Abstract
Purpose
Body weight and preoperative weight loss (WL) are controversially discussed as risk factors for postoperative morbidity and mortality in colorectal cancer surgery. The objective of this study is to determine whether body mass index (BMI) or WL is associated with a higher postoperative complication rate.
Methods
In this retrospective cohort study, data analysis of 1241 consecutive patients undergoing colorectal cancer surgery in an 11-year period was performed. The main outcome measures were wound infections (WI), anastomotic leakages (AL), and in-house mortality.
Results
A total of 697 (56%) patients with colon and 544 (44%) with rectum carcinoma underwent surgery. The rate of WI for each location increased with rising BMI. The threshold value was 28.8 kg/m2. Obese patients developed significantly more WI than normal-weight patients did following rectal resection (18.0% vs. 8.2%, p = 0.018). Patients with preoperative WL developed significantly more AL following colon resections than did patients without preoperative WL (6.2% vs. 2.5%, p = 0.046). In-house mortality was significantly higher in obese patients following colon resections than in overweight patients (4.3% vs. 0.4%, p = 0.012). Regression analysis with reference to postoperative in-house mortality revealed neither increased BMI nor WL as an independent risk factor.
Conclusions
Increased preoperative BMI is associated with a higher WI rate. AL rate after colon resection was significantly higher in patients showing preoperative WL. Preoperative BMI and WL are therefore risk factors for postoperative morbidity in this study. Nevertheless, this has to be further clarified by means of prospective studies.
Trial registration DRKS00025359, 21.05.2021, retrospectively registered.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Colorectal carcinomas are among the most prevalent carcinomas worldwide with an estimated number of new cases of about 1.1 million for colon and about 700,000 for rectal cancer, an estimated number of deaths of about 550,000 for colon, and about 310,000 for rectal cancer [1]. In the treatment of colorectal carcinoma, multimodal concepts depend on various influencing factors such as patient-specific factors (e.g., age, comorbidities), tumor-specific characteristics (e.g., stage, localization), treatment-associated factors (e.g., type of intervention, neoadjuvant therapy), and postoperative complications [2]. In colorectal surgery, risk factors of postoperative complications such as positive tobacco and alcohol anamnesis, age > 65 years, and existing comorbidities with an American Society of Anesthesiologists (ASA) score > III have already been determined [3, 4]. These are associated with higher treatment costs due to longer hospital stays, poorer functional and oncological outcomes, and increased mortality [3,4,5,6,7,8].
However, the influence of BMI is the subject of an ongoing controversial discussion [6, 9]. Also, the influence of unintentional WL in colorectal surgery on the postoperative complication rate is largely unexplored.
The objective of this retrospective cohort study is to determine the influence of preoperative BMI and WL on postoperative surgery-associated complications, specifically AL, WI, and in-hospital mortality (IHM).
Methods
Study design and setting
The study is a retrospective single-centered cohort study. All patients who underwent resection of colorectal carcinomas between the years 2004 and 2014 at the Department of General, Visceral and Transplant Surgery of Tübingen University Hospital were analyzed. Patients were identified by the hospital data system (I.S.H.*med; Siemens Medical Solutions GSD GmbH, Berlin; SAP SE; Walldorf, Germany) using DRG and OPS codes. A 1-year follow-up by means of standardized clinical data collection was added to the data analysis.
All oncological carcinoma resections including lymphadenectomy were performed in a standardized manner according to current evidence-based guidelines for colorectal cancer [10]. Carcinomas were evaluated as colon carcinomas up to the sigmoid colon. Carcinomas of the rectosigmoidal junction were classified as rectal carcinomas.
Multimodal therapies were administered in accordance with the recommendations of the interdisciplinary Center for Gastrointestinal Oncology of Tübingen University Hospital (Comprehensive Cancer Center).
BMI as measurement defines the quantitative variable “weight” statistically by relating body mass [kg] to height [m]. It is broken down by the World Health Organization into underweight (BMI < 18.5 kg/m2), normal weight (BMI 18.5–25 kg/m2), overweight (BMI 25–30 kg/m2), and obesity (BMI > 30 kg/m2) [11]. Unintentional WL as a dynamic variable can be seen as malnutrition. To rule out physiological weight fluctuations, in this study, preoperative WL was considered only if a documented, unintentional WL of at least 3 kg occurred in the last 6 months to achieve a weight loss of 5% according to the statistical average weight [12]. However, there is no uniform definition of preoperative WL in other studies [13,14,15].
Postoperative complications were classified in severity grades I–VI based on the Accordion Severity Classification [16], and comorbidities were graded with the Charlson Comorbidity Index [17]. Patients’ physical condition was classified according to the ASA score.
The main outcome measures were WI, AL, and IHM rates. WI were rated as a local inflammation with microbiological detection of pathogens [18] and AL with clinical and radiographic proof according to the literature [7, 9].
Categorization of quantitative variables into scores was performed for standardized data collection and better comparability. A confounding bias cannot be excluded from the study conducted. An adjustment of the effect was attempted to compensate by regression models.
Statistical analysis
Statistical analysis was performed with SPSS® (IBM®, Armonk, NY, USA) using an Access database (Microsoft, Redmond, WA, USA).
The level of significance was determined with Fisher’s exact test (n > 60)/the Pearson chi2-test (n < 60) or the Mann–Whitney rank-sum test. To illustrate the relationship between metric and nominal values, a receiver operating characteristic (ROC) curve was compiled, and the Youden index was determined, if necessary. Logistic regression analysis was performed to determine the risk of occurrence of an event based on various factors. p values listed are two-sided, and p values < 0.05 are deemed statistically significant.
Ethics
Ethics approval for the conduct of the study in compliance with the protection of the rights and welfare of human subjects participating in medical research (Ethics Review Board of the University of Tübingen, Germany; 273/2010BO2 and 379/2014BO2) was obtained and complies with the criteria of the STROBE checklist for cohort studies in surgery [19]. The study is registered in the German Clinical Trials Register (DRKS00025359). Informed consent has been obtained from the study participants.
Cohort
A total of 1241 patients were recruited: 697 (56%) patients with colon and 544 (44%) with rectal carcinomas. Descriptive data analysis with patient population and differentiation of tumor entity distinguished by weight for colon and rectal carcinoma is presented in Tables 1, 2, 3, and 4.
Results
A total of 1241 patients underwent colorectal surgery in an 11-year period. Descriptive data analysis of the 697 (56.2%) treated colon carcinomas and the 544 (43.8%) rectal carcinomas referred to as preoperative BMI is described in Tables 1 and 3. Eleven patients with colon cancer and 3 patients with rectal cancer could not be categorized into a BMI group. However, weight loss was reported in these patients. In the case of colon carcinoma, preoperative WL was observed in 130 (19%) of the 697 patients and in the case of rectal carcinoma in 79 (15%) of the 544 patients (Tables 2 and 4).
Rate of wound infection
As shown in Table 1, a total of 54 (7.9%) patients developed WI following colon resection. The rate of WI increased with higher BMI (normal weight: 6.8%; overweight: 9%; obesity: 9.6%). However, BMI was not seen to have a significant impact on the WI rate (p = 0.11) or WI severity.
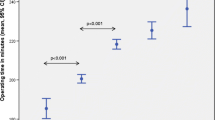
Of the patients who underwent rectal resection, 64 (11.8%) developed WI (Table 3). A significant difference in WI rate was observed between normal weight and obese patients (p = 0.02). The ROC curve illustrates the influence of preoperative BMI on postoperative WI rate. A threshold level of 28.8 kg/m2 was detected. In abundance distribution, no significant difference was observed in the degree of severity relative to BMI.
Tables 2 and 4 show the influence of preoperative WL on the WI rate. After colon (p = 0.36) and rectal resection (p = 0.85), no significant difference in WI rate between patients with and without preoperative WL could be demonstrated as well as in the severity of WI.
Rate of anastomotic leakage
As shown in Table 1, AL was diagnosed in 22 (3.2%) of the 686 patients following colon resection. The ROC curve showed no significant influence of preoperative BMI on the AL rate (p = 0.52), nor was a significant difference demonstrated with regard to the degree of severity.
In the 541 patients with rectal resection with primary anastomosis, AL was detected in 37 (8.7%) patients. A significant difference in the occurrence of AL among the different BMI groups could not be proven (p = 0.81). There was also no significant difference in the distribution of the severity of AL as shown in Table 3.
A significant difference in AL rate following colon resection between patients with and without preoperative WL was demonstrated (p = 0.046). There were no significant differences in the severity of AL (Table 2).
After rectal resection, an AL rate of 8.7% (37 out of 429) was observed with no significant difference between patients with and without preoperative WL (p = 0.81) nor was a significant difference in the severity of AL observed (Table 4). Moreover, no significant difference (p = 0.2) was seen between patients with an ileostomy (n = 14; 6.5%) and without an ileostomy (n = 23; 10.8%).
Rate of in-hospital mortality
IHM following colon resection referred to preoperative BMI was 4.1% (n = 28), caused by multi-organ failure as a result of sepsis in 39.3% (n = 11) and cardiovascular or cardiopulmonary events in 60.7% (n = 17). There was a significant difference in IHM from multi-organ failure as a result of sepsis between overweight and obese patients (p = 0.01), as shown in Table 1. No further significant differences were detected between the other BMI groups.
IHM following rectal resection referred to preoperative BMI was 0.9% (n = 5), caused by multi-organ failure as a result of sepsis in 40% (n = 2) and cardiovascular or cardiopulmonary events in 60% (n = 3) (Table 3). Mortality affected only normal weight and obese patients.
IHM following colon and rectal resection referred to as preoperative weight loss showed no significant differences between the two groups with regard to multi-organ failure as a result of sepsis or cardiovascular or cardiopulmonary events.
Regression analysis
Regression analysis as shown in Table 5 demonstrated that Charlson Comorbidity Index, ASA score, UICC stage, age, and emergency surgery are factors exerting a significant influence on IHM. With an increasing ASA score, the risk of IHM rises significantly (p = 0.049). An increase of one score point doubles the risk of IHM (OR 2.008; 95% CI: 1.003–4.021). With an increase in Charlson Comorbidity Index, the risk of IHM rises significantly, namely by 34% per score point (OR 1.340; 95% CI: 1.157–1.553; p = 0.001). Patients with UICC IV also had a significantly increased risk of IHM as compared to stages 0–I (OR 3.228; 95% CI: 1.014–10.283; p = 0.047). Age (OR 1.072; 95% CI: 1.020–1.126; p = 0.006) and emergency surgery (OR 0.126; 95% CI: 0.047–0.334; p = 0.001) were seen to have an increased risk for IHM. Regression analyses of the incidence of postoperative WI and AL were not reliable due to the small numbers of cases.
Discussion
In our retrospective study of 1241 patients who underwent colorectal cancer surgery in an 11-year period median age [20], sex ratio [21], UICC stages [22], surgical techniques [23], rate of emergency surgery [24], and preoperative BMI [25] were similar compared to other studies. Normal weight patients constituted the largest group in this study population. Compared to the weight distribution of the average population underweight, overweight and obese patients were disproportionately represented [12]. Even though BMI is uncomplicated to determine clinically and well compared to other studies, it is critically discussed in the literature due to its nonuniform use. For example, BMI ≥ 25 kg/m2 was rated overweight or obese without considering other factors such as muscle mass [26]. Other parameters, such as waist-to-size ratio [15], waist circumference, waist-to-hip ratio, or visceral fat, are described as being more sensitive [27]. Nevertheless, BMI is the value most frequently used in publications so that it was also chosen as a reference in the present study.
Of the patients in our study, 17% (n = 209) presented with preoperative WL, which is comparable to other studies with 7–28% [28, 29]. Unintentional WL as a dynamic value is dealt with variously by different studies and can be seen as malnutrition. The pathophysiology of unintentional WL is poorly understood. Malignant diseases are cited as the most frequent cause of unintentional WL prior to nonmalignant gastrointestinal diseases, psychiatric conditions, and unknown causes [13, 30, 31].
Our study demonstrated a significant influence of neoadjuvant therapy for colon carcinoma on preoperative WL (p = 0.004) (Table 2). However, the small number of this subgroup should be taken into account (26/697; 3.7%). In rectal carcinoma, 48% of the patients received neoadjuvant therapy, of which 53.2% presented with preoperative WL. Here, neoadjuvant therapy had no significant influence on preoperative WL (p = 0.33) (Table 4).
The WI rate following colon and rectal resection increased with rising BMI. After colon resection, the WI rate increased significantly in patients with BMI ≥ 30 kg/m2 as compared to patients with BMI ≤ 30 kg/m2. Underweight patients did not develop WI following colon resection in either this study or other studies [18].
After rectal resection, obese patients developed significantly more WI than normal weight patients did. This is comparable to other study results [32, 33]. The threshold value for developing WI after rectal resection was seen to be 28.8 kg/m2. There are no comparable threshold values available.
Patients in our study with or without preoperative WL showed no significant difference in the WI rate following a colon or rectal resection. An increased WI rate caused by WL from malnutrition proved to be an independent risk factor [34]. Furthermore, the possibility of lowering the WI rate for WL by using immunonutrition has already been described [35]. However, another study performed with preoperative immunonutrition therapy showed no change in the postoperative complication rate when bloodwork improved [36]. In contrast to our study, Tang et al. showed an increased WI rate after preoperative WL [37]. Evidence on the influence of preoperative WL on the WI rate following colon and rectal resection is therefore inconsistent.
With regard to AL following a colon or rectal resection, no significant differences between the different BMI groups were found in our study. The highest rate of AL was seen in the group of obese patients but without significant difference. Nevertheless, the evidence is inconsistent. Gessler et al. did not report any significant difference in a retrospective evaluation of 600 patients [7]. In retrospective national analyses, Midura et al. demonstrated no significant difference in the AL rate between obese (BMI > 30 kg/m2) and nonobese patients in a collective of 13,684 patients [8] as well as Bakker et al. in a collective of 15,667 patients [38]. In contrast, Qiu et al. conducted a meta-analysis of ten studies with a total of 3660 obese and 10,829 nonobese patients and demonstrated a significantly increased risk of AL in patients who already had a BMI > 25 kg/m2 (8% versus 2.2%; p < 0.001) [39]. This was also evident in Amri et al. [18]. Geiger et al. pointed out a significant difference in the AL rate following colon resection between nonobese (BMI < 30 kg/m2) and obese patients (BMI > 30 kg/m2). That study did not discriminate between colon and rectal carcinoma [40]. The AL rate following rectal resection ranged from 7.6 to 9.5% for the various BMI groups. Benoist et al. as well as Qu et al. in a meta-analysis of 1619 prospectively and 2967 retrospectively evaluated patients identified obesity as a risk factor for AL following rectal resection [6, 41]. As possible reasons for difficult operative conditions, technical difficulties due to visceral obesity and unclear anatomical conditions were discussed [6, 33, 42]. Following colon resection, there was a significant increase in the AL rate in patients with preoperative WL. This was also demonstrated by Rencuzogullari et al. in patients > 65 years after elective resections [43]. After rectal resection, a more likely occurrence of AL was demonstrated in patients with preoperative WL than in those without, however with no significant difference. In contrast, Kang et al. showed a significantly increased AL rate following rectal resection in patients with preoperative WL [44]. Midura et al. also demonstrated a significantly higher rate of AL in patients compared to patients without WL [8]. Like the influence of BMI on the AL rate, the influence of preoperative WL also needs to be further discussed in different data situations.
Following colon resection, obese patients died significantly more frequently than did overweight patients due to septic multiorgan failure. This was also demonstrated by Amri et al., who, however, did not discriminate between colon and rectal carcinoma [18]. Hu et al. pointed out that 5.9% underweight patients had a significantly higher 30-day mortality rate than did non-underweight patients [45].
Following rectal resection, no significant difference in mortality rate was demonstrated between the different BMI groups. Here, Hu et al. demonstrated also a higher mortality rate after rectal resection in underweight patients [45]. In our study, however, no underweight patient died after rectal resection. Bakker et al. did not demonstrate a correlation between increased BMI and AL or associated higher mortality [38].
Following colon resection, significantly more patients with preoperative WL died than did patients without preoperative WL. After rectal resection, no patient with preoperative WL died. The mortality rate in patients without preoperative WL was also very low, namely 0.9%. Midura et al. were able to demonstrate a significant correlation between preoperative WL and AL. They also demonstrated a correlation between AL and the 30-day mortality rate, which was significantly increased. Thus, it can be concluded that preoperative WL influences the 30-day mortality rate [8]. To further determine the influence of BMI and preoperative WL on the 30-day mortality rate, further studies are needed to clarify the inconsistent data.
Significant differences in IHM following colon and rectal resection between the two groups were not demonstrated in our study. This was supported by regression analysis that revealed Charlson Comorbidity Index, ASA score, UICC stage, age, and emergency resection to be significant influencing factors for IHM. Neither preoperative BMI, WL, nor the occurrence of WI or AL showed a significant influence. In other studies, a higher mortality rate following colorectal cancer surgery was pointed out in patients with comorbidities, higher UICC stages, age > 65 years, and obesity [17, 24, 45]. Regression analysis with reference to postoperative IHM revealed neither increased BMI nor WL as an independent risk factor.
Limitations of the present study include the retrospective study design and the difficult comparability with the data of other studies as a consequence of different BMI and WL definitions. Also, selection and information bias have to be pointed out in the context of retrospective data evaluation. Postoperative complications after colorectal cancer surgery are multifactorial so that a confounding bias of the individual complications cannot be excluded.
Nevertheless, it must be pointed out that despite retrospective data collection, the complete data sets of an 11-year period could be evaluated. A strict distinction between entities of colon and rectum was performed. This resulted in high case numbers of the two collectives in our study. Both preoperative BMI and WL represent a risk factor of increased morbidity in colorectal cancer surgery in our study.
Conclusions
Obese patients were seen to have a significantly higher WI rate than normal weight patients following rectal resection. There was also a significant increase in the AL rate following colon resection in patients with preoperative WL. IHM was significantly increased in obese patients as compared to overweight patients following colon resection. Nevertheless, in multivariable regression analysis, neither preoperative BMI nor WL was an independent risk factor for increased IHM.
While the reasons for WI, AL, and IHM are multifactorial, they are not independent risk factors for the occurrence of these surgery-associated complications. With a simple evaluation of these factors, they could possibly be components of preoperative risk stratification. This should be further investigated in prospective randomized controlled trials. The studies available to date are inhomogeneous and controversial.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request. Further study data can also be accessed in the German Clinical Trials Register (DRKS00025359).
References
Ferlay J, Colombet M, Soerjomataram I, Mathers C, Parkin DM, Pineros M, Znaor A, Bray F (2019) Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int J Cancer 144:1941–1953. https://doi.org/10.1002/ijc.31937
Chadi SA, Fingerhut A, Berho M, DeMeester SR, Fleshman JW, Hyman NH, Margolin DA, Martz JE, McLemore EC, Molena D, Newman MI, Rafferty JF, Safar B, Senagore AJ, Zmora O, Wexner SD (2016) Emerging trends in the etiology, prevention, and treatment of gastrointestinal anastomotic leakage. J Gastrointest Surg 20:2035–2051. https://doi.org/10.1007/s11605-016-3255-3
McDermott FD, Heeney A, Kelly ME, Steele RJ, Carlson GL, Winter DC (2015) Systematic review of preoperative, intraoperative and postoperative risk factors for colorectal anastomotic leaks. Br J Surg 102:462–479. https://doi.org/10.1002/bjs.9697
Pommergaard HC, Gessler B, Burcharth J, Angenete E, Haglind E, Rosenberg J (2014) Preoperative risk factors for anastomotic leakage after resection for colorectal cancer: a systematic review and meta-analysis. Colorectal Dis 16:662–671. https://doi.org/10.1111/codi.12618
Artinyan A, Orcutt ST, Anaya DA, Richardson P, Chen GJ, Berger DH (2015) Infectious postoperative complications decrease long-term survival in patients undergoing curative surgery for colorectal cancer: a study of 12,075 patients. Ann Surg 261:497–505. https://doi.org/10.1097/SLA.0000000000000854
Qu H, Liu Y, Bi DS (2015) Clinical risk factors for anastomotic leakage after laparoscopic anterior resection for rectal cancer: a systematic review and meta-analysis. Surg Endosc 29:3608–3617. https://doi.org/10.1007/s00464-015-4117-x
Gessler B, Eriksson O, Angenete E (2017) Diagnosis, treatment, and consequences of anastomotic leakage in colorectal surgery. Int J Colorectal Dis 32:549–556. https://doi.org/10.1007/s00384-016-2744-x
Midura EF, Hanseman D, Davis BR, Atkinson SJ, Abbott DE, Shah SA, Paquette IM (2015) Risk factors and consequences of anastomotic leak after colectomy: a national analysis. Dis Colon Rectum 58:333–338. https://doi.org/10.1097/DCR.0000000000000249
Almasaudi AS, McSorley ST, Edwards CA, McMillan DC (2018) The relationship between body mass index and short term postoperative outcomes in patients undergoing potentially curative surgery for colorectal cancer: a systematic review and meta-analysis. Crit Rev Oncol Hematol 121:68–73. https://doi.org/10.1016/j.critrevonc.2017.12.004
Oncology guideline program (German Cancer Society GCA, AWMF); Working Group of the Scientific Medical Societies S3 guideline colorectal cancer, long version 2019 30.08.2020; 2.1. http://www.leitlinienprogramm-onkologie.de/leitlinien/kolorektales-karzinom/
Group WW (1986) Use and interpretation of anthropometric indicators of nutritional status. Bull World Health Organ 64:929–941
Office GFS (2018) Shares of the German adult population by body measurements (BMI) and age groups in 2017 https://www.destatis.de: German Federal Statistical Office; [24.07.2020]
Gaddey HL, Holder K (2014) Unintentional weight loss in older adults. Am Fam Physician 89:718–722
Metalidis C, Knockaert DC, Bobbaers H, Vanderschueren S (2008) Involuntary weight loss. Does a negative baseline evaluation provide adequate reassurance? Eur J Intern Med 19:345–349. https://doi.org/10.1016/j.ejim.2007.09.019
Ashwell M, Hsieh SD (2005) Six reasons why the waist-to-height ratio is a rapid and effective global indicator for health risks of obesity and how its use could simplify the international public health message on obesity. Int J Food Sci Nutr 56:303–307. https://doi.org/10.1080/09637480500195066
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213. https://doi.org/10.1097/01.sla.0000133083.54934.ae
Whitmore RG, Stephen JH, Vernick C, Campbell PG, Yadla S, Ghobrial GM, Maltenfort MG, Ratliff JK (2014) ASA grade and Charlson Comorbidity Index of spinal surgery patients: correlation with complications and societal costs. Spine J 14:31–38. https://doi.org/10.1016/j.spinee.2013.03.011
Amri R, Bordeianou LG, Sylla P, Berger DL (2014) Obesity, outcomes and quality of care: body mass index increases the risk of wound-related complications in colon cancer surgery. Am J Surg 207:17–23. https://doi.org/10.1016/j.amjsurg.2013.05.016
Vandenbroucke JP, von Elm E, Altman DG, Gotzsche PC, Mulrow CD, Pocock SJ, Poole C, Schlesselman JJ, Egger M, Initiative S (2014) Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): explanation and elaboration. Int J Surg 12:1500–1524. https://doi.org/10.1016/j.ijsu.2014.07.014
Smith RK, Broach RB, Hedrick TL, Mahmoud NN, Paulson EC (2014) Impact of BMI on postoperative outcomes in patients undergoing proctectomy for rectal cancer: a national surgical quality improvement program analysis. Dis Colon Rectum 57:687–693. https://doi.org/10.1097/DCR.0000000000000097
Damhuis RA, Wereldsma JC, Wiggers T (1996) The influence of age on resection rates and postoperative mortality in 6457 patients with colorectal cancer. Int J Colorectal Dis 11:45–48
Robert-Koch-Institute (2006) Cancer data, cancer in Germany, colorectal cancer https://www.krebsdaten.de/Krebs/DE/Content/Krebsarten/Darmkrebs/darmkrebs_node.html;jsessionid=C1766F47106B4E69DFC08089780632B2.1_cid363: Robert-Koch-Institute; Center for cancer registry datam, [30.07.2020]
Thomassen I, van Gestel YR, Lemmens VE, de Hingh IH (2013) Incidence, prognosis, and treatment options for patients with synchronous peritoneal carcinomatosis and liver metastases from colorectal origin. Dis Colon Rectum 56:1373–1380. https://doi.org/10.1097/DCR.0b013e3182a62d9d
Kirchhoff P, Clavien PA, Hahnloser D (2010) Complications in colorectal surgery: risk factors and preventive strategies. Patient Saf Surg 4:5. https://doi.org/10.1186/1754-9493-4-5
de Vries S, Jeffe DB, Davidson NO, Deshpande AD, Schootman M (2014) Postoperative 30-day mortality in patients undergoing surgery for colorectal cancer: development of a prognostic model using administrative claims data. Cancer Causes Control 25:1503–1512. https://doi.org/10.1007/s10552-014-0451-x
Prentice AM, Jebb SA (2001) Beyond body mass index. Obes Rev 2:141–147. https://doi.org/10.1046/j.1467-789x.2001.00031.x
Cakir H, Heus C, Verduin WM, Lak A, Doodeman HJ, Bemelman WA, Houdijk AP (2015) Visceral obesity, body mass index and risk of complications after colon cancer resection: a retrospective cohort study. Surgery 157:909–915. https://doi.org/10.1016/j.surg.2014.12.012
Smith D, Ballal M, Hodder R, Soin G, Selvachandran SN, Cade D (2006) Symptomatic presentation of early colorectal cancer. Ann R Coll Surg Engl 88:185–190. https://doi.org/10.1308/003588406X94904
Vokurka J, Spurny V, Kabela M (2005) Combination therapy of colorectal carcinoma. Bratisl Lek Listy 106:416–420
Atalayer D, Astbury NM (2013) Anorexia of aging and gut hormones. Aging Dis 4:264–275. https://doi.org/10.14336/AD.2013.0400264
Ruscin JM, Page RL 2nd, Yeager BF, Wallace JI (2005) Tumor necrosis factor-alpha and involuntary weight loss in elderly, community-dwelling adults. Pharmacotherapy 25:313–319. https://doi.org/10.1592/phco.25.3.313.61607
Aytac E, Lavery IC, Kalady MF, Kiran RP (2013) Impact of obesity on operation performed, complications, and long-term outcomes in terms of restoration of intestinal continuity for patients with mid and low rectal cancer. Dis Colon Rectum 56:689–697. https://doi.org/10.1097/DCR.0b013e3182880ffa
Denost Q, Quintane L, Buscail E, Martenot M, Laurent C, Rullier E (2013) Short- and long-term impact of body mass index on laparoscopic rectal cancer surgery. Colorectal Dis 15:463–469. https://doi.org/10.1111/codi.12026
Putwatana P, Reodecha P, Sirapo-ngam Y, Lertsithichai P, Sumboonnanonda K (2005) Nutrition screening tools and the prediction of postoperative infectious and wound complications: comparison of methods in presence of risk adjustment. Nutrition 21:691–697. https://doi.org/10.1016/j.nut.2004.10.015
Gianotti L, Braga M, Nespoli L, Radaelli G, Beneduce A, Di Carlo V (2002) A randomized controlled trial of preoperative oral supplementation with a specialized diet in patients with gastrointestinal cancer. Gastroenterology 122:1763–1770. https://doi.org/10.1053/gast.2002.33587
Sorensen LS, Thorlacius-Ussing O, Rasmussen HH, Lundbye-Christensen S, Calder PC, Lindorff-Larsen K, Schmidt EB (2014) Effects of perioperative supplementation with omega-3 fatty acids on leukotriene B(4) and leukotriene B(5) production by stimulated neutrophils in patients with colorectal cancer: a randomized, placebo-controlled intervention trial. Nutrients 6:4043–4057. https://doi.org/10.3390/nu6104043
Tang R, Chen HH, Wang YL, Changchien CR, Chen JS, Hsu KC, Chiang JM, Wang JY (2001) Risk factors for surgical site infection after elective resection of the colon and rectum: a single-center prospective study of 2,809 consecutive patients. Ann Surg 234:181–189. https://doi.org/10.1097/00000658-200108000-00007
Bakker IS, Grossmann I, Henneman D, Havenga K, Wiggers T (2014) Risk factors for anastomotic leakage and leak-related mortality after colonic cancer surgery in a nationwide audit. Br J Surg 101:424–32. https://doi.org/10.1002/bjs.9395. discussion 32
Qiu Y, Liu Q, Chen G, Wang W, Peng K, Xiao W, Yang H (2016) Outcome of rectal cancer surgery in obese and nonobese patients: a meta-analysis. World J Surg Oncol 14:23. https://doi.org/10.1186/s12957-016-0775-y
Geiger TM, Muldoon R (2011) Complications following colon rectal surgery in the obese patient. Clin Colon Rectal Surg 24:274–282. https://doi.org/10.1055/s-0031-1295692
Benoist S, Panis Y, Alves A, Valleur P (2000) Impact of obesity on surgical outcomes after colorectal resection. Am J Surg 179:275–281. https://doi.org/10.1016/s0002-9610(00)00337-8
Clark W, Siegel EM, Chen YA, Zhao X, Parsons CM, Hernandez JM, Weber J, Thareja S, Choi J, Shibata D (2013) Quantitative measures of visceral adiposity and body mass index in predicting rectal cancer outcomes after neoadjuvant chemoradiation. J Am Coll Surg 216:1070–1081. https://doi.org/10.1016/j.jamcollsurg.2013.01.007
Rencuzogullari A, Benlice C, Valente M, Abbas MA, Remzi FH, Gorgun E (2017) Predictors of anastomotic leak in elderly patients after colectomy: nomogram-based assessment from the American College of Surgeons National Surgical Quality Program Procedure-Targeted Cohort. Dis Colon Rectum 60:527–536. https://doi.org/10.1097/DCR.0000000000000789
Kang CY, Halabi WJ, Chaudhry OO, Nguyen V, Pigazzi A, Carmichael JC, Mills S, Stamos MJ (2013) Risk factors for anastomotic leakage after anterior resection for rectal cancer. JAMA Surg 148:65–71. https://doi.org/10.1001/2013.jamasurg.2
Hu WH, Cajas-Monson LC, Eisenstein S, Parry L, Cosman B, Ramamoorthy S (2015) Preoperative malnutrition assessments as predictors of postoperative mortality and morbidity in colorectal cancer: an analysis of ACS-NSQIP. Nutr J 14:91. https://doi.org/10.1186/s12937-015-0081-5
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
Each of the listed authors meets the requirements for listing according to the “Uniform Requirements for Manuscripts Submitted to Biomedical Journals” statement of the International Committee of Medical Journal Editors. All authors read and approved the final manuscript. Also, each author has agreed to be personally accountable for the author’s own contributions and to ensure that questions related to the accuracy or integrity of any part of the work, even ones in which the author was not personally involved, are appropriately investigated, resolved, and the resolution documented in the literature. Steffen Axt: substantial contributions to the conception, interpretation of data, and drafted the work. Peter Wilhelm: the acquisition, analysis, interpretation of data, and substantively revised the work. Ricarda Spahlinger: the acquisition, analysis, interpretation of data, and substantively revised the work. Jens Rolinger: the acquisition, analysis, interpretation of data, and substantively revised the work. Jonas Johannink: the acquisition, analysis, interpretation of data, and substantively revised the work. Lena Axt: validation, roles/writing—original draft, and writing—review and editing. Andreas Kirschniak: substantial contributions to the conception and interpretation of data. Claudius Falch: substantial contributions to the conception, interpretation of data, and drafted the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Ethics Review Board of the University of Tübingen, Germany; 273/2010BO2 and 379/2014BO2.
Consent to participate
Informed consent has been obtained from the study participants.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Axt, S., Wilhelm, P., Spahlinger, R. et al. Impact of preoperative body mass index and weight loss on morbidity and mortality following colorectal cancer—a retrospective cohort study. Int J Colorectal Dis 37, 1983–1995 (2022). https://doi.org/10.1007/s00384-022-04228-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-022-04228-1