Abstract
Purpose
The impact of anastomotic leaks (AL) on oncological outcomes after low anterior resection for mid-low rectal cancer is still debated. The aim of this study was to evaluate overall survival (OS), disease-free survival (DFS), and local and distant recurrence in patients with AL following low anterior resection.
Methods
This is an extension of a multicentre RCT (NCT01110798). Kaplan–Meier method and the log-rank test were used to estimate and compare the 3-, 5-, and 10-year OS and DFS, and local and distant recurrence in patients with and without AL. Predictors of OS and DFS were evaluated using the Cox regression analysis as secondary aim.
Results
Follow-up was available for 311 patients. Of them, 252 (81.0%) underwent neoadjuvant chemoradiotherapy and 138 (44.3%) adjuvant therapy. AL occurred in 63 (20.3%) patients. At a mean follow-up of 69.5 ± 31.9 months, 23 (7.4%) patients experienced local recurrence and 49 (15.8%) distant recurrence. The 3-, 5-, and 10-year OS and DFS were 89.2%, 85.3%, and 70.2%; and 80.7%, 75.1%, and 63.5% in patients with AL, and 88.9%, 79.8% and 72.3%; and 83.7, 74.2 and 62.8%, respectively in patients without (p = 0.89 and p = 0.84, respectively). At multivariable analysis, AL was not an independent predictor of OS (HR 0.65, 95%CI 0.34–1.28) and DFS (HR 0.70, 95%CI 0.39–1.25), whereas positive circumferential resection margins and pathological stage impaired both.
Conclusions
In the context of modern multimodal rectal cancer treatment, AL does not affect long-term OS, DFS, and local and distant recurrence in patients with mid-low rectal cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rectal cancer represents a major cause of morbidity and mortality worldwide, accounting for about 736,000 new estimated cases and 340,000 estimated deaths in 2020 [1]. The standard of care for locally advanced mid-low rectal cancer is surgical resection with total mesorectal excision (TME) associated with neoadjuvant chemoradiotherapy (nCRT) [2].
Anastomosis-related complications represent the main source of morbidity after TME. Anastomotic leak (AL) occurs in up to 20% of low anterior resection (LAR) [3], and may require interventional treatment and, eventually, the need for a temporary or permanent stoma [4]. Several studies investigated risk factors for AL, and no differences were found using different reconstructive techniques [5]. In pre-multimodal treatment era, sex, obesity, and distance of the anastomosis from the anal verge were recognized as independent risk factors for AL after rectal resection [6]. Recently, many retrospective studies reported an increased rate of AL following nCRT [7,8,9]; nevertheless, results from randomised controlled trials (RCT) showed no difference in AL rate between nCRT and non-nCRT patients [5, 10, 11]. Regardless from these uncertainties, it is widely accepted that AL results in an increased overall postoperative morbidity and mortality, and prolonged in-hospital length-of-stay.
The impact of AL on oncological long-term outcomes have been previously investigated, with various results. The presence of a defect of the intestinal wall at anastomosis may promote the spilling of neoplastic cells and increase the risk of local recurrences. Furthermore, local inflammation caused by AL was found to promote the upregulation of receptors associated with adhesion of tumour cells [12, 13]; finally, the occurrence of an AL delays the beginning of adjuvant therapy, preventing an optimal local and distant control of the disease, resulting in a decreased disease-free survival (DFS) and overall survival (OS). In the meta-analyses by Mirnezami et al. and by Ha et al., AL following colorectal resection was reported to affect local recurrence and OS [14, 15]. More recently, other meta-analyses investigated specifically survival in patients with AL after rectal resection, reporting that AL had an adverse impact on survival and local recurrence [16,17,18]. Adverse effects of AL were also reported in a long-term analysis of series including patients with previous nCRT [19]. Other retrospective and observational studies, however, reported no impact of AL on long-term oncological outcomes [20,21,22,23].
Considering a potential adverse effect on recurrence, OS and DFS, the aim of the current study was to evaluate the impact of AL following LAR on OS, DFS, and local and distant recurrence, in patients enrolled in a multicentre RCT [5].
Methods
Study design
The present study represents an extended follow-up secondary analysis of a prospective multicentre RCT (NCT01110798) that enrolled patients affected by mid-low rectal cancer undergoing curative-intent surgery at 16 Italian centres between October 2009 and February 2016 [5, 24]. Long-term survival analyses were originally designed as secondary outcome of the trial. Inclusion criteria of the trial were the following: age > 18 years, biopsy-proven adenocarcinoma of the rectum up to 11 cm from the anal verge, resectable disease with LAR and stapled anastomosis, and curative-intent resection (R0-R1). Exclusion criteria were non-curative resection (R2), metastatic disease, previous history of colonic resection, and handsewn coloanal anastomosis.
The study was first approved by the Ethics Committee of the coordinating centre and subsequently approved by the local committee of every participating centre. Patients provided written informed consent to participate in this study. The current analysis was reported following the The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement [25].
Endpoints and outcome measures
The primary endpoint on this study was the impact of AL on long-term oncologic outcomes. The outcome measures were OS, DFS, and incidence of local and distant recurrence. The secondary endpoints included the rate and pattern of recurrence, and factors associated with oncologic outcomes.
Data of interest and treatment details
The following data were recorded for each patient: gender, age, American Society of Anaesthesiologists (ASA) score, Eastern Cooperative Oncology Group (ECOG) performance status scale, Body Mass Index (BMI), distance of the tumour from the anal verge, baseline Carcinoembryonic Antigen (CEA) level, clinical staging, and nCRT regimen. The race/ethnicity distribution of the enrolled patients was not available, since it was not collected in the original study. All the patients underwent a LAR with standard TME [26], and were randomised to receive either a stapled colonic J-Pouch or a straight colorectal anastomosis. The creation of a covering stoma was mandatory. Long-course preoperative CRT (capecitabine-based chemotherapy + 45-–50.4-Gy radiotherapy) or short-course radiotherapy (5 Gy in 5 fractions) was administered as recommended in national guidelines [27]. Adjuvant treatment was offered to patients with pTNM II stage with high-risk features, or pTNM III stage. In patients who underwent adjuvant treatment, stoma reversal was recommended after the completion of the treatment.
Definitions
Major non-anastomotic complications were defined as any complication requiring interventional treatment (i.e. surgical or radiological procedures, Clavien-Dindo grade ≥ 3a [28]). According to the International Study Group of Rectal Cancer, AL was defined as any defect of the anastomosis leading to a communication between intra- and extra-luminal compartments. Leak originating from suture or staple line of the colonic J-Pouch and pelvic abscess in the proximity of the anastomosis were considered as AL [29]. According to the study protocol, the evaluation of anastomotic integrity was mandatory within 30 days of the index surgery and was assessed by endoscopic or radiologic (i.e. soluble contrast medium enema/CT) investigation. The severity of AL was classified according to the definition and grading system proposed by Rahbari et al. [29]. Grade A was defined as AL resulting in no change in patient’s management, grade B as AL requiring therapeutic intervention without re-laparotomy, and grade C as AL requiring re-laparotomy.
For every patient, pathological data were collected, including tumour size and grading, circumferential resection margin (CRM) status, pathologic tumour (pT) and nodal (pN) stage, number of harvested lymph nodes, and radicality of resection (R0-R1). Clinical and pathological TNM staging were reported according to the American Joint Committee on Cancer 8th Edition [30].
Long-term outcomes definition
Patients underwent a standard oncological follow-up, according to national guidelines [27]. Physical examination, routine blood tests, and CEA were performed every 4 months in the first 2 years, and then every 6 months for a total of 5 years. Chest-abdominal CT was performed every 6 months for 5 years. Colonoscopy was recommended 1 year after surgery. For the purpose of this study, every participating centre was contacted to update the oncological outcomes (date of death or last follow-up, and date of local or distant recurrence). Local recurrence was defined as any pelvic endoluminal or extra-luminal recurrence, while recurrences outside the pelvis were defined as distant.
Statistical analysis
Continuous data are expressed as mean value with standard deviation. Significant differences between the two groups were test by the Fisher exact test for categorical variables, and independent sample t test for continuous variables. To estimate the OS and DFS, the Kaplan–Meier method and log-rank test were used to compare the survival of the groups. Each outcome was calculated from the date of surgery to the date of the event (local or distant recurrence, death, or the last follow-up). Multivariable survival analysis for OS and DFS was performed using Cox regression model. Results are reported as Hazard Ratio with 95% confidence intervals (CI). All analyses were carried out with STATA version 13.0 (StataCorp, College Station, TX).
Results
Patients, tumour, and treatment characteristics
Eleven out of 16 centres agreed to participate to this study, resulting in the inclusion of long-term outcomes of 311 out of 379 patients who were initially enrolled in the trial.
The following baseline clinical stage was available in 308 patients: cT1 (n = 8, 2.6%), cT2 (n = 43, 14%), cT3 (n = 249, 81.0%), and cT4 (n = 8, 2.6%). Lymph nodes were found to be clinically positive in 192 (63.0%) patients. A total of 252 (81.0%) patients underwent preoperative treatment, 46 (14.8%) underwent a short-course radiotherapy, 204 (65.6%) a long-course nCRT, and two underwent chemotherapy only. In patients who received nCRT, the most common chemotherapy regimens included capecitabine/5-FU alone (n = 139, 44.7%), or in associations with oxaliplatin (n = 13, 4.2%).
A conventional open LAR was performed in 200 (64.3%) patients, while a minimally invasive approach was performed in 111 (35.7%). Overall, 145 (46.6%) patients underwent a colonic J-pouch, and 166 (53.3%) a straight colorectal anastomosis. The mean distance of the anastomosis from the anal verge was 4.3 ± 1.5 cm. The mean postoperative hospital length-of-stay was 10.7 ± 10.2 days. Postoperative not AL-related complications were found in 94 (30.2%) patients; of these, 54 (17.4%) were minor and 40 (12.9%) were major. Readmission was required in 25 (8.0%) patients, and re-operation in 21 (6.8%).
Anastomotic leak
Patients, tumour, and treatment characteristics of the study group according to the presence or absence of AL are summarised in Table 1. Sixty-three (20.3%) patients were found to have an AL. Twenty (6.4%) patients had grade A, 31 (10.0%) grade B, and 12 (3.9%) grade C AL. Most of the patients with AL (n = 54, 85.7%) had received nCRT. AL was more common in men than in women (24.3% vs 14.3%, p = 0.03). ASA score and BMI were higher in the AL than in non-AL group (p = 0.03 and p = 0.01 respectively). The mean distance of the tumour from the anal verge was 7.3 ± 2.2 cm and was shorter in patients with AL than in those without AL (6.7 ± 2.3 vs 7.5 ± 2.1 cm, p = 0.008). Pathological stage, rate of CRM infiltration, number of lymph nodes harvested, and rate of patients who underwent nCRT and adjuvant chemotherapy did not differ between patients with and without AL (Table 1). The postoperative hospital length-of-stay was longer in patients with AL than in those without AL (median (IQR) 9(7–14) vs 8(7–11) days, p = 0.001).
Long-term outcomes and pattern of recurrence
At a mean follow-up of 69.5 (31.9) months, recurrence occurred in 61 (19.6%) patients. Of these, 12 (3.9%) patients developed local recurrence, 38 (12.2%) distant recurrence, and 11 (3.5%) both local and distant recurrence. The most common sites of recurrence were lungs (n = 33, 10.6%) and liver (n = 22, 7.1%). The mean time from surgery to local and distant recurrence was 26.0 ± 19.3 and 29.5 ± 23.4 months, respectively.
The pathological stage of 63 patients with AL was the following: pT0 (n = 9, 14.3%), pT1 (n = 10, 15.8%), pT2 (n = 20, 31.7%), pT3, (n = 23, 36.5%), and pT4 (n = 1, 1.5%). In the same patients, 46 (73.0%) were found to be pN0. The final pTNM stage was the following: stage 0 (n = 9, 14.3%), stage I (n = 23, 36.5%), stage II (n = 14, 22.2%), and stage III (n = 17, 27.0%). Adjuvant chemotherapy was performed in 23 (36.5%) patients, 16 of whom have had a clinically relevant AL (grades II–III).
Among the patients who experienced recurrence, 10 (16.3%) also experienced an AL (grade A n = 2, grade B n = 4, grade C n = 4). In these patients, the recurrences were local (n = 1), distant (n = 4), and both local and distant (n = 5). The mean time to local and distant recurrence was 25.7 ± 14 and 28.2 ± 19.3 months, respectively.
Among the 20 patients with a grade A AL, 2 (10.0%) patients experience recurrence (distant only, n = 1, local + distant, n = 1), and 3 of them deceased, only 1 for progression disease. Out of 31 patients with a grade B AL, 4 (12.9%) patients experience recurrence (local only, n = 1, distant only, n = 2, and local + distant n = 1), and 8 of them deceased, only 2 for progression disease. Among 12 patients with a grade C AL, 4 (33.3%) patients experienced recurrence (distant only, n = 1, and local + distant n = 3), and 3 of them deceased, 2 for progression disease.
Primary aim: AL and long-term oncologic outcomes
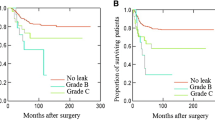
In patients with AL, the estimated cumulative 3-, 5- and 10-year OS were 89.2%, 85.3%, and 70.2%, respectively, whereas the estimated cumulative 3-, 5- and 10-year DFS were 80.7%, 75.1%, and 63.5%, respectively. Comparing these curves with those of patients without AL, no differences were found between the two groups (p = 0.89, and p = 0.84 respectively) (Figs. 1 and 2).
No differences between patients with and without AL were found in terms of local and distant recurrence (p = 0.41, and p = 0.89 respectively) (Figs. 3 and 4).
Secondary aim: predictors of survival
On multivariable analysis (Table 2), preoperative CEA level (HR 1.01. 95%CI 1.00–1.01, p = 0.02), distance from the anal verge (HR 0.89, 95%CI 0.79–1.00, p = 0.05), pT stage (HR 1.43, 95%CI 1.03–1.99, p = 0.033), pN stage (HR 1.64, 95%CI 1.11–2.42, p = 0.014), and a positive CRM (HR 4.44, 95%CI 1.52–12.9, p = 0.006) were found to be independent predictors of OS.
Preoperative CEA level (HR 1.02 95%CI 1.01–1.02, p < 0.001), pT stage (HR 1.33, 95%CI 1.01–1.74, p = 0.045), pN stage (HR 1.76, 95%CI 1.24–2.49, p = 0.002), and a positive CRM (HR 4.96, 95%CI 1.90–12.9, p = 0.001) were found to be independent predictors of DFS.
AL was not an independent predictor of OS (HR 0.65, 95%CI 0.34–1.28, p = 0.2) nor of DFS (HR 0.70, 95%CI 0.39–1.25, p = 0.2).
Discussion
This study aimed to investigate whether AL following LAR for mid-low rectal cancer may have a negative impact on oncological outcomes, focusing on OS and DFS. AL did not impact OS, DFS, and local and distant recurrence, but other factors including tumour-related characteristics and surgical clearance did.
Over the last decades, several studies have looked at a potential correlation between AL and long-term outcomes after rectal cancer surgery, but results are still controversial, because of several confounders and factors that need to be considered in these patients. The findings of the current study are in line with those of Crippa et al. from the Mayo Clinic, who found a local recurrence rate of 4.8% in patients with AL, which was not different compared to patients without AL. Moreover, AL was not found to be an independent predictor of local recurrence [21]. Similar findings were previously found by Smith et al. who reported the Memorial Sloan Kettering Cancer Center experience. Even if a clinical AL was associated with a delay of adjuvant treatment administration, it was not associated with an increased local recurrence rate. Again, the AL was not found to be an independent risk factor for local recurrence [23]. The same considerations apply for OS [21, 23]. In the present study, the survival outcomes in terms of OS and DFS did not differ between patients with and without AL. In a more recent RCT (COLOR II) on 764 patients with rectal cancer randomised to laparoscopic versus open surgery, AL was independently associated with reduced DFS and higher risk of local recurrence, but not with OS and distant recurrence [31].
A significant number of meta-analyses have been published on this topic, using data from retrospective and prospective studies [14,15,16,17,18], and the more recent did found a correlation between AL and survival and local recurrence [16, 17]. However, the significant heterogeneity between the included studies, the wide time frames of treatment, and the inclusion of studies on patients who did not receive neoadjuvant treatments make it difficult to draw definitive conclusions.
Apart from tumour stage and CEA levels, an important factor to consider when it comes to assessing long-term outcomes and disease control after curative LAR for cancer, is whether the patients underwent concomitant treatments. Jang et al. reported on 698 patients with locally advanced mid-low rectal cancer treated with long-course nCRT followed by TME [22], with an AL rate of 6.7%. Most of these AL were clinically severe and required re-operation (grade C = 83%), resulting in a statistically significant delay in adjuvant therapy in patients with AL compared with those without AL. However, survival analysis reported no differences concerning the 5-year OS (90.9% vs 86.2%, p = 0.242) and DFS (93.3% vs 94.9%, p = 0.653). An analysis of the prospective Spanish Rectal Cancer Project Registry on 1153 patients reported a clinically relevant AL rate of 9.4% after LAR, which was not an independent predictor of OS (HR 1.10 95%CI 0.73–1.65; p = 0.648) nor cancer-specific survival (HR 1.23 95%CI 0.75–2.02; p = 0.421) [20]. However, a retrospective propensity-score analysis by Kulu et al. found AL to be an independent risk factor for worse OS and DFS [32]. Of note, only 50% of their patients received nCRT and 25% received adjuvant therapy. The long-term survival analysis of the German Rectal cancer trial found that AL was associated with impaired 10-year OS (51.0% vs 65.2%, p = 0.02) in all treatment group. However, the rate of distant metastases and local recurrence was higher after AL only in those patients who did not receive neoadjuvant or adjuvant CRT, even in stage I cancers [19]. The association with chemotherapy and radiotherapy, either before or after surgery, seems therefore to play a relevant role on the outcomes. Most patients in the current study received concomitant therapy. Overall, 81% and 44% of the patients underwent nCRT and adjuvant therapy respectively, and more than 1/3 of the patients (37.0%) to both neoadjuvant and adjuvant therapy, further advocating a protective effect in term of long-term outcomes irrespective from if AL occurs, but neither nCRT nor adjuvant therapy were protective factors for OS and DFS at regression analysis.
Finally, another relevant aspect that needs attention is the importance of performing adequate, radical surgery during LAR, achieving complete oncological clearance. Patients with R2 resection were not included in the study, and a microscopically positive CRM was the strongest predictor of worse survival, increasing the risk of shorter OS and DFS by more than 4 times (Table 2). The surgeon factor should therefore not be underestimated [33], as a proper oncological resection remains the most important prognosticator of survival.
Study limitations and strengths
This study does have limitations. Some data concerning the adjuvant therapy (e.g. regimen, date of start, potential delay) were not consistently captured. Also, not all centres joined the extended follow-up analyses, reducing the available sample of the patients included. As matter-of-fact, data in only 11 out of 16 centres, resulting in 311 out of 379 patients initially enrolled in the trial, were available. The study sample size of was estimated on the primary outcome of the RCT, and the trial was interrupted at the second ad interim analysis. Of course, by interrupting the RCT and reducing the sample size, the risk of type 2 statistical error on this secondary outcome is increased. However, by considering the size of the population with an AL, setting the power of the study at 0.80, the estimable difference in survival is up to 20% approximately.
However, published studies have several limitations that were removed in the current analysis. The most common shortcomings of the previous reports include the retrospective design, the lack of a shared and validated definition and grading of AL, the inclusion of both colon and rectal cancer resections, a wide inclusion of patients not treated with neoadjuvant or adjuvant treatment, and the lack of data regarding neoadjuvant and adjuvant treatment. On the contrary, the current study included long-term analysis as scheduled secondary outcome of a multicentre RCT. The anastomosis was systematically evaluated after surgery, and events were reported according to a shared and validated definition and grading system [29], and the overall AL rate is high. However, due to the low rate of recurrences in grades A, B, and C, AL is difficult to assess a statistical difference, even if the rate is relatively higher in most severe leakage (grades A, B, and C 10.0, vs 12.9, vs 33.3% respectively). Additional strengths of this study include its multicentric design, the long-term follow-up, the prospective collection of data, and the inclusion of mid and low rectal cancer only. Furthermore, most of the patients (80%) underwent nCRT, and 45% adjuvant treatment in high-risk stage II and stage III disease, making results easily generalizable.
Of note, patients in the current study who experienced an AL were promptly treated at tertiary centres, thereby optimising the chances of long-term success—potentially reducing the long-term impact on survival.
Conclusions
In patients with mid-low rectal cancer who undergo LAR in the context of a multidisciplinary team and multimodal management, AL do not impair OS, DFS, and do not increase local and distant recurrence rates.
Pathological stage, preoperative CEA levels, and CRM status are independent predictors of long-term survival. This suggests the importance of adequate preoperative planning, and the relevance of following the oncological principles of radical surgery. On the other hand, the study allows to reassure patients who experience AL that they should not expect worse long-term outcomes following such a debilitating complication.
Data availability
The dataset used in the current study is available on reasonable request to the authors.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer J Clinici
Ma B, Gao P, Wang H, Xu Q, Song Y, Huang X, Sun J, Zhao J, Luo J, Sun Y, Wang Z (2017) What has preoperative radio(chemo)therapy brought to localized rectal cancer patients in terms of perioperative and long-term outcomes over the past decades? A systematic review and meta-analysis based on 41,121 patients. Int J Cancer 141(5):1052–1065. https://doi.org/10.1002/ijc.30805
Borstlap WAA, Westerduin E, Aukema TS, Bemelman WA, Tanis PJ (2017) Anastomotic leakage and chronic presacral sinus formation after low anterior resection: results from a large cross-sectional study. Ann Surg 266(5):870–877. https://doi.org/10.1097/sla.0000000000002429
Dinnewitzer A, Jäger T, Nawara C, Buchner S, Wolfgang H, Öfner D (2013) Cumulative incidence of permanent stoma after sphincter preserving low anterior resection of mid and low rectal cancer. Dis Colon Rectum 56(10):1134–1142. https://doi.org/10.1097/DCR.0b013e31829ef472
Pucciarelli S, Del Bianco P, Pace U, Bianco F, Restivo A, Maretto I, Selvaggi F, Zorcolo L, De Franciscis S, Asteria C, Urso EDL, Cuicchi D, Pellino G, Morpurgo E, La Torre G, Jovine E, Belluco C, La Torre F, Amato A, Chiappa A, Infantino A, Barina A, Spolverato G, Rega D, Kilmartin D, De Salvo GL, Delrio P (2019) Multicentre randomized clinical trial of colonic J pouch or straight stapled colorectal reconstruction after low anterior resection for rectal cancer. Br J Surg 106(9):1147–1155. https://doi.org/10.1002/bjs.11222
Rullier E, Laurent C, Garrelon JL, Michel P, Saric J, Parneix M (1998) Risk factors for anastomotic leakage after resection of rectal cancer. Br J Surg 85(3):355–358. https://doi.org/10.1046/j.1365-2168.1998.00615.x
Eriksen MT, Wibe A, Norstein J, Haffner J, Wiig JN (2005) Anastomotic leakage following routine mesorectal excision for rectal cancer in a national cohort of patients. Colorectal Dis 7(1):51–57. https://doi.org/10.1111/j.1463-1318.2004.00700.x
Matthiessen P, Hallböök O, Andersson M, Rutegård J, Sjödahl R (2004) Risk factors for anastomotic leakage after anterior resection of the rectum. Colorectal Dis 6(6):462–469. https://doi.org/10.1111/j.1463-1318.2004.00657.x
Park JS, Choi GS, Kim SH, Kim HR, Kim NK, Lee KY, Kang SB, Kim JY, Lee KY, Kim BC, Bae BN, Son GM, Lee SI, Kang H (2013) Multicenter analysis of risk factors for anastomotic leakage after laparoscopic rectal cancer excision: the Korean laparoscopic colorectal surgery study group. Ann Surg 257(4):665–671. https://doi.org/10.1097/SLA.0b013e31827b8ed9
Sebag-Montefiore D, Stephens RJ, Steele R, Monson J, Grieve R, Khanna S, Quirke P, Couture J, de Metz C, Myint AS, Bessell E, Griffiths G, Thompson LC, Parmar M (2009) Preoperative radiotherapy versus selective postoperative chemoradiotherapy in patients with rectal cancer (MRC CR07 and NCIC-CTG C016): a multicentre, randomised trial. Lancet 373(9666):811–820. https://doi.org/10.1016/s0140-6736(09)60484-0
Marijnen CA, Kapiteijn E, van de Velde CJ, Martijn H, Steup WH, Wiggers T, Kranenbarg EK, Leer JW (2002) Acute side effects and complications after short-term preoperative radiotherapy combined with total mesorectal excision in primary rectal cancer: report of a multicenter randomized trial. J Clin Oncol 20(3):817–825. https://doi.org/10.1200/jco.2002.20.3.817
Umpleby HC, Fermor B, Symes MO, Williamson RC (1984) Viability of exfoliated colorectal carcinoma cells. Br J Surg 71(9):659–663. https://doi.org/10.1002/bjs.1800710902
Fermor B, Umpleby HC, Lever JV, Symes MO, Williamson RC (1986) Proliferative and metastatic potential of exfoliated colorectal cancer cells. J Natl Cancer Inst 76(2):347–349
Mirnezami A, Mirnezami R, Chandrakumaran K, Sasapu K, Sagar P, Finan P (2011) Increased local recurrence and reduced survival from colorectal cancer following anastomotic leak: systematic review and meta-analysis. Ann Surg 253(5):890–899. https://doi.org/10.1097/SLA.0b013e3182128929
Ha GW, Kim JH, Lee MR (2017) Oncologic impact of anastomotic leakage following colorectal cancer surgery: a systematic review and meta-analysis. Ann Surg Oncol 24(11):3289–3299. https://doi.org/10.1245/s10434-017-5881-8
Lu ZR, Rajendran N, Lynch AC, Heriot AG, Warrier SK (2016) Anastomotic leaks after restorative resections for rectal cancer compromise cancer outcomes and survival. Dis Colon Rectum 59(3):236–244. https://doi.org/10.1097/dcr.0000000000000554
Yang J, Chen Q, Jindou L, Cheng Y (2020) The influence of anastomotic leakage for rectal cancer oncologic outcome: a systematic review and meta-analysis. J Surg Oncol 121(8):1283–1297. https://doi.org/10.1002/jso.25921
Wang S, Liu J, Wang S, Zhao H, Ge S, Wang W (2017) Adverse effects of anastomotic leakage on local recurrence and survival after curative anterior resection for rectal cancer: a systematic review and meta-analysis. World J Surg 41(1):277–284. https://doi.org/10.1007/s00268-016-3761-1
Sprenger T, Beißbarth T, Sauer R, Tschmelitsch J, Fietkau R, Liersch T, Hohenberger W, Staib L, Gaedcke J, Raab HR, Rödel C, Ghadimi M (2018) Long-term prognostic impact of surgical complications in the German Rectal Cancer Trial CAO/ARO/AIO-94. Br J Surg 105(11):1510–1518. https://doi.org/10.1002/bjs.10877
Espín E, Ciga MA, Pera M, Ortiz H (2015) Oncological outcome following anastomotic leak in rectal surgery. Br J Surg 102(4):416–422. https://doi.org/10.1002/bjs.9748
Crippa J, Duchalais E, Machairas N, Merchea A, Kelley SR, Larson DW (2020) Long-term oncological outcomes following anastomotic leak in rectal cancer surgery. Dis Colon Rectum 63(6):769–777. https://doi.org/10.1097/dcr.0000000000001634
Jang JH, Kim HC, Huh JW, Park YA, Cho YB, Yun SH, Lee WY, Yu JI, Park HC, Park YS, Park JO (2019) Anastomotic leak does not impact oncologic outcomes after preoperative chemoradiotherapy and resection for rectal cancer. Ann Surg 269(4):678–685. https://doi.org/10.1097/sla.0000000000002582
Smith JD, Paty PB, Guillem JG, Temple LK, Weiser MR, Nash GM (2012) Anastomotic leak is not associated with oncologic outcome in patients undergoing low anterior resection for rectal cancer. Ann Surg 256(6):1034–1038. https://doi.org/10.1097/SLA.0b013e318257d2c1
Gavaruzzi T, Pace U, Giandomenico F, Pucciarelli S, Bianco F, Selvaggi F, Restivo A, Asteria CR, Morpurgo E, Cuicchi D, Jovine E, Coletta D, La Torre G, Amato A, Chiappa A, Marchegiani F, Rega D, De Franciscis S, Pellino G, Zorcolo L, Lotto L, Boccia L, Spolverato G, De Salvo GL, Delrio P, Del Bianco P (2020) Colonic J-pouch or straight colorectal reconstruction after low anterior resection for rectal cancer: impact on quality of life and bowel function: a multicenter prospective randomized study. Dis Colon Rectum 63(11):1511–1523. https://doi.org/10.1097/dcr.0000000000001745
von Elm E, Altman DG, Egger M, Pocock SJ, Gotzsche PC, Vandenbroucke JP, Initiative S (2007) The strengthening the reporting of observational studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Ann Intern Med 147(8):573–577. https://doi.org/10.7326/0003-4819-147-8-200710160-00010
Heald RJ, Ryall RD (1986) Recurrence and survival after total mesorectal excision for rectal cancer. Lancet 1(8496):1479–1482. https://doi.org/10.1016/s0140-6736(86)91510-2
AIOM (2019) Linee guida NEOPLASIE DEL RETTO E ANO. https://www.aiom.it/wp-content/uploads/2019/10/2019_LG_AIOM_Retto_ano.pdf. Accessed 10 Oct 2020
Dindo D, Demartines N, Clavien P-A (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240(2):205–213
Rahbari NN, Weitz J, Hohenberger W, Heald RJ, Moran B, Ulrich A, Holm T, Wong WD, Tiret E, Moriya Y, Laurberg S, den Dulk M, van de Velde C, Büchler MW (2010) Definition and grading of anastomotic leakage following anterior resection of the rectum: a proposal by the International Study Group of Rectal Cancer. Surgery 147(3):339–351. https://doi.org/10.1016/j.surg.2009.10.012
Amin MB, Greene FL, Edge SB, Compton CC, Gershenwald JE, Brookland RK, Meyer L, Gress DM, Byrd DR, Winchester DP (2017) The Eighth Edition AJCC Cancer Staging Manual: Continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA: A Cancer J Clinic 67 (2):93–99. https://doi.org/10.3322/caac.21388
Koedam TWA, Bootsma BT, Deijen CL, van de Brug T, Kazemier G, Cuesta MA, Fürst A, Lacy AM, Haglind E, Tuynman JB, Daams F, Bonjer HJ, group obotCCIs (2020) Oncological outcomes after anastomotic leakage after surgery for colon or rectal cancer: increased risk of local recurrence. Annal Surg Publish Ahead of Print. https://doi.org/10.1097/sla.0000000000003889
Kulu Y, Tarantio I, Warschkow R, Kny S, Schneider M, Schmied BM, Büchler MW, Ulrich A (2015) Anastomotic leakage is associated with impaired overall and disease-free survival after curative rectal cancer resection: a propensity score analysis. Ann Surg Oncol 22(6):2059–2067. https://doi.org/10.1245/s10434-014-4187-3
Garcia-Granero E, Navarro F, Cerdan Santacruz C, Frasson M, Garcia-Granero A, Marinello F, Flor-Lorente B, Espi A (2017) Individual surgeon is an independent risk factor for leak after double-stapled colorectal anastomosis: an institutional analysis of 800 patients. Surgery 162(5):1006–1016. https://doi.org/10.1016/j.surg.2017.05.023
Funding
Open access funding provided by Università degli Studi di Padova within the CRUI-CARE Agreement. This work was supported partially by the Italian Ministry of Health (Progetti ordinari di Ricerca Finalizzata, RF-2011–02349645). The study was endorsed by the Italian Society of Surgical Oncology and Italian Society of Colorectal Surgery. No external funding for this manuscript.
Author information
Authors and Affiliations
Contributions
QRB: conception and design of the study, acquisition, analysis and interpretation of data, drafting and revision of the article, and final approval. GP: acquisition, analysis and interpretation of data, drafting and revision of the article, and final approval. GS: conception and design, analysis of data, interpretations of data, revision of the article and final approval. AR: conception and design, analysis and interpretation of data, revision of the article, and final approval. SD: acquisition, analysis and interpretation of data, drafting and revision of the article, and final approval. GC: acquisition and interpretation of data, drafting and revision of the article, and final approval. CR: interpretation of data, revision of the article, and final approval. FB: acquisition of data, revision of the article, and final approval. DC: acquisition of data, revision of the article, and final approval. EJ: acquisition of data, revision of the article, and final approval. RL: acquisition of data, revision of the article, and final approval. CB: acquisition of data, revision of the article, and final approval. AA: acquisition of data, revision of the article, and final approval. FLT: acquisition of data, revision of the article, and final approval. CA: acquisition of data, revision of the article, and final approval. AI: acquisition of data, revision of the article, and final approval. TC: acquisition of data, revision of the article, and final approval. PDB: conception and design, data analysis, interpretation of data, statistical analysis, revision of the article, final approval.PD: conception, interpretation of data, revision of the article, and final approval. SP: conception and design, interpretation of data, revision of the article, final approval.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The preliminary results of the study were presented on March 18, 2021, at SSO 2021—International Conference on Surgical Cancer Care virtual meeting.
Paolo Delrio and Salvatore Pucciarelli are two last co-authors.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bao, Q.R., Pellino, G., Spolverato, G. et al. The impact of anastomotic leak on long-term oncological outcomes after low anterior resection for mid-low rectal cancer: extended follow-up of a randomised controlled trial. Int J Colorectal Dis 37, 1689–1698 (2022). https://doi.org/10.1007/s00384-022-04204-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-022-04204-9