Abstract
Purpose
There has been no comparative study on the clinical value of magnetic resonance tumor regression grade (mrTRG)1–2 and ycT0–1N0 for the prediction of ypT0–1N0 after concurrent chemoradiotherapy (CCRT) for rectal cancer. We compared the diagnostic performance between mrTRG1–2 and ycT0–1N0 for predicting ypT0–1N0 as a selection criterion for non-radical management after CCRT in locally advanced rectal cancer.
Methods
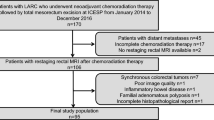
This retrospective study enrolled 291 patients from three referral hospitals between January 2018 and March 2020. The diagnostic performance of ycT0–1N0 and mrTRG1–2 for the prediction of ypT0–1N0 was compared in terms of sensitivity, specificity, positive-predictive value, negative-predictive value, and area under the curve (AUC).
Results
Sixty-eight patients (23.4%) achieved ypT0–1N0. Nineteen patients (6.5%) had ycT0–1N0, and 91 patients (31.2%) had mrTRG1–2. For predicting ypT0–1N0, ycT0–1N0 had a sensitivity of 16.2% (95% confidence interval [CI]: 8.36‒27.10) and positive-predictive value of 57.9% (95% CI: 36.57‒76.63), while mrTRG1–2 had a sensitivity of 58.8% (95% CI: 46.23‒70.63) and positive-predictive value of 44.0% (95% CI: 36.46‒51.74). When predicting ypT0–1N0, mrTRG1–2 showed a higher AUC (0.680, 95% CI: 0.604‒0.756) than ycT0–1N0 (0.563, 95% CI: 0.481‒0.645) (P < 0.001).
Conclusion
mrTRG1–2 might be a better indicator than ycT0–1N0 for the selection of non-radical management of advanced rectal cancer post-CCRT. However, additional diagnostic tools are required for predicting ypT0–1N0 because mrTRG1–2 or yc stage on MRI has insufficient evidence for diagnosing ypT0–1N0.



Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article (and/or) its supplementary materials.
Code availability
Not applicable.
References
Hoffmann KT, Rau B, Wust P, Stroszczynski C, Hünerbein M, Schneider U, Felix R (2002) Restaging of locally advanced carcinoma of the rectum with MR imaging after preoperative radio-chemotherapy plus regional hyperthermia. Strahlenther Onkol 178:386–392. https://doi.org/10.1007/s00066-002-0938-3
Guillem JG, Ruby JA, Leibold T, Akhurst TJ, Yeung HW, Gollub MJ, Ginsberg MS, Shia J, Suriawinata AA, Riedel ER, Mazumdar M, Saltz LB, Minsky BD, Nash GM, Paty PB, Temple LK, Weiser MR, Larson SM (2013) Neither FDG-PET Nor CT can distinguish between a pathological complete response and an incomplete response after neoadjuvant chemoradiation in locally advanced rectal cancer: a prospective study. Ann Surg 258:289–295. https://doi.org/10.1097/SLA.0b013e318277b625
Heo SH, Kim JW, Shin SS, Jeong YY, Kang HK (2014) Multimodal imaging evaluation in staging of rectal cancer. World J Gastroenterol 20:4244–4255. https://doi.org/10.3748/wjg.v20.i15.4244
Furey E, Jhaveri KS (2014) Magnetic resonance imaging in rectal cancer. Magn Reson Imaging Clin N Am 22:165–90, v, v–vi. https://doi.org/10.1016/j.mric.2014.01.004
Patel UB, Taylor F, Blomqvist L, George C, Evans H, Tekkis P, Quirke P, Sebag-Montefiore D, Moran B, Heald R, Guthrie A, Bees N, Swift I, Pennert K, Brown G (2011) Magnetic resonance imaging-detected tumor response for locally advanced rectal cancer predicts survival outcomes: mercury experience. J Clin Oncol 29:3753–3760. https://doi.org/10.1200/JCO.2011.34.9068
van Gijn W, Marijnen CA, Nagtegaal ID, Kranenbarg EM, Putter H, Wiggers T, Rutten HJ, Påhlman L, Glimelius B, van de Velde CJ, Dutch Colorectal Cancer Group (2011) Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer: 12-year follow-up of the multicentre, randomised controlled TME trial. Lancet Oncol 12:575–582. https://doi.org/10.1016/S1470-2045(11)70097-3
Park JH, Yoon SM, Yu CS, Kim JH, Kim TW, Kim JC (2011) Randomized phase 3 trial comparing preoperative and postoperative chemoradiotherapy with capecitabine for locally advanced rectal cancer. Cancer 117:3703–3712. https://doi.org/10.1002/cncr.25943
Bryant CLC, Lunniss PJ, Knowles CH, Thaha MA, Chan CLH (2012) Anterior resection syndrome. Lancet Oncol 13:e403–e408. https://doi.org/10.1016/S1470-2045(12)70236-X
Lindgren R, Hallböök O, Rutegård J, Sjödahl R, Matthiessen P (2011) What is the risk for a permanent stoma after low anterior resection of the rectum for cancer? A six-year follow-up of a multicenter trial. Dis Colon Rectum 54:41–47. https://doi.org/10.1007/DCR.0b013e3181fd2948
Fischer J, Eglinton TW, Richards SJ, Frizelle FA (2021) Predicting pathological response to chemoradiotherapy for rectal cancer: a systematic review. Expert Rev Anticancer Ther 21:489–500. https://doi.org/10.1080/14737140.2021.1868992
Fernandez LM, são Julião GP, Figueiredo NL, Beets GL, van der Valk MJM, Bahadoer RR, Hilling DE, Meershoek-Klein Kranenbarg E, Roodvoets AGH, Renehan AG, van de Velde CJH, Habr-Gama A, Perez RO, International Watch & Wait Database Consortium (2021) Conditional recurrence-free survival of clinical complete responders managed by watch and wait after neoadjuvant chemoradiotherapy for rectal cancer in the International Watch & Wait Database: a retrospective, international, multicentre registry study. Lancet Oncol 22:43–50. https://doi.org/10.1016/S1470-2045(20)30557-X
Lee JK, Cho JR, Song KS, Oh JH, Jeong SY, Kim MJ, Lee J, Kim MH, Oh HK, Kim DW, Kang SB; Seoul Colorectal Research Group (SECOG) Collaborative (2021) Oncologic comparison between nonradical management and total mesorectal excision in good responders after chemoradiotherapy in patients with mid-to-low rectal cancer. Ann Surg Treat Res 101:93–101. https://doi.org/10.4174/astr.2021.101.2.93
Fayaz MS, Demian GA, Fathallah WM, Eissa HE, El-Sherify MS, Abozlouf S, George T, Samir SM (2016) Significance of magnetic resonance imaging–assessed tumor response for locally advanced rectal cancer treated with preoperative long-course chemoradiation. J Glob Oncol 2:216–221. https://doi.org/10.1200/JGO.2015.001479
Spiros P, Hiotis SM, Cohen AM, Minsky BD, Paty PB, Guillem JG, Wagman R, Saltz LB, Wong WD (2002) Assessing the predictive value of clinical complete response to neoadjuvant therapy for rectal cancer: an analysis of 488 patients. J Am Coll Surg 194:131–135
Jang JK, Choi SH, Park SH, Kim KW, Kim HJ, Lee JS, Kim AY (2020) MR tumor regression grade for pathological complete response in rectal cancer post neoadjuvant chemoradiotherapy: a systematic review and meta-analysis for accuracy. Eur Radiol 30:2312–2323. https://doi.org/10.1007/s00330-019-06565-2
Cho MS, Kim H, Han YD, Hur H, Min BS, Baik SH, Cheon JH, Lim JS, Lee KY, Kim NK (2019) Endoscopy and magnetic resonance imaging-based prediction of ypT stage in patients with rectal cancer who received chemoradiotherapy: results from a prospective study of 110 patients. Med (Baltim) 98:e16614. https://doi.org/10.1097/MD.0000000000016614
Onaitis MW, Noone RB, Fields R, Hurwitz H, Morse M, Jowell P, McGrath K, Lee C, Anscher MS, Clary B, Mantyh C, Pappas TN, Ludwig K, Seigler HF, Tyler DS (2001) Complete response to neoadjuvant chemoradiation for rectal cancer does not influence survival. Ann Surg Oncol 8:801–806. https://doi.org/10.1007/s10434-001-0801-2
Nahas SC, Rizkallah Nahas CS, Sparapan Marques CF, Ribeiro U Jr, Cotti GC, Imperiale AR, Capareli FC, Chih Chen AT, Hoff PM, Cecconello I (2016) Pathologic complete response in rectal cancer: can we detect it? Lessons learned from a proposed randomized trial of watch-and-wait treatment of rectal cancer. Dis Colon Rectum 59:255–263. https://doi.org/10.1097/DCR.0000000000000558
Jeong SY, Park JW, Nam BH, Kim S, Kang SB, Lim SB, Choi HS, Kim DW, Chang HJ, Kim DY, Jung KH, Kim TY, Kang GH, Chie EK, Kim SY, Sohn DK, Kim DH, Kim JS, Lee HS, Kim JH, Oh JH (2014) Open versus laparoscopic surgery for mid-rectal or low-rectal cancer after neoadjuvant chemoradiotherapy (COREAN trial): survival outcomes of an open-label, non-inferiority, randomised controlled trial. Lancet Oncol 15:767–774. https://doi.org/10.1016/S1470-2045(14)70205-0
Moran B, Brown G, Cunningham D, Daniels I, Heald R, Quirke P, Sebag-Montefiore D (2008) Clarifying the TNM staging of rectal cancer in the context of modern imaging and neo-adjuvant treatment: ‘y’u′ and ‘p’ need ‘mr’ and ‘ct.’ Colorectal Dis 10:242–243. https://doi.org/10.1111/j.1463-1318.2007.01260.x
Horvat N, Rocha CCT, Oliveira BC, Petkovska I, Gollub MJ (2019) MRI of rectal cancer: tumor staging, imaging techniques, and management. Radiographics 39:367–387. https://doi.org/10.1148/rg.2019180114
Bujko K, Nowacki MP, Nasierowska-Guttmejer A, Kepka L, Winkler-Spytkowska B, Suwiński R, Oledzki J, Stryczyńska G, Wieczorek A, Serkies K, Rogowska D, Tokar P, Polish Colorectal Study Group (2005) Prediction of mesorectal nodal metastases after chemoradiation for rectal cancer: results of a randomised trial: implication for subsequent local excision. Radiother Oncol 76:234–240. https://doi.org/10.1016/j.radonc.2005.04.004
Kim DW, Kim DY, Kim TH, Jung KH, Chang HJ, Sohn DK, Lim SB, Choi HS, Jeong SY, Park JG (2006) Is T classification still correlated with lymph node status after preoperative chemoradiotherapy for rectal cancer? Cancer 106:1694–1700. https://doi.org/10.1002/cncr.21794
Nagtegaal ID, Glynne-Jones R (2020) How to measure tumour response in rectal cancer? An explanation of discrepancies and suggestions for improvement. Cancer Treat Rev 84:101964. https://doi.org/10.1016/j.ctrv.2020.101964
Author information
Authors and Affiliations
Contributions
Conceptualization and guided writing and performing study design: SBK, KJP, and JHO. Data collecting: HKO, DWK, MJK, SBR, SYJ, DWL, SCP, HCP, and HMA. Data analysis: JHL, JWS, and IJY. Data collecting and writing final manuscript: IJY.
Corresponding author
Ethics declarations
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, I.J., Suh, J.W., Lee, J. et al. Comparison of tumor regression grade and clinical stage based on MRI image as a selection criterion for non-radical management after concurrent chemoradiotherapy in locally advanced rectal cancer: a multicenter, retrospective, cross-sectional study. Int J Colorectal Dis 37, 1561–1568 (2022). https://doi.org/10.1007/s00384-022-04193-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-022-04193-9