Abstract
Aims
This review aims to evaluate the feasibility of robot-assisted laparoscopic surgery (RALS) as an alternative to standard laparoscopic surgery (SLS) for the treatment of bowel deep-infiltrative endometriosis. Additionally, it aims to provide guidance for future study design, by gaining insight into the current state of research, in accordance with the IDEAL framework.
Method
A systematic review was conducted to identify relevant studies on RALS for bowel deep infiltrating endometriosis in Medline, Embase, Cochrane Library and PubMed databases up to August 2023 and reported in keeping with PRISMA guidelines. The study was registered with PROSPERO Registration: CRD42022308611
Results
Eleven primary studies were identified, encompassing 364 RALS patients and 83 SLS patients, from which surgical details, operative and postoperative outcomes were extracted. In the RALS group, mean operating time was longer (235 ± 112 min) than in the standard laparoscopy group (171 ± 76 min) (p < 0.01). Patients in the RALS group experienced a shorter hospital stay (5.3 ± 3.5 days vs. 7.3 ± 4.1 days) (p < 0.01), and appeared to have fewer postoperative complications compared to standard laparoscopy. Research evidence for RALS in bowel DE is at an IDEAL Stage 2B of development.
Conclusion
RALS is a safe and feasible alternative to standard laparoscopy for bowel endometriosis treatment, with a shorter overall length of stay despite longer operating times. Further robust randomized trials recommended to delineate other potential advantages of RALS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Endometriosis is a chronic condition characterized by the presence of endometrial-like tissue outside the uterus, affecting around 10% of women of childbearing age [1, 2]. The ectopic endometrial tissue responds to hormonal fluctuations during the menstrual cycle, leading to pain, inflammation and adhesion formation in affected organs [3]. Clinical presentations can vary, but women with endometriosis commonly present with pelvic pain, dysmenorrhea and infertility [1].
Deep infiltrative endometriosis (DE) [4] is defined as lesions with ≥ 5-mm penetration into the peritoneum or lesions that invade the muscularis propria of surrounding viscera, affecting more than 20% of women with said condition [2, 5]. Among the known sites of DE, bowel involvement is reported in 5–12% of women with endometriosis and can be associated with additional symptoms such as abdominal pain, hematochezia, and dyschezia [6]. A substantial subset of this, ranging from 70 to 93%, exhibits rectosigmoid involvement [5, 7].
The primary approach for managing symptoms of bowel deep infiltrating endometriosis [4] involves medical therapy, including non-steroidal anti-inflammatory drugs, oral contraceptives and progestins [6, 8]. Once medical options have been exhausted, surgical removal has been considered as the next step in the treatment pathway, with standard laparoscopic surgery (SLS) being the current standard approach [1, 6, 9]. Various surgical techniques have been described for treating bowel DE, including rectal shaves, discoid excisions or segmental resections.
Each of the above techniques involves an increasing degree of surgical complexity, exposing patients to the risk of more significant complications. Therefore, the selection of the surgical technique depends on the extent, length and depth of bowel involvement and may vary depending on preferences and experience of the surgeon involved [6, 9,10,11].
Robot-assisted laparoscopic surgery (RALS) has been shown to be a viable alternative to conventional laparoscopy, providing technical advantages such as improved dexterity and ergonomics, increased range of motion, three-dimensional visualization and elimination of tremors [1, 9, 11]. However, arguments for the feasibility of RALS over standard laparoscopy in the treatment of symptomatic Bowel DE remains limited in the literature, confounded with discrepancies in outcomes reporting.
The main aim of this review is to collate and examine the existing data on RALS in the treatment of bowel DE among symptomatic patients. This review aims to assess surgical procedural details, operative parameters and postoperative outcomes to provide insights into the feasibility of the RALS approach for this population.
Additionally, this review also aims to evaluate the developmental status of the RALS technique based on the IDEAL framework. The framework describes five stages—Idea, Development, Exploration, Assessment and Long-term study—in the evaluation of emerging surgical techniques, determined by the types of studies already published on said technique [12,13,14]. Understanding the current developmental stage allows researchers to identify gaps in the current evidence base and guides the direction of future research in this space.
Methods
This review was designed according to PRISMA guidelines [15] and was submitted to the PROSPERO database, CRD42022308611. The search was last accessed in August 2023, of all publications in Medline, Embase, PubMed and Cochrane Database, reporting on the outcomes of robot-assisted laparoscopic interventions for bowel DE. Table 1 shows the key terms and Boolean operators used for the search in the title and abstract fields. A manual search for relevant studies was also carried out to identify additional studies.
Inclusion and exclusion criteria
Primary studies published up to August 2023 were considered for inclusion to ensure the relevance and timeliness of the findings. This review considered studies discussing RALS procedures for treating bowel DE, including rectal shaves, discoid excisions and segmental resections. These studies can either be non-comparative (RALS only) or comparative studies (RALS vs. SLS). The review population was restricted to symptomatic patients diagnosed with bowel DE through clinical evaluations and/or imaging.
Studies with an emphasis on standard laparoscopic or laparotomy interventions only and those examining DE which did not involve bowel were excluded from consideration. Additionally, literature reviews, conference abstracts, single-case reports and studies not providing outcomes of interest were excluded.
Study characteristics and outcomes of interest
The assessment of the study characteristics included variables such as authorship, publication year, geographical origin, study design, sample size, procedures of interest and key findings. Furthermore, outcomes from each study were extracted using a prospectively designed collection sheet, encompassing technical details, intraoperative parameters and postoperative outcomes.
Risk of bias assessment
The methodological quality and risk of bias in included studies were assessed using the Joanna-Briggs Institute (JBI) appraisal tools for case series [16] and cohort studies [17], summarised in Table 8.
Data extraction and statistical analysis
Two independent reviewers (PN, NS) conducted title and abstract screening, full-text inclusion and data extraction, with disputes settled with another reviewer (HO) if consensus was not reached. Data were summarized in a narrative synthesis and the chi-squared test, Fisher’s exact test and Student’s t-test were used for the comparison of categorical and numerical variables, where appropriate. In cases where variables were presented in the form of median/range, the mean/standard deviation were estimated employing methods outlined by Luo et al. [18] and Wan et al. [19]. All reported p-values were two-sided, and a significant difference was noted when p < 0.05. Statistical analysis was performed using commercially available software.
Results
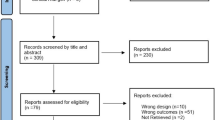
The steps for identifying studies for this review were summarized in the PRISMA diagram (Fig. 1). The initial search resulted in 223 studies. After the removal of duplicates, 192 studies underwent title and abstract screening, leaving 25 full-text studies to be reviewed. A further 14 were excluded for reasons highlighted in Fig. 1, leaving 11 studies [20,21,22,23,24,25,26,27,28,29,30] to be included in the review.
Study characteristics
Table 2 compiles the data describing the studies’ characteristics. In summary, the study range spanned from 2012 to 2022. Among these studies, nine studies [20,21,22,23,24,25,26,27,28] were non-comparative case series focusing on RALS, whereas two studies [29, 30] were comparative cohort studies. Overall, the studies included 527 participants, of which 451 received surgical treatment for bowel DE (which may have also included concurrent removal of other sites of disease)—consisting of 368 RALS and 83 SLS participants. All participants who had treatment for DE which did not involve bowel were excluded.
Surgical procedural details
Nine studies reported the utilization of the Da Vinci System (Intuitive Surgical, Inc.) as their robotic platform (Table 3), with Ercoli et al. likely to have also used this system in 2010. Where patient positioning was described, most were positioned in lithotomy, a configuration commonly used in gynecological, rectal and urological procedures [31]. The depictions of port placements were mostly consistent with what has been described in existing literature [11]. For the two comparative studies [29, 30], the assignment of patients into either the RALS or SLS approach did not follow a formal randomization process, as it solely depended on the availability of the robotic theatre and equipment.
In addition, all studies provided insight into at least one of the following techniques: rectal shaves, discoid excisions and segmental resections. Most studies provided indications regarding the characteristics of endometrial lesions for each procedure (Table 3). However, the reporting of indications was not standardized, as each paper puts forward its own distinct criteria. As depicted in Fig. 2, among the participants who underwent RALS and SLS for bowel DE, 59% and 35% had shaves, 8% and 19% had discoid excisions, and 33% and 42% had segmental resections.
Surgeon experience
When interpreting the results from these studies, it is important to consider the surgical experience, particularly in the RALS group, as many of these studies may represent the surgical learning curve of this new technology.
There was significant heterogeneity in the disclosure and experience of surgeons participating in these studies, as summarized in Table 4, but a trend towards more explicit disclosure of surgical robotic experience can be seen in more recent studies. As expected, all surgeons involved were also trained in laparoscopic surgery.
Operative and postoperative parameters
The operative and postoperative findings from each study were compiled in Table 5. Most studies reported these metrics using means and standard deviations [33] or medians and ranges. However, a few studies either omitted information on some outcomes or reported means/medians without corresponding SD/ranges, resulting in their exclusion from calculations done for Table 6. Regarding patient characteristics, the groups exhibited comparable age and body mass index profiles (Table 6). The mean operating times was 227 ± 104 in the RALS group and 171 ± 76 min in the SLS group (p < 0.01). For the remaining intraoperative metrics, there were no significant differences in blood loss, intraoperative complications and laparotomy conversions.
Regarding postoperative outcomes, the mean hospital stay was 5.1 ± 3.6 days in the robotic group and 7.3 ± 4.1 days in the standard group (p < 0.01). Postoperative complications were stratified based on the Clavien-Dindo classification system to grade adverse events occurring after a surgical procedure [34, 35]. The classification categorizes complications into grades ranging from I to V, in increasing severity. For the purposes of this study, complications were grouped into minor (I–II) and major (III–IV) complications, excluding grade V (death) as it was not reported in any studies. Overall, major and minor complications occurred in 4.4% and 6.4% of cases in the robotic group, compared to 3.6% and 22.9% in the standard group. Anastamotic leak rate of 0.6% in the RALS group (n = 2) lies within acceptable limits. It is important to note that as the overall number of patients were low, none of these differences reached statistical significance.
From a functional perspective, 4 studies reported significant improvements in dysmenorrhoea, dyspareunia, dyschezia and chronic pelvic pain within the RALS group at least 6 months post operatively [20, 21, 25, 29] according to the 10 point VAS scale. Raimondo et al. also found an equal improvement when compared to SLS [29]. Morelli et al. found an improvement in quality of life at 6 months [26] and Abo et al. found improvements in bowel function up to 24 months post operatively [27]. However, the heterogeneity of these measures precluded statistical analysis within the context of this systematic review.
Discussion
Minimally invasive laparoscopy has been the standard in the surgical treatment of endometriosis, including deep infiltrative lesions of the bowel [1, 6, 9]. However, there are known constraints to standard laparoscopy, which include the loss of three-dimensional visualization, reduced degrees of movement and the fulcrum effect [36]. The introduction of robotic surgical platforms aimed to address some of these limitations [36, 37]. While meta-analyses [38, 39] established the feasibility of robotic surgery as an alternative to standard laparoscopy for treating endometriosis, none addressed it in the context of bowel DE. A review by Hur and Falcone [11] had a similar focus, providing a general overview of the role of RALS in bowel endometriosis. However, this present study placed an additional emphasis on conducting a comprehensive review in accordance with the PRISMA guidelines and comparative analysis of data using descriptive statistics.
Choice of surgical techniques using RALS
Significant differences in the choice of surgical techniques emerged between RALS and SLS, with rectal shaves more frequently utilized with RALS (Table 6). The higher prevalence of shaves in the robotic group could be attributed to the benefits associated with robot-assisted surgery, which offers better vision to aid in assessment of disease depth, allowing surgeons to perform complete excisions with clear margins with rectal shaves, and improved dexterity enabling easier and secure suturing within the confines of the pelvis. When more extensive resection is performed, the technical complexities involved may diminish some of the advantages offered by robotic surgery.
Another factor to consider is the inherent variation of surgical indications described in the studies. The choice of technique itself is dependent upon several factors, including the extent and depth of bowel involvement [6, 9,10,11]. This inherent variability in decision-making reflects the multitude of factors which influence the personalized nature of treatment decisions.
The decision to perform segmental resections balance a lower recurrence risk against the increased risk of major complications, particularly anastomotic leak and requirement for stoma. In the cohort of patients reviewed, 2 anastomotic leaks were identified, both within the RALS group. This could be explained by a number of factors. Sampling bias is likely to have played a significant role in this finding, particularly in the SLS group, where only 35 patients underwent segmental resection. A large multicenter meta-analysis, including 1622 patients puts this risk at 1.9% [40]. Comparatively, the rate of 0.6% in the RALS group is within acceptable limits. Furthermore, as these cohort studies are unblinded, selection bias may be present, where more complex cases were undertaken when the robotic system is available, as acknowledged by Graham et al. [28].
However, the increased prevalence of segmental resections within the SLS cohort itself introduces a potential confounding variable, given its association with prolonged surgical duration and an increased risk of postoperative complications [41, 42]. These factors can naturally influence other surgical outcomes, such as recovery times and complication rates.
Operative and postoperative outcomes of RALS
This review suggests that RALS is feasible and safe for treating symptomatic patients with bowel deep infiltrating endometriosis, leading to minimal surgical complications and infrequent conversion to laparotomy. Additionally, there is a trend towards fewer intraoperative complications in the robotic group, although this has not reached clinical significance (Table 6). One study [26] even associates RALS with the preservation of urinary function and sexual well-being up to a year post-surgery. Intraoperative blood loss, another important metric influencing surgical complications, was likewise found to exhibit no significant differences between the groups. The studies included in this review indicate that RALS confers at least an equal benefit in terms of resolution of preoperative symptoms to SLS, but true statistical analysis was not possible due to the heterogeneity of the data.
However, when compared to standard laparoscopy, RALS is associated with significantly longer operating duration, consistent with other reports within the existing literature. Numerous reviews and randomized trials have explored the differences in operating times between RALS and SLS for various subsets of endometriosis patients [38, 39, 43]. While many of these studies reached similar conclusions regarding extended operating times with RALS, there were notable variations in the reported data. Factors contributing to these variations include the selective exclusion of patients with bowel involvement or inclusions of superficial endometriosis in these studies, leading to shorter reported operating times. This complexity underscores the importance of considering specific study contexts and inclusion criteria when interpreting data on operating times.
Postoperatively, the robotic group exhibits a shorter hospital stay, with differences in postoperative complication rates not reaching statistical significance (Table 6). This is likely attributable to a smaller sample size, as compared to larger studies (368 and 83 vs 4721) [44]. Furthermore, many of the included studies acknowledged the learning curve experienced in performing robotic surgery, compared to their significant laparoscopic experience.
In addition, it is important to note that the observational nature of these included studies inherently provides evidence of lower quality compared to RCTs [45]. These studies, due to the lack of controlled interventions, have limitations in their ability to establish causal relationships.
Translation into clinical practice
Prolonged operating time can largely be attributed to the unique characteristics of robotic surgery, with additional time required for tasks such as docking, robot positioning and troubleshooting technical issues [11, 36]. Previous research [46] describing surgical treatments of advanced endometriosis had shown that longer durations of surgery potentially increase the physiological stress on the patient, adversely affecting recovery times and postoperative outcomes.
Another factor to consider is that operating room time is a valuable resource in healthcare. Prolonged surgeries consume more resources, including personnel, equipment and facilities—on top of the costs incurred for implementing and maintaining the robotic equipment [47]. Consequently, this can result in higher immediate costs, which may be offset by the significantly shorter length of stay and lower rates of surgical complications. Furthermore, as the use of the robotic systems mature, it is expected that the operating time will decrease as the learning curve is overcome by both the surgeons and their supporting theatre staff.
In summary, from a practical perspective, the availability of a robotic system can pose a barrier to the adoption of RALS due to its high start-up costs. Nevertheless, as demonstrated in this review, there are potential benefits to using RALS which can only be fully understood through increased availability and utilization of robotic operating systems within the healthcare system. It is important to note that comprehensive cost-benefit analyses specific to RALS in treating bowel DE have been lacking thus far. Future research is essential to provide additional perspectives on the medico-economic implications associated with RALS.
IDEAL staging of RALS for bowel DE
The concept of the IDEAL framework was developed in 2009 by McCulloch et al. to evaluate new and emerging surgical innovation. The life cycle of an innovation is described in 5 stages; Idea, Development, Exploration, Assessment and Long-term Study. Criteria for these 5 stages were developed to evaluate the safety, efficacy and effectiveness of an innovation, and were derived from a Delphi survey, expert workshop and discussions at IDEAL conferences held in Oxford (2016) and New York (2017), with ongoing conferences to update these recommendations [12,13,14].
The evaluation of RALS for the treatment of bowel DE reveals a dynamic landscape of evolving surgical techniques and research methodologies as evidenced by the studies highlighted in this review. In the early 2010s, studies [20,21,22,23,24,25,26,27,28] on this topic predominantly featured single-centre and/or retrospective case series, indicative of the IDEAL Stage 2a (Development) [48]. These studies sought to establish the viability and safety of RALS for bowel DE, along with building consensus on the technical aspects of the procedure and sharing early clinical experiences.
However, recent papers [29, 30] on the technique have transitioned into Stage 2b (Exploration), characterized by prospective, multicentric cohort comparative studies. This evolution reflects the maturity of the technique, supported by robust findings indicating its feasibility and safety, and is summarised in Table 7. Studies directly comparing RALS against standard laparoscopy are emerging, shedding light on the learning curves associated with RALS and highlighting the necessity for RCTs to provide more definitive evidence. Based on these considerations, the overall staging of RALS for bowel DE can be classified as the endpoint of stage 2b. This signifies a consensus on the maturity of the technique, favourable clinical outcomes and an understanding of desirable outcome measures [48].
Looking ahead, it is imperative that multicentre RCTs be undertaken to provide higher-quality evidence to investigate the comparative effectiveness of RALS versus SLS. Notably, the upcoming ROBEndo RCT [49] represents a promising step in this direction and holds the potential to influence future research in this field.
Furthermore, the lack of studies reporting on more specific outcomes such as urinary and sexual function, cost analysis and fertility means there are many avenues to explore for further research as the safety profile of RALS becomes increasingly established.
Limitations
It is important to recognize that like any literature review, this analysis may be susceptible to evidence selection, reporting and publication biases, as well as the inherent biases of the studies previously mentioned [50]. To limit the sources of these biases, several measures were put in place. Firstly, two independent reviewers performed a thorough assessment of the methodology and risk of bias in all the studies (Tables 8 and 9). Additionally, strict adherence to PRISMA guidelines ensured a structured review and reporting process.
However, the nature of the included studies inherently provides lower-quality evidence compared to prospective RCTs [45]. While these studies offer valuable insights into real-world clinical practices, the observational nature and lack of controlled interventions limit their ability to establish definitive causal relationships. Furthermore, their vulnerability to confounders presents a concern, given that this review cannot address all potential factors that might influence the outcomes, such as the surgeons’ learning curve for RALS.
There was significant heterogeneity in the studies available within the current research landscape, which precluded the performance of a formal meta-analysis. Although we performed statistical analyses which included comparable factors from these studies, the findings should be interpreted with these limitations in mind.
Conclusion
Robotic-assisted laparoscopic surgery has emerged as an alternative to conventional laparoscopy in the context of deep infiltrative endometriosis involving the bowel. Our review suggests that RALS offers potential benefits of reduced postoperative hospital stay and a trend towards lower intraoperative complications, with longer operating times. As surgeons and techniques mature, well-designed randomized studies are imperative to further define the safety and efficacy of RALS in bowel endometriosis, aligning with an IDEAL framework stage of 2b. Future studies should address the impact of the learning curve of robotic surgery, potential differences in resection margins and functional outcomes in DE, as well compare each technique; rectal shave, discoid excision and segmental resection, to define the advantages and disadvantages of each system.
Data availability
No datasets were generated or analysed during the current study.
References
Taylor HS, Kotlyar AM, Flores VA (2021) Endometriosis is a chronic systemic disease: clinical challenges and novel innovations. Lancet 397(10276):839–852
Chapron C, Marcellin L, Borghese B, Santulli P (2019) Rethinking mechanisms, diagnosis and management of endometriosis. Nat Rev Endocrinol 15(11):666–682
Alimi Y, Iwanaga J, Loukas M, Tubbs RS (2018) The clinical anatomy of endometriosis: a review. Cureus. 10(9):e3361
Allgayer H, Dietrich CF, Rohde W, Koch GF, Tuschhoff T (2005) Prospective comparison of short- and long-term effects of pelvic floor exercise/biofeedback training in patients with fecal incontinence after surgery plus irradiation versus surgery alone for colorectal cancer: clinical, functional and endoscopic/endosonographic findings. Scand J Gastroenterol 40(10):1168–1175
Daraï E, Cohen J, Ballester M (2017) Colorectal endometriosis and fertility. Eur J Obstet Gynecol Reprod Biol 209:86–94
Meuleman C, Tomassetti C, D’Hoore A, Van Cleynenbreugel B, Penninckx F, Vergote I et al (2011) Surgical treatment of deeply infiltrating endometriosis with colorectal involvement. Hum Reprod Update 17(3):311–326
Wills HJ, Reid GD, Cooper MJ, Morgan M (2008) Fertility and pain outcomes following laparoscopic segmental bowel resection for colorectal endometriosis: a review. Aust N Z J Obstet Gynaecol 48(3):292–295
Vercellini P, Crosignani PG, Somigliana E, Berlanda N, Barbara G, Fedele L (2009) Medical treatment for rectovaginal endometriosis: what is the evidence? Hum Reprod 24(10):2504–2514
Becker CM, Bokor A, Heikinheimo O, Horne A, Jansen F, Kiesel L et al (2022) ESHRE guideline: endometriosis. Hum Reprod Open 2022(2):hoac009
Donnez O, Roman H (2017) Choosing the right surgical technique for deep endometriosis: shaving, disc excision, or bowel resection? Fertil Steril 108(6):931–942
Hur C, Falcone T (2021) Robotic treatment of bowel endometriosis. Best Pract Res Clin Obstet Gynaecol 71:129–143
McCulloch P, Cook JA, Altman DG, Heneghan C, Diener MK (2013) IDEAL framework for surgical innovation 1: the idea and development stages. BMJ : Br Med J 346:f3012
Hirst A, Philippou Y, Blazeby J, Campbell B, Campbell M, Feinberg J et al (2019) No surgical innovation without evaluation: evolution and further development of the IDEAL framework and recommendations. Ann Surg 269(2):211–220
McCulloch P, Altman DG, Campbell WB, Flum DR, Glasziou P, Marshall JC et al (2009) No surgical innovation without evaluation: the IDEAL recommendations. Lancet. 374(9695):1105–1112
Tricco AC, Lillie E, Zarin W, O’Brien KK, Colquhoun H, Levac D et al (2018) PRISMA Extension for Scoping Reviews (PRISMA-ScR): checklist and explanation. Ann Intern Med 169(7):467–473
Munn Z, Barker TH, Moola S, Tufanaru C, Stern C, McArthur A et al (2020) Methodological quality of case series studies: an introduction to the JBI critical appraisal tool. JBI Evid Synth 18(10):2127–2133
Moola S, Munn Z, Sears K, Sfetcu R, Currie M, Lisy K et al (2015) Conducting systematic reviews of association (etiology): the Joanna Briggs Institute’s approach. Int J Evid Based Healthc 13(3):163–169
Luo D, Wan X, Liu J, Tong T (2018) Optimally estimating the sample mean from the sample size, median, mid-range, and/or mid-quartile range. Stat Methods Med Res 27(6):1785–1805
Wan X, Wang W, Liu J, Tong T (2014) Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol 14:135
Ercoli A, D’Asta M, Fagotti A, Fanfani F, Romano F, Baldazzi G et al (2012) Robotic treatment of colorectal endometriosis: technique, feasibility and short-term results. Hum Reprod 27(3):722–726
Cassini D, Cerullo G, Miccini M, Manoochehri F, Ercoli A, Baldazzi G (2013) Robotic hybrid technique in rectal surgery for deep pelvic endometriosis. Surg Innov 21(1):52–58
Collinet P, Leguevaque P, Neme RM, Cela V, Barton-Smith P, Hebert T et al (2014) Robot-assisted laparoscopy for deep infiltrating endometriosis: international multicentric retrospective study. Surg Endosc 28(8):2474–2479
Siesto G, Ieda N, Rosati R, Vitobello D (2014) Robotic surgery for deep endometriosis: a paradigm shift. Int J Med Robot 10(2):140–146
Diguisto C, Hébert T, Paternotte J, Kellal I, Marret H, Ouldamer L et al (2015) Robotic assisted laparoscopy: comparison of segmentary colorectal resection and shaving for colorectal endometriosis. Gynecol Obstet Fertil 43(4):266–270
Pellegrino A, Damiani GR, Trio C, Faccioli P, Croce P, Tagliabue F et al (2015) Robotic shaving technique in 25 patients affected by deep infiltrating endometriosis of the rectovaginal space. J Minim Invasive Gynecol 22(7):1287–1292
Morelli L, Perutelli A, Palmeri M, Guadagni S, Mariniello MD, Di Franco G et al (2016) Robot-assisted surgery for the radical treatment of deep infiltrating endometriosis with colorectal involvement: short- and mid-term surgical and functional outcomes. Int J Color Dis 31(3):643–652
Abo C, Roman H, Bridoux V, Huet E, Tuech JJ, Resch B et al (2017) Management of deep infiltrating endometriosis by laparoscopic route with robotic assistance: 3-year experience. J Gynecol Obstet Hum Reprod 46(1):9–18
Graham A, Chen S, Skancke M, Moawad G, Obias V (2019) A review of deep infiltrative colorectal endometriosis treated robotically at a single institution. Int J Med Robot 15(4):e2001
Raimondo D, Alboni C, Orsini B, Aru AC, Farulla A, Maletta M et al (2021) Comparison of perioperative outcomes between standard laparoscopic and robot-assisted approach in patients with rectosigmoid endometriosis. Acta Obstet Gynecol Scand 100(9):1740–1746
Ferrier C, Le Gac M, Kolanska K, Boudy AS, Dabi Y, Touboul C et al (2022) Comparison of robot-assisted and conventional laparoscopy for colorectal surgery for endometriosis: a prospective cohort study. Int J Med Robot 18(3):e2382
Armstrong M, Moore RA (2023) Anatomy, Patient Positioning. In: In: StatPearls [Internet]. StatPearls Publishing LLC., Treasure Island (FL) Available from: www.ncbi.nlm.nih.gov/books/NBK513320/
Pieniowski EHABC, Nordenvall CAM, Westberg KS, Johar AM, Tumlin Ekelund SF, Larsson KR, Pekkari KJ, Jansson Palmer GC, Lagergren P, Abraham-Nordling M (2023) A randomized controlled clinical trial of transanal irrigation versus conservative treatment in patients with low anterior resection syndrome after rectal cancer surgery. Ann Surg 277(1):30–37
Gerber LH, Hodsdon B, Comis LE, Chan L, Gallin JI, McGarvey CL 3rd. (2017) A brief historical perspective of cancer rehabilitation and contributions from the national institutes of health. PM R 9(9S2):S297–S304
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240(2):205–213
Katayama H, Kurokawa Y, Nakamura K, Ito H, Kanemitsu Y, Masuda N et al (2016) Extended Clavien-Dindo classification of surgical complications: Japan Clinical Oncology Group postoperative complications criteria. Surg Today 46(6):668–685
Catenacci M, Flyckt RL, Falcone T (2011) Robotics in reproductive surgery: strengths and limitations. Placenta. 32:S232–S2S7
Chen CCG, Falcone T (2009) Robotic gynecologic surgery: past, present, and future. Clin Obstet Gynecol 52(3):335–343
Chen S-H, Li Z-A, Du X-P (2016) Robot-assisted versus conventional laparoscopic surgery in the treatment of advanced stage endometriosis: a meta-analysis. CEOG. 43(3):422–426
Restaino S, Mereu L, Finelli A, Spina MR, Marini G, Catena U et al (2020) Robotic surgery vs laparoscopic surgery in patients with diagnosis of endometriosis: a systematic review and meta-analysis. J Robot Surg 14(5):687–694
Bendifallah SP (2021) Anne; Vesale, Elie; Moawad, Gaby; Daraï, Emile; Roman, Horace. Surgical outcomes after colorectal surgery for endometriosis: systematic review and meta-analysis. J Minim Invasive Gynecol 28(3):453–466
Daraï E, Bazot M, Rouzier R, Houry S, Dubernard G (2007) Outcome of laparoscopic colorectal resection for endometriosis. Curr Opin Obstet Gynecol 19(4):308–313
Parra RS, Valério FP, Zanardi JVC, Feitosa MR, Camargo HP, Féres O (2022) Postoperative complications and stoma rates after laparoscopic resection of deep infiltrating endometriosis with bowel involvement. Rev Bras Ginecol Obstet 44(11):1040–1046
Soto E, Luu TH, Liu X, Magrina JF, Wasson MN, Einarsson JI et al (2017) Laparoscopy vs. Robotic Surgery for Endometriosis (LAROSE): a multicenter, randomized, controlled trial. Fertil Steril 107(4):996–1002.e3
Byrne D, Curnow T, Smith P, Cutner A, Saridogan E, Clark TJ et al (2018) Laparoscopic excision of deep rectovaginal endometriosis in BSGE endometriosis centres: a multicentre prospective cohort study. BMJ Open 8(4):e018924
Gilmartin-Thomas JF, Liew D, Hopper I (2018) Observational studies and their utility for practice. Aust Prescr 41(3):82–85
Magrina JF, Espada M, Kho RM, Cetta R, Chang YH, Magtibay PM (2015) Surgical excision of advanced endometriosis: perioperative outcomes and impacting factors. J Minim Invasive Gynecol 22(6):944–950
McBride K, Steffens D, Stanislaus C, Solomon M, Anderson T, Thanigasalam R et al (2021) Detailed cost of robotic-assisted surgery in the Australian public health sector: from implementation to a multi-specialty caseload. BMC Health Serv Res 21(1):108
Pennell CP, Hirst AD, Campbell WB, Sood A, Agha RA, Barkun JS et al (2016) Practical guide to the idea, development and exploration stages of the IDEAL framework and recommendations. Br J Surg 103(5):607–615
Terho AM, Mäkelä-Kaikkonen J, Ohtonen P, Uimari O, Puhto T, Rautio T et al (2022) Robotic versus laparoscopic surgery for severe deep endometriosis: protocol for a randomised controlled trial (ROBEndo trial). BMJ Open 12(7):e063572
Drucker AM, Fleming P, Chan A-W (2016) Research techniques made simple: assessing risk of bias in systematic reviews. J Invest Dermatol 136(11):e109–ee14
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Contributions
Conception: Helen Mohan, Christina Fleming, Deirdre Nally, Uzma Mahmood, Colin Peirce. Methodology: Patrick Nugraha, Nastassia Shulman, Hwa Ian Ong, Stephen Wrenn. Data curation: Patrick Nugraha, Nastassia Shulman, Hwa Ian Ong, Stephen Wrenn. Supervision: David Proud, Adele Burgess, Christina Fleming, Helen Mohan, Uzma Mahmood, Colin Peirce, Deirdre Nally, Satish Warrier, Jacob McCormick. Resources: David Proud, Adele Burgess, Christina Fleming, Helen Mohan, Uzma Mahmood. Project administration: Hwa Ian Ong, Patrick Nugraha, Nastassia Shulman. Analysis: Patrick Nugraha, Nastassia Shulman, Hwa Ian Ong, Stephen Wrenn, Helen Mohan. Writing (original draft): Patrick Nugraha, Nastassia Shulman. Writing (editing and revision): Hwa Ian Ong, Helen Mohan, Christina Fleming, Satish Warrier. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(PDF 84 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ong, H.I., Shulman, N., Nugraha, P. et al. Role of robot-assisted laparoscopy in deep infiltrating endometriosis with bowel involvement: a systematic review and application of the IDEAL framework. Int J Colorectal Dis 39, 98 (2024). https://doi.org/10.1007/s00384-024-04669-w
Accepted:
Published:
DOI: https://doi.org/10.1007/s00384-024-04669-w