardiovascular diseases. In: World Health Organization. https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-cvds. Accessed 22 Jun 2021
Mozaffarian D, Benjamin EJ, Go AS et al (2016) Heart disease and stroke statistics-2016 update a report from the American heart association. Circulation 133:e38–e48. https://doi.org/10.1161/CIR.0000000000000350
Timmis A, Townsend N, Gale CP et al (2020) European society of cardiology: cardiovascular disease statistics 2019. Eur Heart J 41:12–85. https://doi.org/10.1093/eurheartj/ehz859
Roth GA, Mensah GA, Johnson CO et al (2020) Global burden of cardiovascular diseases and risk factors, 1990–2019: update from the GBD 2019 study. J Am Coll Cardiol 76:2982–3021. https://doi.org/10.1016/j.jacc.2020.11.010
WHO Cvd Risk Chart Working Group (2019) World health organization cardiovascular disease risk charts: revised models to estimate risk in 21 global regions. Lancet Glob Heal 7:e1332–e1345. https://doi.org/10.1016/S2214-109X(19)30318-3
Eckel RH, Jakicic JM, Ard JD et al (2014) 2013 AHA/ACC guideline on lifestyle management to reduce cardiovascular risk: a report of the American college of cardiology/American heart association task force on practice guidelines. J Am Coll Cardiol 63:2960–2984. https://doi.org/10.1016/j.jacc.2013.11.003
Tappia PS, Blewett H (2020) Nutrition and cardiovascular health. Int. J. Mol. Sci. 21(7):2284. https://doi.org/10.3390/ijms21072284
Medina-Remón A, Kirwan R, Lamuela-Raventós RM, Estruch R (2018) Dietary patterns and the risk of obesity, type 2 diabetes mellitus, cardiovascular diseases, asthma, and neurodegenerative diseases. Crit Rev Food Sci Nutr 58:262–296. https://doi.org/10.1080/10408398.2016.1158690
Ruel MT (2003) Operationalizing dietary diversity: a review of measurement issues and research priorities. J Nutr 133:3911S-3926S. https://doi.org/10.1093/jn/133.11.3911S
England CY, Andrews RC, Jago R, Thompson JL (2015) A systematic review of brief dietary questionnaires suitable for clinical use in the prevention and management of obesity, cardiovascular disease and type 2 diabetes. Eur J Clin Nutr 69:977–1003. https://doi.org/10.1038/ejcn.2015.6
Nestel PJ, Beilin LJ, Clifton PM et al (2021) Practical guidance for food consumption to prevent cardiovascular disease. Heart Lung Circ 30:163–179. https://doi.org/10.1016/j.hlc.2020.08.022
Stoody EE, Obbagy J, Pannucci TR, Rahavi E, Altman J, Adler M, Clarissa (Claire) Brown, Kelley S, Scanlon R (2020) Dietary Guidelines for Americans. In: DGA. https://www.dietaryguidelines.gov/. Accessed 22 Jun 2021
Arnett DK, Blumenthal RS, Albert MA et al (2019) 2019 ACC/AHA guideline on the primary prevention of cardiovascular disease: a report of the American college of cardiology/American heart association task force on clinical practice guidelines. Circulation 140:e596–e646. https://doi.org/10.1161/CIR.0000000000000678
Mach F, Baigent C, Catapano AL et al (2019) 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk: the task force for the management of dyslipidaemias of the European society of cardiology (ESC) and European atherosclerosis society (EAS). Eur Heart J 41:111–188. https://doi.org/10.1093/eurheartj/ehz455
Schwingshackl L, Christoph M, Hoffmann G (2015) Effects of olive oil on markers of inflammation and endothelial function-a systematic review and meta-analysis. Nutrients 7:7651–7675. https://doi.org/10.3390/nu7095356
Zhu Y, Bo Y, Liu Y (2019) Dietary total fat, fatty acids intake, and risk of cardiovascular disease: a dose-response meta-analysis of cohort studies. Lipids Health Dis 18:91. https://doi.org/10.1186/s12944-019-1035-2
Astrup A, Magkos F, Bier DM et al (2020) Saturated fats and health: a reassessment and proposal for food-based recommendations: JACC state-of-the-art review. J Am Coll Cardiol 76:844–857. https://doi.org/10.1016/j.jacc.2020.05.077
Bhupathi V, Mazariegos M, Cruz Rodriguez JB, Deoker A (2020) Dairy intake and risk of cardiovascular disease. Curr Cardiol Rep 22:10–15. https://doi.org/10.1007/s11886-020-1263-0
Institute of Medicine (2005) Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein, and amino acids (Macronutrients). DC, The National Academies Press, Washington
Voortman T, Chen Z, Girschik C et al (2021) Associations between macronutrient intake and coronary heart disease (CHD): the rotterdam study. Clin Nutr 40:5494–5499. https://doi.org/10.1016/j.clnu.2021.08.022
Shan Z, Rehm CD, Rogers G et al (2019) Trends in dietary carbohydrate, protein, and fat intake and diet quality among US adults, 1999–2016. JAMA - J Am Med Assoc 322:1178–1187. https://doi.org/10.1001/jama.2019.13771
Sacks FM, Lichtenstein AH, Wu JHY et al (2017) Dietary fats and cardiovascular disease: a presidential advisory from the american heart association. Circulation 136:e1–e23. https://doi.org/10.1161/CIR.0000000000000510
Hooper L, Martin N, Jimoh OF et al (2020) Reduction in saturated fat intake for cardiovascular disease. Cochrane database Syst Rev 5:CD011737. https://doi.org/10.1002/14651858.CD011737.pub2
Santiago S, Zazpe I, Fernandez-Lazaro CI et al (2021) Macronutrient quality and all-cause mortality in the SUN cohort. Nutrients. https://doi.org/10.3390/nu13030972
Burggraf C, Teuber R, Brosig S, Meier T (2018) Review of a priori dietary quality indices in relation to their construction criteria. Nutr. Rev. 76:747–764. https://doi.org/10.1093/nutrit/nuy027
Proyecto SUN. Departamento de Medicina Preventiva y Salud Pública. Facultad de Medicina. Universidad de Navarra. https://www.unav.edu/web/departamento-de-medicina-preventiva-y-salud-publica/proyecto-sun. Accessed 8 Jul 2021
Seguí-Gómez M, de la Fuente C, Vázquez Z et al (2006) Cohort profile: the “Seguimiento Universidad de Navarra” (SUN) study. Int J Epidemiol 35:1417–1422. https://doi.org/10.1093/ije/dyl223
Willet WC (2012) Nutritional epidemiology, 3rd edn. Oxford University Press, New York, NY, USA
De La Fuente-Arrillaga C, Vzquez Ruiz Z, Bes-Rastrollo M et al (2010) Reproducibility of an FFQ validated in Spain. Public Health Nutr 13:1364–1372. https://doi.org/10.1017/S1368980009993065
Martin-Moreno JM, Boyle P, Gorgojo L et al (1993) Development and validation of a food frequency questionnaire in Spain. Int J Epidemiol 22:512–519. https://doi.org/10.1093/ije/22.3.512
Fernández-Ballart JD, Piñol JL, Zazpe I et al (2010) Relative validity of a semi-quantitative food-frequency questionnaire in an elderly Mediterranean population of Spain. Br J Nutr 103:1808–1816. https://doi.org/10.1017/S0007114509993837
Moreiras O, Carbajal Á, Cabrera L, Cuadrado C (2005) Tablas de Composición de Alimentos (Food Composition Tables), 9th edn. Madrid, Spain, Pirámide
Mataix Verdu J (2003) Tabla de Composición de Alimentos Españoles (Spanish Food Composition Tables), 4th edn. Universidad de Granada, Granada, Spain
Zazpe I, Santiago S, Gea A et al (2016) Association between a dietary carbohydrate index and cardiovascular disease in the SUN (Seguimiento Universidad de Navarra) Project. Nutr Metab Cardiovasc Dis 26:1048–1056. https://doi.org/10.1016/j.numecd.2016.07.002
Bulló M, Papandreou C, Ruiz-Canela M et al (2021) Plasma metabolomic profiles of glycemic index, glycemic load, and carbohydrate quality index in the PREDIMED study. J Nutr 151:50–58. https://doi.org/10.1093/jn/nxaa345
Martínez-González MA, Fernandez-Lazaro CI, Toledo E et al (2020) Carbohydrate quality changes and concurrent changes in cardiovascular risk factors: a longitudinal analysis in the PREDIMED-Plus randomized trial. Am J Clin Nutr 111:291–306. https://doi.org/10.1093/ajcn/nqz298
Sánchez-Tainta A, Zazpe I, Bes-Rastrollo M et al (2016) Nutritional adequacy according to carbohydrates and fat quality. Eur J Nutr 55:93–106. https://doi.org/10.1007/s00394-014-0828-3
Santiago S, Zazpe I, Gea A et al (2018) Fat quality index and risk of cardiovascular disease in the sun project. J Nutr Health Aging 22:526–533. https://doi.org/10.1007/s12603-018-1003-y
Healthy Eating Plate (2011). The Nutrition Source. Harvard T.H. Chan School of Public Health https://www.hsph.harvard.edu/nutritionsource/healthy. Accessed 22 Jun 2021
Trichopoulou A, Costacou T, Bamia C, Trichopoulos D (2003) Adherence to a Mediterranean diet and survival in a Greek population. N Engl J Med 348:2599–2608. https://doi.org/10.1056/NEJMoa025039
Martínez-González MA, Sánchez-Tainta A, Corella D et al (2014) A provegetarian food pattern and reduction in total mortality in the Prevención con Dieta Mediterránea (PREDIMED) study. Am J Clin Nutr 100(Suppl):320S-S328. https://doi.org/10.3945/ajcn.113.071431
Thygesen Kr, Alpert J S, Jaffe Al S, Simoons M L, Chaitman B R, HDW, (2012) Third universal definition of myocardial infarction. Eur Heart J 33(20):2551–2567. https://doi.org/10.1093/eurheartj/ehs184
Bes-Rastrollo M, Valdivieso J, Sanchez-Villegas A et al (2005) Validación del peso e índice de masa corporal auto-declarados de los participantes de una cohorte de graduados universitarios. Rev Esp Obes. https://doi.org/10.1016/j.gaceta.2011.02.009
Mendonça RD, Carvalho NC, Martin-Moreno JM et al (2019) Total polyphenol intake, polyphenol subtypes and incidence of cardiovascular disease: the SUN cohort study. Nutr Metab Cardiovasc Dis 29:69–78. https://doi.org/10.1016/j.numecd.2018.09.012
Martínez-González MA, García-López M, Bes-Rastrollo M et al (2011) Mediterranean diet and the incidence of cardiovascular disease: a Spanish cohort. Nutr Metab Cardiovasc Dis 21:237–244. https://doi.org/10.1016/j.numecd.2009.10.005
Satija A, Hu FB (2012) Cardiovascular benefits of dietary fiber. Curr Atheroscler Rep 14:505–514. https://doi.org/10.1007/s11883-012-0275-7
Yamada T, Hayasaka S, Shibata Y et al (2011) Frequency of citrus fruit intake is associated with the incidence of cardiovascular disease: the Jichi Medical School cohort study. J Epidemiol 21:169–175. https://doi.org/10.2188/jea.je20100084
Miller V, Mente A, Dehghan M et al (2017) Fruit, vegetable, and legume intake, and cardiovascular disease and deaths in 18 countries (PURE): a prospective cohort study. Lancet (London, England) 390:2037–2049. https://doi.org/10.1016/S0140-6736(17)32253-5
Cho SS, Qi L, Fahey GCJ, Klurfeld DM (2013) Consumption of cereal fiber, mixtures of whole grains and bran, and whole grains and risk reduction in type 2 diabetes, obesity, and cardiovascular disease. Am J Clin Nutr 98:594–619. https://doi.org/10.3945/ajcn.113.067629
Ludwig DS, Hu FB, Tappy L, Brand-Miller J (2018) Dietary carbohydrates: role of quality and quantity in chronic disease. BMJ 361:k2340. https://doi.org/10.1136/bmj.k2340
Zong G, Gao A, Hu FB, Sun Q (2016) Whole grain intake and mortality from all causes, cardiovascular disease, and cancer: a meta-analysis of prospective cohort studies. Circulation 133:2370–2380. https://doi.org/10.1161/CIRCULATIONAHA.115.021101
Barrett EM, Batterham MJ, Ray S, Beck EJ (2019) Whole grain, bran and cereal fibre consumption and CVD: a systematic review. Br J Nutr 121:914–937. https://doi.org/10.1017/S000711451900031X
Lichtenstein AH (2019) Dietary fat and cardiovascular disease: Ebb and flow over the last half century. Adv Nutr 10:S332–S339. https://doi.org/10.1093/advances/nmz024
Clifton PM, Keogh JB (2017) A systematic review of the effect of dietary saturated and polyunsaturated fat on heart disease. Nutr Metab Cardiovasc Dis 27:1060–1080. https://doi.org/10.1016/j.numecd.2017.10.010
Kris-Etherton PM, Petersen K, Van Horn L (2018) Convincing evidence supports reducing saturated fat to decrease cardiovascular disease risk. BMJ Nutr Prev Heal 1:23–26. https://doi.org/10.1136/bmjnph-2018-000009
Ference BA, Ginsberg HN, Graham I et al (2017) Low-density lipoproteins cause atherosclerotic cardiovascular disease. 1. Evidence from genetic, epidemiologic, and clinical studies. A consensus statement from the European atherosclerosis society consensus panel. Eur Heart J 38:2459–2472. https://doi.org/10.1093/eurheartj/ehx144
Zhubi-Bakija F, Bajraktari G, Bytyçi I et al (2021) The impact of type of dietary protein, animal versus vegetable, in modifying cardiometabolic risk factors: a position paper from the international lipid expert panel (ILEP). Clin Nutr 40:255–276. https://doi.org/10.1016/j.clnu.2020.05.017
Budhathoki S, Sawada N, Iwasaki M et al (2019) Association of animal and plant protein intake with all-cause and cause-specific mortality in a Japanese cohort. JAMA Intern Med 179:1509–1518. https://doi.org/10.1001/jamainternmed.2019.2806
Chen Z, Glisic M, Song M et al (2020) Dietary protein intake and all-cause and cause-specific mortality: results from the rotterdam Study and a meta-analysis of prospective cohort studies. Eur J Epidemiol 35:411–429. https://doi.org/10.1007/s10654-020-00607-6
Michas G, Micha R, Zampelas A (2014) Dietary fats and cardiovascular disease: putting together the pieces of a complicated puzzle. Atherosclerosis 234:320–328. https://doi.org/10.1016/j.atherosclerosis.2014.03.013
Nettleton JA, Lovegrove JA, Mensink RP, Schwab U (2016) Dietary fatty acids: is it time to change the recommendations? Ann Nutr Metab 68:249–257. https://doi.org/10.1159/000446865
Rosato V, Temple NJ, La Vecchia C et al (2019) Mediterranean diet and cardiovascular disease: a systematic review and meta-analysis of observational studies. Eur J Nutr 58:173–191. https://doi.org/10.1007/s00394-017-1582-0
Papadaki A, Nolen-Doerr E (2020) Mantzoros CS (2020) the effect of the mediterranean diet on metabolic health: a systematic review and meta-analysis of controlled trials in adults. Nutrients 12(11):3342. https://doi.org/10.3390/nu12113342
Kim H, Caulfield LE, Garcia-Larsen V et al (2019) Plant-based diets are associated with a lower risk of incident cardiovascular disease, cardiovascular disease mortality, and all-cause mortality in a general population of middle-aged adults. J Am Heart Assoc 8:e012865. https://doi.org/10.1161/JAHA.119.012865
Jarvis S, Tinajero M, Khan T, Hanley A, Jenkins D, Malik V (2021) Plant-based dietary patterns and cardiometabolic risk: a systematic review and meta-analysis of prospective cohort studies. Curr Dev Nutr. https://doi.org/10.1093/cdn/nzab038_028
Satija A, Bhupathiraju SN, Spiegelman D et al (2017) Healthful and unhealthful plant-based diets and the risk of coronary heart disease in U.S. Adults J Am Coll Cardiol 70:411–422. https://doi.org/10.1016/j.jacc.2017.05.047
Hemler EC, Hu FB (2019) Plant-based diets for cardiovascular disease prevention: all plant foods are not created equal. Curr Atheroscler Rep 21:18. https://doi.org/10.1007/s11883-019-0779-5
Acknowledgements
We thank very specially all participants in the SUN cohort for their long-standing and enthusiastic collaboration and our advisors from Harvard TH Chan School of Public Health Walter Willett, Alberto Ascherio, Frank B. Hu and Meir J. Stampfer who helped us to design the SUN Project, the PREDIMED study and the PREDIMED-PLUS on-going trial. We thank other members of the SUN Group: Alonso A, Álvarez-Álvarez I, Balaguer A, Barbagallo M, Barrientos I, Barrio-López MT, Basterra-Gortari FJ, Battezzati A, Bazal P, Benito S, Bertoli S, Beulen Y, Beunza JJ, Buil-Cosiales P, Canales M, Carlos S, Carmona L, Cervantes S, Cristobo C, de Irala J, de la Fuente-Arrillaga C, de la Rosa PA, Delgado-Rodríguez M, Díaz-Gutiérrez J, Díez Espino J, Domínguez L, Donat-Vargas C, Donazar M, Eguaras S, Fernández-Montero A, Fresán U, Galbete C, García-Arellano A, García López M, Gardeazábal I, Gutiérrez-Bedmar M, Goméz-Domingos AL, Gómez-Donoso C, Gómez-Gracia E, Goñi E, Guillén F, Henríquez P, Hernández A, Hershey MS, Hidalgo-Santamaría M, Hu E, Lahortiga F, Leone A, Llorca J, López del Burgo C, Marí A, Marques I, Martí A, Martín-Moreno JM, Martínez-Lapiscina EH, Mendonça R, Menéndez C, Molendijk M, Molero P, Murphy K, Muñoz M, Núñez-Córdoba JM, Pajares R, Papadaki A, Parletta N, Pérez de Ciriza P, Pérez Cornago A, Pérez de Rojas J, Pimenta AM, Pons J, Ramallal R, Razquin C, Rico A, Romanos A, Ruano C, Ruiz L, Ruiz Zambrana A, Salgado E, San Julián B, Sánchez D, Sánchez-Bayona R, Sánchez-Tainta A, Sánchez-Villegas A, Sayón-Orea C, Schlatter J, Serrano-Martinez M, Toledo E, Toledo J, Tortosa A, Valencia F, Vázquez Z, Zarnowiecki D.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. The SUN Project is supported by the Instituto de Salud Carlos III and the European Regional Development Fund (FEDER) (RD 06/0045, CIBER-OBN, grant PI10/02658, PI10/02293, PI13/00615, PI14/01668, PI14/01798, P14/01764, PI17/01795, PI20/00564 y G03/140), PNSD (2020/2021), Navarra Regional Government (45/2011, 122/2014, 41/2016) and the University of Navarra. The funding sources had no role in the design, execution, analyses, and interpretation of the data or decision to submit the results of this study.
Author information
Authors and Affiliations
Contributions
MAM-G: designed and started the cohort study. IZ and MAM-G: design the present study and were responsible for study oversight, PV, IZ and MAM-G: performed the statistical analysis. PV, SS and IZ: drafted the manuscript and all authors contributed to its development. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Ethical approval
The SUN study has been conducted in accordance with the Declaration of Helsinki, with the approval of the Institutional Review Board of the University of Navarra. This cohort is registered in Clinicaltrials.gov as NCT02669602.
Consent for publication
All co-authors have read and approved submission of this article for publication.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Vanegas, P., Zazpe, I., Santiago, S. et al. Macronutrient quality index and cardiovascular disease risk in the Seguimiento Universidad de Navarra (SUN) cohort. Eur J Nutr 61, 3517–3530 (2022). https://doi.org/10.1007/s00394-022-02901-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-022-02901-3