Abstract
Background
Sex differences and causality of the association between heart rate variability (HRV) and atrial fibrillation (AF) in the general population remain unclear.
Methods
12,334 participants free of AF from the population-based Rotterdam Study were included. Measures of HRV including the standard deviation of normal RR intervals (SDNN), SDNN corrected for heart rate (SDNNc), RR interval differences (RMSSD), RMSSD corrected for heart rate (RMSSDc), and heart rate were assessed at baseline and follow-up examinations. Joint models, adjusted for cardiovascular risk factors, were used to determine the association between longitudinal measures of HRV with new-onset AF. Genetic variants for HRV were used as instrumental variables in a Mendelian randomization (MR) analysis using genome-wide association studies (GWAS) summary-level data.
Results
During a median follow-up of 9.4 years, 1302 incident AF cases occurred among 12,334 participants (mean age 64.8 years, 58.3% women). In joint models, higher SDNN (fully-adjusted hazard ratio (HR), 95% confidence interval (CI) 1.24, 1.04–1.47, p = 0.0213), and higher RMSSD (fully-adjusted HR, 95% CI 1.33, 1.13–1.54, p = 0.0010) were significantly associated with new-onset AF. Sex-stratified analyses showed that the associations were mostly prominent among women. In MR analyses, a genetically determined increase in SDNN (odds ratio (OR), 95% CI 1.60, 1.27–2.02, p = 8.36 × 10–05), and RMSSD (OR, 95% CI 1.56, 1.31–1.86, p = 6.32 × 10–07) were significantly associated with an increased odds of AF.
Conclusion
Longitudinal measures of uncorrected HRV were significantly associated with new-onset AF, especially among women. MR analyses supported the causal relationship between uncorrected measures of HRV with AF. Our findings indicate that measures to modulate HRV might prevent AF in the general population, in particular in women.
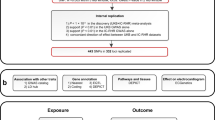
Graphical abstract

AF; atrial fibrillation, GWAS; genome-wide association study, IVW; inverse variance weighted, MR; Mendelian randomization, MR-PRESSO; MR-egger and mendelian randomization pleiotropy residual sum and outlier, RMSSD; root mean square of successive RR interval differences, RMSSDc; root mean square of successive RR interval differences corrected for heart rate, SDNN; standard deviation of normal to normal RR intervals, SDNNc; standard deviation of normal to normal RR intervals corrected for heart rate, WME; weighted median estimator.
aRotterdam Study n=12,334
bHRV GWAS n=53,174
cAF GWAS n=1,030,836
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Atrial fibrillation (AF), the most common cardiac arrhythmia, is associated with substantial morbidity and mortality and represents a significant burden on healthcare [1,2,3,4]. The exact AF pathogenesis remains to be identified. It has recently been suggested that cardiac autonomic imbalance could play a role in AF pathophysiology by promoting a decline in cardiac function [5,6,7,8,9,10].
Heart rate variability (HRV) is considered a non-invasive, accessible measure that may reflect the complex interaction between the autonomic nervous system and the heart [11, 12]. A complex relationship between HRV and AF has been suggested [5,6,7,8,9,10, 13]. Specifically, lower and higher levels of HRV may lead to decline in cardiac function and subsequently give rise to AF [5,6,7,8,9,10, 13]. Recent evidence shows that sex differences with regard to AF burden, pathophysiology, and prognosis exist [14]. However, the previous observational studies have been limited to either a cross-sectional design or a single measurement of HRV and did not evaluate sex differences. In addition, observational studies are prone to residual confounding and reverse causality [15].
Genome-wide association studies (GWAS) have identified genetic variants/single nucleotide polymorphisms (SNPs) for multiple assessments of HRV [16] and AF [17, 18]. Pathway and tissue enrichment analyses suggest that HRV SNPs are preferentially expressed within the sinoatrial node [16]. Moreover, AF SNPs have been suggested to affect the cardiac ion channels, cardiac calcium signaling, and the heart and skeletal muscles [17, 18]. This suggests that there may be a genetic foundation underlying the association between HRV and AF.
We aimed to investigate the association between longitudinal measures of HRV and heart rate with the risk of new-onset AF in the general population. Additionally, we used a comprehensive Mendelian randomization (MR) analysis using summary-level data from GWAS on measures of HRV and AF to investigate the potential causal relationship between HRV and AF.
Methods
Study population
The current study was embedded within the Rotterdam Study [19, 20]. The Rotterdam Study is a prospective population-based cohort study that aims to assess the occurrence and progression of risk factors for chronic diseases in middle-age and elderly persons. During 1990–1993, all inhabitants of the Ommoord district in the city of Rotterdam in The Netherlands aged ≥ 55 years were invited for the study. A total of 7983 (78% of all invitees) agreed to participate (RS-I). In 2000, the cohort was extended with 3011 participants who had become ≥ 55 years or had migrated into the research area (RS-II). In 2006, the cohort was again extended with 3932 participants who were ≥ 45 years (RS-III). The overall response rate at baseline was 72%. Participants attended follow-up examinations every 3–5 years. Outcome data on morbidity and mortality were continuously collected through linkage with digital files from general practitioners in the study area [19, 20].
For the present study, we included participants at study entry of the three recruitment waves. Participants with prevalent AF at baseline (n = 559), no informed consent for follow-up data collection (n = 305), no follow-up time (n = 6) or no measures of HRV (n = 1722) were excluded. Among the 12,334 free of AF included participants, 12,334 had at least 1 measurement for standard deviation of normal RR intervals (SDNN), SDNN corrected for heart rate (SDNNc), RR interval differences (RMSSD), RMSSD corrected for heart rate (RMSSDc), and heart rate. 8,832 participants had 2 measurements, 3837 had 3 measurements, 1817 had 4 measurements, and 787 participants had 5 measurements that were available during follow-up (before date of onset of AF, date of death, loss to follow-up, or to January 1, 2014, whichever occurred first).
Assessment of heart rate variability
Participants underwent a 10-s 12-lead resting electrocardiogram (ECG) using an ACTA Gnosis IV ECG recorder (Esaote Biomedica, Florence, Italy) and the ECG records were digitally stored. Subsequently, Modular ECG Analysis System (MEANS) was used to interpret the ECGs [21]. ECGs of individuals with a pacemaker, < 5 RR intervals between normal beats or > 5 premature supra- and/or ventricular complexes were excluded for the assessment of HRV [22]. In addition, the remaining ECGs marked as non-sinus arrhythmia and sinus arrhythmia by MEANS were manually assessed by 2 medical doctors to rule out and exclude atrial fibrillation/flutter, other arrhythmias, and ECGs with poor signal quality. Sinus rhythm (including sinus arrhythmia) is based on the detection by MEANS of regular P waves that have a fixed coupling interval with the following QRS complexes [21]. Furthermore, a random sample of 200 ECGs marked as sinus rhythm by MEANS were also manually checked by 2 medical doctors and 199 ECGs were found to be in sinus rhythm during the manual assessment indicating a very high positive predictive value of MEANS which was also demonstrated in earlier work [21]. RR intervals between 2 adjacent normal beats were used to compute the mean heart rate and time-domain indices of HRV; SDNN and RMSSD. Moreover, as HRV is potentially inversely and exponentially associated with heart rate, we additionally used heart rate corrected values of RMSSD (RMSSDc), and SDNN (SDNNc) using an exponential model [22,23,24,25,26]. The reproducibility of the HRV data was evaluated in a later cohort of the Rotterdam Study, in which ECG recordings of 3–5 min were made. From a sample of 310 3–5 min ECGs, we extracted from each recording two 10-s ECGs, one after the first minute of recording, the second after 2 minutes. The sample of 310 pairs of 10-s ECGs was also manually assessed by 2 medical doctors to rule out and exclude arrhythmias, and ECGs with poor signal quality. After exclusion, 211 ECG pairs remained and were used to calculate the HRV measures. Differences were examined using the paired T-test. The HRV measures did not statistically significantly differ from each other (p = 0.087 for RR, p = 0.415 for SDNN, and p = 0.427 for RMSSD).
Assessment of atrial fibrillation
AF was defined in accordance with the European Society of Cardiology (ESC) guidelines [4]. The methods on event adjudication for prevalent and incident AF within the Rotterdam Study have been described in detail earlier [20]. In short, AF was assessed at baseline and follow-up examinations using a 10-s 12-lead ECG with an ACTA Gnosis IV ECG recorder (Esaote Biomedica, Florence, Italy). The ECG records were then stored digitally and analyzed with MEANS [21]. Thereafter, 2 medical doctors validated the diagnosis of AF and in case of disagreement a cardiologist was consulted [3]. Additional follow-up data were obtained from medical files of participating general practitioners, hospitals, outpatient clinics, national registration of all hospitals discharge diagnoses, and follow-up examinations at the research center. The date of incident AF was defined as the date of the first occurrence of symptoms suggestive of AF with subsequent ECG verification obtained from the medical records. Participants were followed from the date of enrollment in the Rotterdam Study until the date of onset of AF, date of death, loss to follow-up, or to January 1, 2014, whichever occurred first.
Assessment of cardiovascular risk factors
The cardiovascular risk factors included in this study were body mass index (BMI), total cholesterol, high-density lipoprotein cholesterol, hypertension, smoking status, history of diabetes mellitus, history of coronary heart disease, history of heart failure, left ventricular hypertrophy on the ECG, use of cardiac medication, use of antihypertensive medication, use of beta blockers, use of calcium blockers, and use of lipid lowering medication. Methods for measurements of cardiovascular risk factors are explained in detail in the Methods S1 [3, 19, 20].
Selection of genetic variants on heart rate variability and atrial fibrillation
Genetic variants associated with HRV were used as instrumental variables for the MR analyses. The genetic variants were retrieved from publically available summary statistics from 2 GWAS [16,17,18]. Details regarding these study populations are depicted in Tables S4 and S5. For HRV, we retrieved independent genetic variants from a GWAS on HRV that assessed SDNN and RMSSD as log transformed continuous measures. This GWAS meta-analysis on HRV included 53,174 participants from European descent [16]. In addition, we retrieved independent genetic variants that were associated with AF from a GWAS that included 1,030,836 European participants (60,620 AF cases and 970,216 controls) [17]. We only included independent genetic variants in the subsequent MR analyses (p-value < 5.0 × 10–08 genome-wide significant and r2 < 0.1).
Statistical analyses
Joint model analyses
The baseline characteristics of the study population are presented as mean with standard deviation (SD) or number (n) with percentages as appropriate. Differences between men and women were examined by Student’s T-test (normal distribution) or the Mann Whitney U-test (skewed distribution) for continuous variables, and Chi-Square Test for categorical variables. The distributions of the different HRV measures and heart rate were skewed. Therefore, a natural logarithmic transformation was used to obtain a normal distribution.
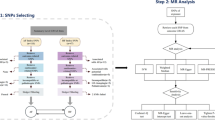
Competing risk analyses were employed using joint models for longitudinal and time-to-event data. To investigate the association between longitudinal measures of HRV with the risk of new-onset AF with mortality as a competing event, cause-specific hazard ratios (HRs) with their 95% confidence intervals (CIs) were calculated to quantify the associations (Fig. 1). See the Methods S2 for more details on the rationale, imputation and sensitivity analyses of the joint model analyses [27, 28].
Flow chart for the conducted analyses. Abbreviations: AF, atrial fibrillation; GWAS, genome-wide association study; HRV, heart rate variability; SDNN, standard deviation of normal to normal RR intervals; SDNNc, standard deviation of normal to normal RR intervals corrected for heart rate; RMSSD, root mean square of successive RR interval differences; RMSSDc, root mean square of successive RR interval differences corrected for heart rate; SNPs, single-nucleotide polymorphisms
The analyses were done in the total study population and for men and women separately. Additionally, we reported the p-values of sex interaction from the joint model. All models (mixed- and survival models) were adjusted for age, sex (if applicable), and cohort (model 1), and additionally for cardiovascular risk factors including body mass index, total cholesterol, high-density lipoprotein cholesterol, hypertension, smoking status, history of diabetes mellitus, history of coronary heart disease, history of heart failure, left ventricular hypertrophy on the ECG, cardiac medication, beta blockers, calcium blockers, and use of lipid lowering medication (model 2). Time was measured in years after baseline and the variables from model 1 and 2 were treated as covariates in the subsequent models.
Mendelian randomization analyses
We conducted two-sample MR analyses to examine the potential causal association between HRV and AF. The inverse variance weighted (IVW) method is the main method used in our analyses [29]. MR estimates were presented as odds ratios (ORs) with corresponding 95% CIs (Fig. 1). See the Methods S3, for more details on the rationale, assumptions and sensitivity analyses of the MR analyses [15, 29,30,31,32,33,34,35].
A two-tailed p < 0.05 was considered statistically significant. The data management was done using IBM SPSS Statistics version 25.0 for Windows (IBM Corp, Armonk, New York). The statistical analyses were done using the R packages “JMbayes2”, [36] and “TwoSampleMR”(30, 34, 35) in R software (R 4.0.2; R Foundation for Statistical Computing, Vienna, Austria) [37].
Results
Joint model analyses
A total of 12,334 participants, 5140 men (41.7%) and 7194 women (58.3%), were eligible for the analyses. Baseline characteristics for the total study population and stratified by sex are presented in Table 1. The mean age of the total study population was 64.8 ± 9.5 years and 58.3% were women. Median values for SDNN, SDNNc, RMSSD, RMSSDc, and heart rate were 16.2 ms, 27.0 ms, 16.3 ms, 33.7 ms, and 69.0 beats/min, respectively. See Table 1 for more details.
During a median follow-up of 9.4 years (interquartile range (IQR), 6.2–15.1), 1302 incident AF cases (10.6%) (613 in men and 691 in women) and 4004 mortality cases (32.5%) (1,740 in men and 2264 in women) occurred. The incidence rate of AF was 9.6 per 1000 person-years in the total study population (11.5 per 1000 person-years in men, 8.4 per 1000 person-years in women) and the incidence rate of mortality was 29.5 per 1000 person-years in the total study population (32.6 per 1000 person-years in men, 27.5 per 1000 person-years in women).
Joint models showed significant associations in model 2 with the risk of new-onset AF in the total study population for a higher SDNN (HR, 95% CI 1.24, 1.04–1.47, p = 0.0213), and a higher RMSSD (HR, 95% CI 1.33, 1.13–1.54, p = 0.0010). However, a higher SDNNc (HR, 95% CI 1.06, 0.89–1.23, p = 0.4784), higher RMSSDc (HR, 95% CI 1.09, 0.96–1.22, p = 0.1774), and a lower heart rate (HR, 95% CI 1.21, 0.74–1.99, p = 0.4781) were not significantly associated with the risk of new-onset AF in the total study population. The effect estimates slightly attenuated in model 2 in comparison to model 1, but SDNN, and RMSSD remained significant. See Table 2 for more details.
The sex-stratified analyses from model 2 showed that in men only the association for a higher RMSSD (HR, 95% CI 1.23, 1.01–1.48, p = 0.0414) with the risk of new-onset AF was significant. The analyses in women showed significant associations for a higher SDNN (HR, 95% CI 1.36, 1.03–1.79, p = 0.0278), higher RMSSD (HR, 95% CI 1.47, 1.16–1.89, p = 0.0018), and lower heart rate (HR, 95% CI 1.88, 1.02–3.67, p = 0.0408) with the risk of new-onset AF. See Table 2 for more information. In model 2, the p-values of the sex interaction in the joint model for SDNN, SDNNc, RMSSD, RMSSDc, and heart rate were p = 0.1077, p = 0.7638, p = 0.0065, p = 0.8465, and p = 0.1298, respectively.
All results of the joint model sensitivity analyses are depicted in Results S1.
Mendelian randomization analyses
A total of 33 genome-wide significant independent genetic variants were associated with HRV represented by SDNN (n = 16) and RMSSD (n = 17), respectively. A total of 5 SNPs for SDNN and 6 SNPs for RMSSD were available in the AF GWAS and were used for the MR analyses after removal of potential outliers. All individual genetic instruments for SDNN and RMSSD had a F statistic > 10 (median for SDNN, 53.2 (IQR, 51.8–70.8) and median for RMSSD, 54.7 (IQR, 43.1–69.3)) and were, therefore, considered to be of sufficient strength to be used in the MR analyses. The effect estimates of the genetic variants associated with SDNN, RMSSD and AF that were used in the MR analyses are presented in Table S4.
The MR estimates from the association between HRV and AF based on the IVW, weighted median estimator (WME), and MR-Egger methods are presented in Table 3. Specifically, MR analyses supported the causal effects of genetically determined SDNN and RMSSD on AF risk (for SDNN: n = 5 SNPs, OR, 95% CI 1.60, 1.27–2.02, p = 8.36 × 10–05 and for RMSSD: n = 6 SNPs, OR, 95% CI 1.56, 1.31–1.86, p = 6.32 × 10–07). A graphical presentation of the results can be found in Fig. 2.
Scatter plot which visualizes the causal effect estimate for each individual genetic variant on SDNN, and RMSSD and its effect on atrial fibrillation. Abbreviations: IVW, inverse variance weighted; RMSSD, the root mean square of successive RR interval differences; SDNN, standard deviation of normal to normal RR intervals; SNPs, single nucleotide polymorphisms; WME, weighted median estimator. The dots represent the corresponding effect estimates of each individual genetic variant. The crosses around the dots represent the 95% confidence intervals of the corresponding effect estimates of each individual genetic variant
The results of the MR sensitivity analyses are depicted in Results S2 [33].
Discussion
Our study shed light on the complex interaction between HRV and AF. Our joint model analyses showed that longitudinal measures of SDNN, and RMSSD were significantly associated with new-onset AF in the general population while SDNNc, RMSSDc, and heart rate were not significantly associated. Sex-stratified analyses showed that RMSSD among men, and SDNN, RMSSD, and heart rate among women were significantly associated with new-onset AF. MR analyses supported the causal association between SDNN, and RMSSD with AF. Our findings indicate that treatment to modulate HRV might prevent AF in the general population, in particular in women.
The exact mechanism that underlies the relationship between HRV and AF remains incompletely understood. Shared underlying risk factors, such as obesity, diabetes mellitus, and coronary heart disease, could influence HRV and are also implicated in AF pathophysiology [1, 5, 12, 38, 39]. In our study, however, the associations of HRV with incident AF slightly attenuated, but remained significant after extensive adjustment for shared cardiovascular risk factors. The increase in left atrial size that has been associated with HRV could suggest a role for HRV in AF pathogenesis that is mediated by the left atrium [13]. Moreover, autonomic imbalance could trigger an inflammatory response that can subsequently lead to AF [12]. Finally, the effect of the GWAS-identified HRV SNPs on the genes (especially, GNG11, RGS6) that are preferentially expressed within the sinoatrial node underlines the genetic basis that potentially underlies the association between HRV and AF. In short, these genes may affect acetylcholine release of the vagal nerves within the sinoatrial node and thereby influence HRV [16]. More specifically, GNG11 codes for the γ11 subunit of the heterotrimeric G-protein complex Gαβγ and may cause a decreased expression of this subunit [16]. This lower availability of this subunit may then reduce Gβγ induced GIRK activation. This potentially blunts heart rate changes caused by oscillatory changes in cardiac vagal activity, ultimately decreasing HRV [16]. Furthermore, RGS6 regulates the heterotrimeric G-protein complex signaling type 6 and may increase its availability. This leads to a decreased GIRK activation and potentially blunts the effects in cardiac vagal activation, and may thereby decrease HRV [16]. Subsequently, it has been suggested that sinus node disease (SND) may cause AF by promoting atrial extrasystoles, and re-entry [40, 41]. Atrial extrasystoles may occur during the slow atrial cycle in the presence of SND. Atrial extrasystoles are mostly followed by a compensatory pause. The pause may then be prolonged which allows other atrial ectopic activity to arise which possibly triggers AF [40]. Early premature beats that originate from areas other than the sinus node may result in conduction block and initiate re-entry, which may be a mechanism underlying AF [40]. Furthermore, stenosis in the sinus nodal artery is also common in patients with AF which implies that ischemic damage to the sinus node alone without atrial fibrosis, stretch or muscle loss may result in AF [40]. Overall, a combination of atrial extrasystoles, re-entry, and ischemia to the sinus node are mechanisms by which SND may cause and promote AF.
We investigated the longitudinal measures of HRV during a long follow-up time in relation to new-onset AF. Taking into account repeated measurements of HRV in relation to new-onset AF may provide more insight and prognostic information over a single baseline measurement that has been done by most of the previous studies [5,6,7,8,9,10, 13]. Longitudinal measures of HRV during follow-up were associated with an increased risk of incident AF, especially among women. These findings extend previous evidence by simultaneously evaluating the repeated measurements of uncorrected and corrected HRV, heart rate, and sex differences while investigating the link between HRV and AF [5,6,7,8,9,10, 13]. To some extent our findings support the association between heart rate and AF that has been previously reported in observational, [6, 7, 42] and Mendelian randomization studies [43]. However, we only found a significant association for heart rate in association with AF among women. One potential explanation could be differences in sex hormones. It has been demonstrated that an acute ovarian hormone withdrawal induced by oophorectomy leads to decline in different measures of HRV (SDNN, RMSSD), and an increase in heart rate in women [44]. The same study also showed that estrogen replacement therapy for three months within the oophorectomized women restored the HRV and heart rate to a pre-surgery level [44]. This might explain why uncorrected HRV and heart rate were only associated with incident AF in women, and not in men, in our study. We further hypothesize that competing risk of death is a possible explanation for the observed sex differences. AF is strongly associated with age, [1,2,3] so it is likely that men die of other (cardiovascular) diseases before development of AF. This hypothesis was supported by our competing risk analyses which showed that SDNN, RMSSD, RMSSDc, and heart rate were significantly associated with mortality, especially among men. Nevertheless, we found a higher incidence of AF in men, than in women in our study.
Our MR approach sheds light on the causality of the association between HRV and AF. Our effect estimates were more or less in line with previous observational studies. However, we were unable to assess the association between SDNNc, RMSSDc and AF, since not enough instrumental variables for SDNNc and RMSSDc were available to be used for the MR analyses. Future GWAS with a larger sample size could identify new additional genetic variants that could be used to assess the association between heart rate corrected HRV and AF. This could be of importance, because of the strong inverse association that exists between HRV and heart rate [24, 25]. This relation is further underlined by Nolte et al. who showed attenuation in the HRV SNP associations when they corrected for heart rate [16]. This might imply that uncorrected measures of HRV may be, in part, confounded by heart rate. Although, we showed that heart rate itself was not significantly associated with new-onset AF (except in women), we cannot rule out the possibility that heart rate is the overall determining factor instead of HRV after all. Since our uncorrected measures of HRV were indeed not significantly associated with new-onset AF. Further, as heart rate is also associated with AF and cardiovascular mortality proper adjustment for heart rate is of importance [26, 43, 45, 46]. However, excluding a genetic variant that was also associated with heart rate, a potential confounder or horizontal mediator, did not substantially change our MR results. Future studies on HRV measures corrected for heart rate could further aid in elucidating the exact mechanisms underlying HRV and AF.
The major strengths of this study are its population-based nature, large sample size with detailed information on cardiovascular risk factors, meticulous adjudication of incident AF and long follow-up time, multiple sensitivity analyses including complete-case analyses, excluding prevalent and incident CHD prior to AF diagnosis, use of competing risk analyses to compute cause-specific hazards, and use of large-scale GWAS summary statistics. The availability of repeated measurements for different HRV measures during follow-up also enabled us to investigate longitudinal measures of HRV in association with new-onset AF in a joint modeling approach which may provide more insight and give more prognostic information over a single baseline measurement. Moreover, using a MR approach we were able to gain more insight in the complex interaction between HRV and AF and to avoid certain biases that are more common in traditional observational epidemiological studies, such as residual confounding and reverse causation [15]. However, our study also has some limitations that should be taken into consideration. Our HRV measures were based on 10-s ECGs, although HRV guidelines recommend that HRV measures are based on preferably 5-min or 24-h ECG recordings [22]. Nevertheless, 10-s ECGs are more commonly performed in healthcare, are cheaper, are faster, and thereby more patient friendly than longer ECG recordings. Additionally, HRV measures from 10-s ECGs have already been associated with left ventricular function, [47] heart failure, [47, 48] cardiac-[49] and all-cause mortality [50]. Additionally, other studies that investigated the reliability of 10-s ECGs in comparison to 5-min ECGs to assess HRV showed that 10-s ECGs are also a reliable tool for HRV risk assessment, in particular within population-based studies [51, 52]. We could not distinguish between paroxysmal, persistent, long-term persistent, and permanent AF as Holter monitoring has not been done in this large population-based cohort. In our MR analyses, we cannot rule out unobserved horizontal pleiotropy, although we tried to address horizontal pleiotropy using multiple MR sensitivity analyses, such as MR-Egger, WME, MR-PRESSO, and sensitivity plots, to identify and correct for horizontal pleiotropy. Additionally, not enough sex-stratified SNPs were available in the publically available genetic dataset to perform the MR for men and women separately. Furthermore, there was partial overlap in the samples that were used to obtain the genetic instruments which may cause bias toward observational findings [33]. However, the potential bias was probably negligible given that the maximum potential overlap was 2.1%. Finally, our findings may not be generalizable to younger populations and other ethnicities, as our analysis included mainly older participants from European descent.
In conclusion, longitudinal measures of SDNN, RMSSD, but not SDNNc, RMSSDc, and heart rate, were significantly associated with new-onset AF. In sex-stratified analyses, RMSSD among men and SDNN, RMSSD, and heart rate among women were significantly associated with new-onset AF. MR analysis confirmed the complex association between HRV and AF that has been indicated by our and previous observational studies. These findings indicate that measures to modulate HRV might prevent AF in the general population, especially among women, but future MR studies that investigate the causality between heart rate corrected measures of HRV and AF are warranted.
Data availability
For the longitudinal study, data can be obtained upon request. Requests should be directed toward the management team of the Rotterdam Study (secretariat.epi@erasmusmc.nl), which has a protocol for approving data requests. Because of restrictions based on privacy regulations and informed consent of the participants, data cannot be made freely available in a public repository. For the Mendelian randomization study, genetic variants associated with HRV, used as instrumental variables for the MR analyses, were retrieved from publically available summary statistics from the 2 used GWAS.
References
Chugh SS, Havmoeller R, Narayanan K, Singh D, Rienstra M, Benjamin EJ et al (2014) Worldwide epidemiology of atrial fibrillation: a Global Burden of Disease 2010 Study. Circulation 129(8):837–847
Krijthe BP, Kunst A, Benjamin EJ, Lip GY, Franco OH, Hofman A et al (2013) Projections on the number of individuals with atrial fibrillation in the European Union, from 2000 to 2060. Eur Heart J 34(35):2746–2751
Heeringa J, van der Kuip DA, Hofman A, Kors JA, van Herpen G, Stricker BH et al (2006) Prevalence, incidence and lifetime risk of atrial fibrillation: the Rotterdam study. Eur Heart J 27(8):949–953
Hindricks G, Potpara T, Dagres N, Arbelo E, Bax JJ, Blomstrom-Lundqvist C et al (2021) 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J 42(5):373–498
Khan AA, Lip GYH, Shantsila A (2019) Heart rate variability in atrial fibrillation: the balance between sympathetic and parasympathetic nervous system. Eur J Clin Invest 49(11):e13174
Agarwal SK, Norby FL, Whitsel EA, Soliman EZ, Chen LY, Loehr LR et al (2017) Cardiac autonomic dysfunction and incidence of atrial fibrillation: results from 20 years follow-up. J Am Coll Cardiol 69(3):291–299
Habibi M, Chahal H, Greenland P, Guallar E, Lima JAC, Soliman EZ et al (2019) Resting heart rate, short-term heart rate variability and incident atrial fibrillation (from the Multi-Ethnic Study of Atherosclerosis (MESA)). Am J Cardiol 124(11):1684–1689
Perkiomaki J, Ukkola O, Kiviniemi A, Tulppo M, Ylitalo A, Kesaniemi YA et al (2014) Heart rate variability findings as a predictor of atrial fibrillation in middle-aged population. J Cardiovasc Electrophysiol 25(7):719–724
Singh JP, Larson MG, Levy D, Evans JC, Tsuji H, Benjamin EJ (2004) Is baseline autonomic tone associated with new onset atrial fibrillation?: Insights from the framingham heart study. Ann Noninvasive Electrocardiol 9(3):215–220
Kalisnik JM, Avbelj V, Vratanar J, Santarpino G, Gersak B, Fischlein T et al (2019) Cardiac autonomic regulation and PR interval determination for enhanced atrial fibrillation risk prediction after cardiac surgery. Int J Cardiol 289:24–29
Ernst G (2017) Heart-rate variability-more than heart beats? Front Public Health 5(5):240
Sajadieh A, Nielsen OW, Rasmussen V, Hein HO, Abedini S, Hansen JF (2004) Increased heart rate and reduced heart-rate variability are associated with subclinical inflammation in middle-aged and elderly subjects with no apparent heart disease. Eur Heart J 25(5):363–370
Friedman HS (2004) Heart rate variability in atrial fibrillation related to left atrial size. Am J Cardiol 93(6):705–709
Ko D, Rahman F, Schnabel RB, Yin X, Benjamin EJ, Christophersen IE (2016) Atrial fibrillation in women: epidemiology, pathophysiology, presentation, and prognosis. Nat Rev Cardiol 13(6):321–332
Smith GD, Ebrahim S (2003) “Mendelian randomization”: can genetic epidemiology contribute to understanding environmental determinants of disease? Int J Epidemiol 32(1):1–22
Nolte IM, Munoz ML, Tragante V, Amare AT, Jansen R, Vaez A et al (2017) Genetic loci associated with heart rate variability and their effects on cardiac disease risk. Nat Commun 8:15805
Nielsen JB, Thorolfsdottir RB, Fritsche LG, Zhou W, Skov MW, Graham SE et al (2018) Biobank-driven genomic discovery yields new insight into atrial fibrillation biology. Nat Genet 50(9):1234–1239
Christophersen IE, Rienstra M, Roselli C, Yin X, Geelhoed B, Barnard J et al (2017) Large-scale analyses of common and rare variants identify 12 new loci associated with atrial fibrillation. Nat Genet 49(6):946–952
Ikram MA, Brusselle G, Ghanbari M, Goedegebure A, Ikram MK, Kavousi M et al (2020) Objectives, design and main findings until 2020 from the Rotterdam Study. Eur J Epidemiol 35(5):483–517
Leening MJ, Kavousi M, Heeringa J, van Rooij FJ, Verkroost-van Heemst J, Deckers JW et al (2012) Methods of data collection and definitions of cardiac outcomes in the Rotterdam Study. Eur J Epidemiol 27(3):173–185
van Bemmel JH, Kors JA, van Herpen G (1990) Methodology of the modular ECG analysis system MEANS. Methods Inf Med 29(4):346–353
Malik M (1996) Heart rate variability. Standards of measurement, physiological interpretation, and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Eur Heart J 17(3):354–381
Gasior JS, Sacha J, Jelen PJ, Pawlowski M, Werner B, Dabrowski MJ (2015) Interaction between heart rate variability and heart rate in pediatric population. Front Physiol 6:385
Monfredi O, Lyashkov AE, Johnsen AB, Inada S, Schneider H, Wang R et al (2014) Biophysical characterization of the underappreciated and important relationship between heart rate variability and heart rate. Hypertension 64(6):1334–1343
Sacha J (2014) Interaction between heart rate and heart rate variability. Ann Noninvasive Electrocardiol 19(3):207–216
van den Berg ME, Rijnbeek PR, Niemeijer MN, Hofman A, van Herpen G, Bots ML et al (2018) Normal values of corrected heart-rate variability in 10-second electrocardiograms for all ages. Front Physiol 9:424
Rizopoulos D (2012) Joint models for longitudinal and time-to-event data With applications in R. Chapman and Hall, London
Sv B, Groothuis-Oudshoorn K (2011) MICE: multivariate imputation by chained equations in R. J Stat Softw 45(3):1–67
Burgess S, Scott RA, Timpson NJ, Davey Smith G, Thompson SG, Consortium E-I (2015) Using published data in Mendelian randomization: a blueprint for efficient identification of causal risk factors. Eur J Epidemiol 30(7):543–552
Hemani G, Zheng J, Elsworth B, Wade KH, Haberland V, Baird D et al (2018) The MR-Base platform supports systematic causal inference across the human phenome. Elife 7:e34408
Verbanck M, Chen CY, Neale B, Do R (2018) Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat Genet 50(5):693–698
Bowden J, Davey Smith G, Haycock PC, Burgess S (2016) Consistent estimation in mendelian randomization with some invalid instruments using a weighted median estimator. Genet Epidemiol 40(4):304–314
Burgess S, Davies NM, Thompson SG (2016) Bias due to participant overlap in two-sample Mendelian randomization. Genet Epidemiol 40(7):597–608
Yavorska OO, Burgess S (2017) MendelianRandomization: an R package for performing Mendelian randomization analyses using summarized data. Int J Epidemiol 46(6):1734–1739
Broadbent JR, Foley CN, Grant AJ, Mason AM, Staley JR, Burgess S (2020) Mendelian Randomization v0.5.0: updates to an R package for performing Mendelian randomization analyses using summarized data. Wellcome Open Res 5:252
Rizopoulos D, Papageorgiou G, Miranda Afonso P (2021) JMbayes2: extended joint models for longitudinal and time-to-event data. https://cran.r-project.org/web/packages/JMbayes2/JMbayes2.pdf
Team R, Core (2020) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Miller JD, Aronis KN, Chrispin J, Patil KD, Marine JE, Martin SS et al (2015) Obesity, exercise, obstructive sleep apnea, and modifiable atherosclerotic cardiovascular disease risk factors in atrial fibrillation. J Am Coll Cardiol 66(25):2899–2906
Carnethon MR, Prineas RJ, Temprosa M, Zhang ZM, Uwaifo G, Molitch ME et al (2006) The association among autonomic nervous system function, incident diabetes, and intervention arm in the Diabetes Prevention Program. Diabetes Care 29(4):914–919
Kezerashvili A, Krumerman AK, Fisher JD (2008) Sinus node dysfunction in atrial fibrillation: cause or effect? J Atr Fibrillation 1(3):30
Jackson LR 2nd, Rathakrishnan B, Campbell K, Thomas KL, Piccini JP, Bahnson T et al (2017) Sinus node dysfunction and atrial fibrillation: a reversible phenomenon? Pacing Clin Electrophysiol 40(4):442–450
Okin PM, Wachtell K, Kjeldsen SE, Julius S, Lindholm LH, Dahlof B et al (2008) Incidence of atrial fibrillation in relation to changing heart rate over time in hypertensive patients: the LIFE study. Circ Arrhythm Electrophysiol 1(5):337–343
Larsson SC, Drca N, Mason AM, Burgess S (2019) Resting heart rate and cardiovascular disease. Circ Genom Precis Med 12(3):e002459
Mercuro G, Podda A, Pitzalis L, Zoncu S, Mascia M, Melis GB et al (2000) Evidence of a role of endogenous estrogen in the modulation of autonomic nervous system. Am J Cardiol 85(6):787–789
Liu X, Guo N, Zhu W, Zhou Q, Liu M, Chen C et al (2019) Resting heart rate and the risk of atrial fibrillation. Int Heart J 60(4):805–811
Zhang D, Shen X, Qi X (2016) Resting heart rate and all-cause and cardiovascular mortality in the general population: a meta-analysis. CMAJ 188(3):E53–E63
Arshi B, Geurts S, Tilly MJ, van den Berg M, Kors JA, Rizopoulos D et al (2022) Heart rate variability is associated with left ventricular systolic, diastolic function and incident heart failure in the general population. BMC Med 20(1):91
Rautaharju PM, Kooperberg C, Larson JC, LaCroix A (2006) Electrocardiographic predictors of incident congestive heart failure and all-cause mortality in postmenopausal women: the Women’s Health Initiative. Circulation 113(4):481–489
de Bruyne MC, Kors JA, Hoes AW, Klootwijk P, Dekker JM, Hofman A et al (1999) Both decreased and increased heart rate variability on the standard 10-second electrocardiogram predict cardiac mortality in the elderly: the Rotterdam Study. Am J Epidemiol 150(12):1282–1288
Dekker JM, Schouten EG, Klootwijk P, Pool J, Swenne CA, Kromhout D (1997) Heart rate variability from short electrocardiographic recordings predicts mortality from all causes in middle-aged and elderly men the Zutphen Study. Am J Epidemiol 145(10):899–908
Baek HJ, Cho CH, Cho J, Woo JM (2015) Reliability of ultra-short-term analysis as a surrogate of standard 5-min analysis of heart rate variability. Telemed J E Health 21(5):404–414
Ernst G (2017) Hidden signals-the history and methods of heart rate variability. Front Public Health 5:265
Acknowledgements
The authors are grateful for the dedication, commitment and contribution of the study participants, the general practitioners, pharmacists and the staff from the Rotterdam Study. Furthermore, the authors would like to thank the Genetic Variance in Heart Rate Variability (VgHRV), HUNT, deCODE, MGI, DiscovEHR, UK Biobank, AFGen Consortium, and individual studies for sharing their summary statistics in GWAS.
Funding
The Rotterdam Study is funded by Erasmus Medical Center and Erasmus University, Rotterdam, the Netherlands Organization for the Health Research and Development (ZonMw), the Research Institute for Diseases in the Elderly (RIDE), the Ministry of Education, Culture and Science, the Ministry for Health, Welfare and Sports, the European Commission (DG XII), and the Municipality of Rotterdam. This study is further supported by the Senior Scientist Grant from Dutch Heart Foundation (03-004-2021-T050).
Author information
Authors and Affiliations
Contributions
Conceptualization, SG and MK; Data curation, SG, MT, BA and MK; Formal analysis, SG; Funding acquisition, MK and MAI; Investigation, SG, MT, BA, BS, JK, JD, NG, MK and MAI; Methodology, SG, MT, BA and MK; Project administration, SG and MK; Resources, BS, JK, JD, MK and MAI; Software, SG; Supervision, MK; Validation, SG and MK; Visualization, SG; Writing—original draft, SG and MK; Writing—review & editing, SG, MT, BA, BS, JK, JD, NG, MK and MAI.
Corresponding author
Ethics declarations
Conflict of interest
Ikram reports consulting fees from BioGen Inc. The remaining authors have no disclosures to report.
Institutional Review Board Statement
The Rotterdam Study complies with the Declaration of Helsinki and has been approved by the Medical Ethics Committee of the Erasmus MC (registration number MEC 02.1015) and by the Dutch Ministry of Health, Welfare and Sport (Population Screening Act WBO, license number 1071272–159521-PG). The Rotterdam Study has been entered into the Netherlands National Trial Register (NTR; www.trialregister.nl/trials/) and into the WHO International Clinical Trials Registry Platform (ICTRP; https://apps.who.int/trialsearch/) under shared catalog number NL6645/NTR6831.
Informed Consent Statement
For the longitudinal study, all participants provided written informed consent to participate, prior to inclusion, in the study and to have their information obtained from treating physicians. The Mendelian randomization study has been conducted using publicly available summary statistics, so no original data were collected for this bidirectional MR study. Ethical approval and informed consent from each participant in the studies included in the investigation can be found in the original publications. The analysis of other publicly available summary statistics does not require additional ethical approval.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Geurts, S., Tilly, M.J., Arshi, B. et al. Heart rate variability and atrial fibrillation in the general population: a longitudinal and Mendelian randomization study. Clin Res Cardiol 112, 747–758 (2023). https://doi.org/10.1007/s00392-022-02072-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00392-022-02072-5