Abstract
Background
The diagnostic accuracy of Magnetic Resonance Imaging (MRI) in restaging locally advanced rectal cancers (LARC) after neoadjuvant chemo-radio therapy (NCRT) has been under recent scrutiny. There is limited data on the accuracy of MRI and its timing in assessing tumor regression grade (TRG) and in identifying patients with complete response (CR). NCRT seems to cause tissue inflammation and oedema which renders reading the scans difficult for radiologist.
Aim
This study aims to assess the accuracy of MRI at different time intervals after NCRT in staging TRG and in identifying CR. Inter-observer agreement between 2 blinded radiologists will also be assessed.
Method
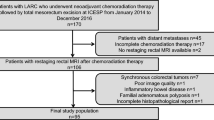
In this retrospective analysis, all patients diagnosed with LARC between January 2003 and 2014, who underwent long-course NCRT, who had at least one post-treatment MRI scan, and who underwent surgery with available pathology results are included. Histopathology staging is considered the reference standard. Accuracy of MRI in T staging and in TRG staging is assessed using weighted kappa. Accuracy, sensitivity, and specificity in identifying CR are calculated from a 2 × 2 contingency table. Inter-observer agreement between two-staging blinded radiologists is calculated using weighted kappa. These are calculated at 2 different time intervals after completion of NCRT.
Results
114 patients were identified who had a first post-treatment MRI scan at an average of 6.2 weeks after completion of NCRT. A subgroup of 68 patients had a second post-treatment MRI at an average of 10.4 weeks. Pathology results were available for 103 patients. By the second post-treatment scan, an additional 25% of patients experienced downstaging; accuracy in T staging increased from 43% to 57.4%; accuracy in TRG staging rose from 28.2% to 38.1%; accuracy in identifying CR rose from 83.4% to 84.1%. Inter-observer agreement in T staging rose from 0.1 for first post-treatment MRI to 0.206 for second post-treatment MRI.
Conclusion
This study advocates that restaging should occur at 10 weeks rather than the standard 6 weeks. This results in higher complete response rates and higher concordance with pathological specimens. Our results also showed that it is easier for radiologists to stage the MRI scans, resulting in higher inter-rater agreements.

Similar content being viewed by others
References
Brown G, Radcliffe AG, Newcombe RG, et al. (2003) Preoperative assessment of prognostic factors in rectal cancer using high-resolution magnetic resonance imaging. Br J Surg 90:355–364
MERCURY Study Group (2006) Diagnostic accuracy of preoperative magnetic resonance imaging in predicting curative resection of rectal cancer: prospective observational study. BMJ (Clinical Research ed.) 333:779
Sloothaak DA, Geijsen DE, van Leersum NI, et al. (2013) Dutch surgical colorectal audit. Optimal time interval between neoadjuvant chemoradiotherapy and surgery for rectal cancer. Br J Surg 100:933–939
Beets-Tan RG, Beets GL, Vliegen RF, et al. (2001) Accuracy of magnetic resonance imaging in prediction of tumour-free resection margin in rectal cancer surgery. Lancet (London, England) 357:497–504
Patel UB, Taylor F, Blomqvist L, et al. (2011) Magnetic resonance imaging-detected tumor response for locally advanced rectal cancer predicts survival outcomes: MERCURY experience. J Clin Oncol 29:3753–3760
Dworak O, Keilholz L, Hoffmann A (1997) Pathological features of rectal cancer after preoperative radiochemotherapy. Int J Colorectal Dis 12:19–23
Mandard AM, Dalibard F, Mandard JC, et al. (1994) Pathologic assessment of tumor regression after preoperative chemoradiotherapy of esophageal carcinoma. Clinicopathologic correlations. Cancer 73:2680–2686
Habr-Gama A, Perez R, Lynn P, São Julião G, Gama Rodrigues J (2012) Selective non-operative management of distal rectal cancer: the watch & wait protocol. In: Schiessel R, Metzger P (eds) Intersphincteric resection for low rectal tumors. Vienna: Springer, pp 43–53
Willis JE (2007) The pathologist’s role in rectal cancer patient assessments. Clin Colon Rectal Surg 20:158–166
Cohen J (1968) Weighted kappa: nominal scale agreement with provision for scaled disagreement or partial credit. Psychol Bull 70:213–220
Sclafani F, Brown G, Cunningham D, et al. (2017) Comparison between MRI and pathology in the assessment of tumour regression grade in rectal cancer. Br J Cancer. 117:1478
Shihab OC, Taylor F, Salerno G, et al. (2011) MRI predictive factors for long-term outcomes of low rectal tumours. Ann Surg Oncol 18:3278–3284
Johnston DF, Lawrence KM, Sizer BF, et al. (2009) Locally advanced rectal cancer: histopathological correlation and predictive accuracy of serial MRI after neoadjuvant chemotherapy. Br J Radiol 82:332–336
Kapiteijn E, Marijnen CA, Nagtegaal ID, et al. (2001) Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer. N Engl J Med 345:638–646
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
There was no funding related to this study.
Conflict of interest
All authors declare that there are no conflicts of interest to disclose related to this study.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was waived by our institution’s research and development department.
Rights and permissions
About this article
Cite this article
Aker, M., Boone, D., Chandramohan, A. et al. Diagnostic accuracy of MRI in assessing tumor regression and identifying complete response in patients with locally advanced rectal cancer after neoadjuvant treatment. Abdom Radiol 43, 3213–3219 (2018). https://doi.org/10.1007/s00261-018-1627-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-018-1627-8