Abstract
Purpose
High rates of early colorectal cancers (CRC) are observed in Tunisia suggesting genetic susceptibility. Nevertheless, up to now, no molecular study has been performed in the Tunisian population. In our research, we evaluated the clinical characteristics of Tunisian families suspected of Lynch syndrome and the contribution of DNA mismatch repair (MMR) genes.
Methods
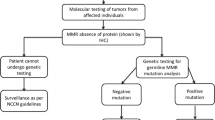
Thirty-one unrelated families suspected of Lynch syndrome were studied. Probands were tested for the presence of germline mutations in the MMR genes MLH1, MSH2, MSH6 and in MUTYH. Available tumours were analysed for microsatellite instability and expression of MMR proteins. Detailed family and medical histories were collected.
Results
A total of 134 cancers were noted in the 31 families, the most frequent type of cancer corresponding to CRC (69%), followed by uterine cancer (7.5%). Germline mutations were identified in 11 (35.5%) families (six MSH2, five MLH1, including seven novel mutations), seven of which fulfilled the Amsterdam criteria (sensitivity, 63.6%; positive predictive value, 58.3%). Noteworthy, germline mutations were detected in 52.6% of male patients tested, but in only 8.3% of females (p = 0.02). Moreover, CRC were essentially left sided in families without detected mutation (p = 0.017). Ages of onset of cancers and tumour spectrum were very similar in families with or without MMR germline mutation, contrasting with previous studies performed in other populations.
Conclusions
MMR genes contribute significantly to CRC susceptibility in the Tunisian population. However, the cause of early CRC susceptibility remains unknown in most cases, especially in women and in patients with early left colon or rectal cancer.


Similar content being viewed by others
Abbreviations
- CRC:
-
Colorectal cancer
- HNPCC:
-
Hereditary nonpolyposis colorectal cancer
- MLPA:
-
Multiplex ligation dependent probe amplification
- MMR:
-
Mismatch repair
- MSI:
-
Microsatellite instability
- MSI-H:
-
Microsatellite instability-high
- MSS:
-
Microsatellite stable
References
Ben Abdalla M (1998) Registre des cancers Nord-Tunisie 1995–1998
Salovaara R, Loukola A, Jristo P, Kääriäinen H, Ahtola H, Eskelinen M, Härkönen N, Julkunen R, Kangas E, Ojala S, Tulikoura J, Valkano E, Järvinen H, Mecklin JP, Aaltonen LA, de La Chapelle A (2000) Population-based molecular detection of HNPCC. J Clin Oncol 18:2193–2200
Lynch HT, de la Chapelle A (2003) Hereditary colorectal cancer. N Engl J Med 348:919–932
Hampel H, Frankel WL, Martin E, Arnold M, Khanduja K, Kuebler P, Clendenning M, Sotamaa K, Prior T, Westman JA, Panescu J, Fix D, Lockman J, LaJeunesse J, Comeras I, de la Chapelle A (2008) Feasibility of screening for lynch syndrome among patients with colorectal cancer. J Clin Oncol 26:5783–5788
Hampel H, Frankel WL, Martin E, Arnold M, Khanduja K, Kuebler P, Nakagawa H, Sotamaa K, Prior TW, Westman J, Panescu J, Fix D, Lockman J, Comeras I, de la Chapelle A (2005) Screening for the Lynch syndrome (hereditary nonpolyposis colorectal cancer). N Engl J Med 352:1851–1860
Vasen HF, Möslein G, Alonso A, Bernstein I, Bertario L, Blanco I, Burn J, Capella G, Engel C, Frayling I, Friedl W, Hes FJ, Hodgson S, Mecklin JP, Møller P, Nagengast F, Parc Y, Renkonen-Sinisalo L, Sampson JR, Stormorken A, Wijnen J (2007) Guidelines for the clinical management of Lynch syndrome (hereditary non-polyposis cancer). J Med Genet 44:353–362
Aaltonen LA, Peltomäki P, Mecklin JP, Järvinen H, Jass JR, Green JS, Lynch HT, Watson P, Tallqvist G, Juhola M, Sistonen P, Hamilton SR, Kinzler KW, Vogelstein B, de la Chapelle A (1994) Replication errors in benign and malignant tumours from hereditary nonpolyposis colorectal cancer patients. Cancer Res 54:1645–1648
Boland CR, Thibodeau SN, Hamilton SR, Sidransky D, Eshleman JR, Burt RW, Meltzer SJ, Rodriguez-Bigas MR, Fodde R, Ranzani GN, Srivastava S (1998) A National Cancer Institute Workshop on Microsatellite Instability for cancer detection and familial predisposition: development of international criteria for the determination of microsatellite instability in colorectal cancer. Cancer Res 8:5248–5257
Cunningham JM, Christensen ER, Tester DJ, Kim CY, Roche PC, Burgart LJ, Thibodeau SN (1998) Hypermethylation of the hMLH1 promoter in colon cancer with microsatellite instability. Cancer Res 58:3455–3460
Veigl ML, Kasturi L, Olechnowicz J, Ma AH, Lutterbaugh JD, Periyasamy S, Li GM, Drummond J, Modrich PL, Sedwick WD, Markowitz SD (1998) Biallelic inactivation of hMLH1 by epigenetic gene silencing, a novel mechanism causing human MSI cancers. Proc Natl Acad Sci USA 95:8698–8702
Vasen HF, Mecklin JP, Khan PM, Lynch HT (1991) The International Collaborative Group on Hereditary Non-Polyposis Colorectal Cancer (ICG-HNPCC). Dis Colon Rectum 34:424–425
Vasen HF, Watson P, Mecklin JP, Lynch HT (1999) New clinical criteria for hereditary nonpolyposis colorectal cancer (HNPCC, Lynch syndrome) proposed by the International Collaborative group on HNPCC. Gastroenterology 116:1453–1456
Rodriguez-Bigas MA, Boland CR, Hamilton SR, Henson DE, Jass JR, Khan PM, Lynch HT, Perucho M, Smyrk T, Sobin L, Srivasta SA (1997) A national cancer institute workshop on hereditary nonpolyposis colorectal cancer syndrome: meeting highlights and Bethesda guidelines. J Natl Cancer Inst 89:1758–1762
Umar A, Boland CR, Terdiman JP, Syngal S, de la Chapelle A, Rüschoff J, Fishel R, Lindor NM, Burgart LJ, Hamelin R, Hamilton SR, Hiatt RA, Jass J, Lindblom A, Lynch HT, Peltomaki P, Ramsey SD, Rodriguez-Bigas MA, Vasen HF, Hawk ET, Barrett JC, Freedman AN, Srivastava S (2004) Revised Bethesda guidelines for hereditary nonpolyposis colorectal cancer (Lynch syndrome) and microsatellite instability. J Natl Cancer Inst 96:261–268
Luo DC, Cai Q, Sun MH, Ni YZ, Ni SC, Chen ZJ, Li XY, Tao CW, Zhang XM, Shi DR (2005) Clinicopathological and molecular genetic analysis of HNPCC in China. World J Gastroenterol 11:1673–1679
Wang J, Luo MH, Zhang ZX, Zhang PD, Jiang XL, Ma DW, Suo RZ, Zhao LZ, Qi QH (2007) Clinical and molecular analysis of hereditary non-polyposis colorectal cancer in Chinese colorectal cancer patients. World J Gastroenterol 13:1612–1617
Nyström-Lahti M, Wu Y, Moisio AL, Hostra RMW, Osinga J, Mecklin JP, Järvinen HJ, Leisti J, Buys CHCM, de La Chapelle A, Peltomäki P (1996) DNA mismatch repair gene mutations in 55 kindreds with verified or putative hereditary non-polyposis colorectal cancer. Hum Mol Genet 5:763–769
Suraweera N, Duval A, Reperant M, Vaury C, Furlan D, Leroy K, Seruca R, Iacopetta B, Hamelin R (2002) Evaluation of tumour microsatellite instability using five quasimonomorphic mononucleotide repeats and pentaplex PCR. Gastroenterology 123:1804–1811
Aissi-Ben Moussa S, Moussa A, Lovecchio T, Kourda N, Najjar T, Ben Jilani S, El Gaaied A, Porchet N, Manai M, Buisine MP (2008) Identification and characterization of a novel MLH1 genomic rearrangement as the cause of HNPCC in a Tunisian family: evidence for a homologous Alu-mediated recombination. Fam Cancer 8:119–126
Al-Tassan N, Chmiel NH, Maynard J, Fleming N, Livingston AL, Williams GT, Hodges AK, Davies DR, David SS, Sampson JR, Cheadle JP (2002) Inherited variants of MYH associated with somatic G:C–>T:A mutations in colorectal tumors. Nat Genet 30:227–232
Fleischmann PJ, Cheadle J, Shah B, Sampson J, Houlston RS (2004) Comprehensive analysis of the contribution of germline MYH variation to early-onset colorectal cancer. Int J Cancer 109:554–558
Wang L, Baudhuin LM, Boardman LA, Steenblock KJ, Petersen GM, Halling KC, French AJ, Johnson RA, Burgart LJ, Rabe K, Lindor NM, Thibodeau SN (2004) MYH mutations in patients with attenuated and classic polyposis and with young-onset colorectal cancer without polyps. Gastroenterology 127:9–16
Croitoru ME, Cleary SP, Di Nicola N, Manno M, Selander T, Aronson M, Redston M, Cotterchio M, Knight J, Gryfe R, Gallinger S (2004) Association between biallelic and monoallelic germline MYH gene mutations and colorectal cancer risk. J Natl Cancer Inst 96:1631–1634
Lejeune S, Guillemot F, Triboulet JP, Cattan S, Mouton C, Porchet N, Manouvrier S, Buisine MP (2006) Low frequency of AXIN2 mutations and high frequency of MUTYH mutations in patients with multiple polyposis. Hum Mutat 27:1064
Park JG, Kim DW, Hong CW, Nam BH, Shin YK, Hong SH, Kim IJ, Lim SB, Aronson M, Bisgaard ML, Brown GJ, Burn J, Chow E, Conrad P, Douglas F, Dunlop M, Ford J, Greenblatt MS, Heikki J, Heinimann K, Lynch EL, Macrae F, McKinnon WC, Möeslein G, Rossi BM, Rozen P, Schofield L, Vaccaro C, Vasen H, Velthuizen M, Viel A, Wijnen J (2006) Germ line mutations of mismatch repair genes in hereditary nonpolyposis colorectal cancer patients with small bowel cancer: International Society for Gastrointestinal Hereditary Tumours Collaborative Study. Clin Cancer Res 12:3389–3393
Wijnen J, Khan PM, Vasen H, Menko F, Van der Klift H, Van dan Broek M, Van Leeuwen-Cornelisse I, Nagengast F, Meijers-Heijboer EJ, Lindhout D, Griffioen G, Cats A, Kleibeuker J, Varesco L, Bertario L, Bisgaard ML, Mohr J, Kolodner R, Fodde R (1996) Majority of hMLH1 mutations responsible for hereditary nonpolyposis colorectal cancer cluster at the exonic region 15–16. Am J Hum Genet 58:300–307
Jacob S, Praz F (2002) DNA mismatch repair defects: role in colorectal carcinogenesis. Biochimie 84:27–47
Kondo E, Horii A, Fukushige S (2001) The interacting domains of three MutL heterodimers in man: hMLH1 interacts with 36 homologous amino acid residues within hMLH3, hPMS1 and hPMS2. Nucleic Acids Res 29:1695–1702
Wimmer K, Etzler J (2008) Constitutional mismatch repair-deficiency syndrome: have we so far seen only the tip of an iceberg? Hum Genet 124:105–122
Wheeler JM, Loukola A, Aaltonen LA, Mortensen NJ, Bodmer WF (2002) The role of hypermethylation of the hMLH1 promoter region in HNPCC versus MSI + sporadic colorectal cancers. J Med Genet 37:588–592
Menigatti M, Di Gregorio C, Borghi F, Sala E, Scarselli A, Pedroni M, Foroni M, Benatti P, Roncucci L, Ponz de Leon M, Percesepe A (2001) Methylation pattern of different regions of the MLH1 promoter and silencing of gene expression in hereditary and sporadic colorectal cancer. Genes Chromosomes Cancer 31:357–361
Bettstetter M, Dechant S, Ruemmele P, Grabowski M, Keller G, Holinski-Feder E, Hartmann A, Hofstaedter F, Dietmaier W (2007) Distinction of hereditary nonpolyposis colorectal cancer and sporadic microsatellite-unstable colorectal cancer through quantification of MLH1 methylation by real-time PCR. Clin Cancer Res 13:3221–3228
Takahashi M, Shimodaira H, Andreutti-Zaugg C, Iggo R, Kolodner RD, Ishioka C (2007) Functional analysis of human MLH1 variants using yeast and in vitro mismatch repair assays. Cancer Res 67:4595–4604
Kowalski LD, Mutch DG, Herzog TJ, Rader JS, Goodfellow PJ (1997) Mutational analysis of MLH1 and MSH2 in 25 prospectively-acquired RER+ endometrial cancers. Genes Chromosomes Cancer 18:219–227
Weber TK, Chin HM, Rodriguez-Bigas M, Keitz B, Gilligan R, O’Malley L, Urf E, Diba N, Pazik J, Petrelli NJ (1999) Novel hMLH1 and hMSH2 germline mutations in African Americans with colorectal cancer. JAMA 281:2316–2320
Järvinen HJ, Aarnio M, Mustonen H, Aktan-Collan K, Aaltonen LA, Peltomäki P, de La Chapelle A, Mecklin JP (2000) Controlled 15-year trial on screening for colorectal cancer in families with hereditary nonpolyposis colorectal cancer. Gastroenterology 118:829–834
Southey MC, Jenkins MA, Mead L, Whitty J, Trivett M, Tesoriero AA, Smith LD, Jennings K, Grubb G, Royce SG, Walsh MD, Barker MA, Young JP, Jass JR, St John DJ, Macrae FA, Giles GG, Hopper JL (2005) Use of molecular tumour characteristics to prioritize mismatch repair gene testing in early-onset colorectal cancer. J Clin Oncol 23:6524–6532
Kievit W, de Bruin JH, Adang EM, Ligtenberg MJ, Nagengast FM, van Krieken JH, Hoogerbrugge N (2004) Current clinical selection strategies for identification of hereditary non-polyposis colorectal cancer families are inadequate: a meta-analysis. Clin Genet 65:308–316
Lipton LR, Johnson V, Cummings C, Fisher S, Risby P, Eftekhar Sadat AT, Cranston T, Izatt L, Sasieni P, Hodgson SV, Thomas HJ, Tomlinson IP (2004) Refining the Amsterdam criteria and Bethesda guidelines: testing algorithms for the prediction of mismatch repair mutation status in the familial cancer clinic. J Clin Oncol 22:4934–4943
Balmaña J, Balaguer F, Castellví-Bel S, Steyerberg EW, Andreu M, Llor X, Jover R, Castells A, Syngal S (2008) Comparison of predictive models, clinical criteria and molecular tumour screening for the identification of patients with Lynch syndrome in a population-based cohort of colorectal cancer patients. J Med Genet 45:557–563
Syngal S, Fox EA, Eng C, Kolodner RD, Garber JE (2000) Sensitivity and specificity of clinical criteria for hereditary non-polyposis colorectal cancer associated mutations in MSH2 and MLH1. J Med Genet 37:641–645
Koch-Muller Y, Vogelsang H, Kopp R, Lohse P, Keller G, Aust D, Muders M, Gross M, Daum J, Schiemann U, Grabowski M, Scholz M, Kerker B, Becker I, Henke G, Holinski-Feder E (2005) Hereditary non-polyposis colorectal cancer: clinical and molecular evidence for a new entity of hereditary colorectal cancer. Gut 54:1733–1740
Lindor NM, Rabe K, Petersen GM, Haile R, Casey G, Baron J, Callinger S, Bapat B, Aronson M, Hopper J, Jass J, Le Marchand L, Grove J, Potter J, Newcomb P, Terdiman JP, Conrad P, Moslein G, Goldberg R, Ziogas A, Anton-Culver H, de Andrade M, Siegmund M, Thibodeau SN, Boardman LA, Seminara D (2005) Lower cancer incidence in Amsterdam-I criteria families without mismatch repair deficiency:familial colorectal cancer type X. JAMA 293:1979–1985
Llor X, Pons E, Xicola RM, Castells A, Alenda C, Piñol V, Andreu M, Castellví-Bel S, Payá A, Jover R, Bessa X, Girós A, Roca A, Gassull MA (2005) Differential features of colorectal cancers fulfilling Amsterdam criteria without involvement of the mutator pathway. Clin Cancer Res 11:7304–7310
Vasen HFA, Stormorken A, Menko FH, Nagengast FM, Kleibeuker JH, Griffioen G, Taal BG, Moller P, Wijnen JT (2001) MSH2 mutation carriers are at higher risk of cancer than MLH1 mutation carriers: a study of hereditary nonpolyposis colorectal cancer families. J Clin Oncol 19:4074–4080
Acknowledgements
This study was the result of collaboration between the Universities of Tunis-El Manar and Lille Nord de France. S.A.B. was supported in part by a grant from the Tunisian government. We are indebted to all family members who agreed to attend our study. We also thank Dr O. Trabelsi, gastroenterologist in Tunis, Tunisia for providing us with families, Dr A. Wacrenier, Department of Pathology of the CHRU of Lille, France, for her help in immunohistochemistry of MMR proteins, Dr M. Crépin, Platform of Molecular Biology of the CHRU of Lille, France, and S. Aissi for revision of the text.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moussa, S.AB., Moussa, A., Kourda, N. et al. Lynch syndrome in Tunisia: first description of clinical features and germline mutations. Int J Colorectal Dis 26, 455–467 (2011). https://doi.org/10.1007/s00384-010-1129-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-010-1129-9